HNMR Without Integrations
In this tutorial I want to show you how to tackle 1H NMR problems when the integration values are missing. Not having integrations means you do not immediately know how many hydrogens correspond to each signal, and that can feel unsettling. The good news is that all such questions fall into two categories, and each category has a clear strategy that you can rely on.
Example 1
The first category involves matching structures to spectra.
These questions can appear in different forms. Sometimes you get several structures and one spectrum. Sometimes you get several spectra and one structure. The format does not matter. The approach is always the same. You begin by examining the structures and predicting the rough layout of the spectra they should produce.
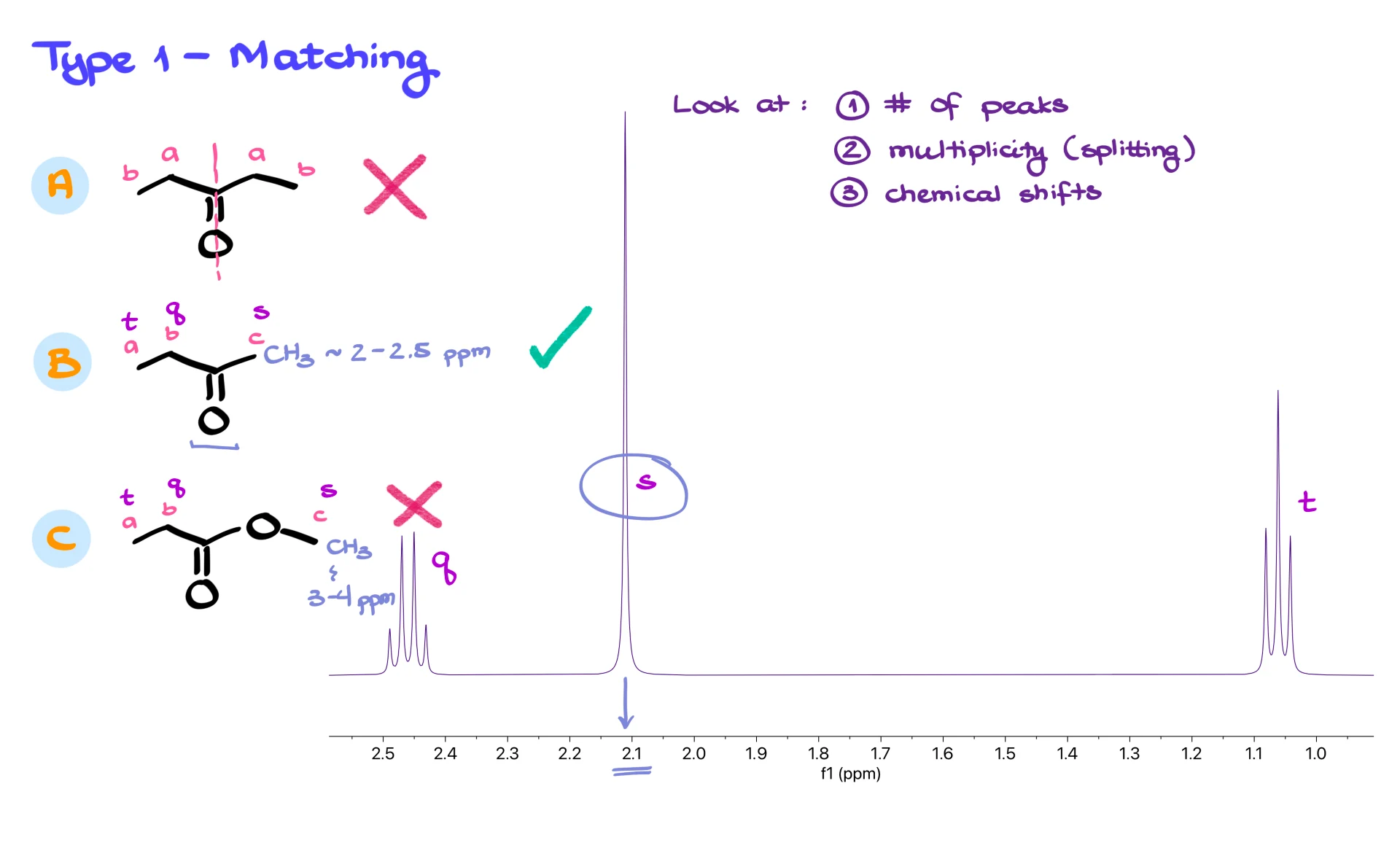
For example, in the first structure, molecule A, you can spot an internal plane of symmetry. That symmetry collapses several hydrogens into equivalent environments, so the molecule only produces two signals. If the spectrum shows three signals, then molecule A cannot match it. Molecule B produces three distinct signals, which we can call (a), (b) and (c). Molecule C also produces three signals. Because both structures give the correct number of peaks, we move on to splitting.
Molecule B should give a triplet, a quartet and a singlet. Molecule C should give the same pattern. If the spectrum contains a triplet, a quartet and a singlet, we still cannot distinguish the two. This is when chemical shifts become the deciding factor. A CH3 group next to a carbonyl typically appears around 2.0 to 2.5 ppm. A CH3 group sitting next to an oxygen appears further downfield, at about 3.0 to 4.0 ppm. In our spectrum the singlet sits at about 2.1 ppm, which matches a CH3 group next to a carbonyl. That immediately eliminates molecule C, leaving molecule B as the correct match.
So when you do not have integrations and you need to match structures to spectra, the workflow is simple:
- check the number of expected signals,
- examine the multiplicity, and finally
- use chemical shifts to distinguish any remaining options.
One of those three steps will always be enough to identify the correct structure.
Example 2
The second type of question is more involved. Here you must determine the complete structure from a molecular formula and an NMR spectrum without integrations. Sometimes the problem also includes IR or other data, but often 1H NMR and the formula are all you get. These problems seem intimidating, but they become manageable when you treat the spectrum as a collection of clues.

The first clue is the molecular formula. From that we calculate the hydrogen deficiency index. I like the version of the formula where we take twice the number of carbons plus two, subtract the number of hydrogens, subtract any halogens, add any nitrogens and ignore oxygens, then divide by two. In the example shown, with C11H14O3, we compute (2×11 + 2 − 14) ÷ 2 = 5. Five degrees of unsaturation usually signal the presence of an aromatic ring because an aromatic ring accounts for four. And our spectrum confirms this because we see signals in the aromatic region. The expansion of that region shows two symmetrical doublets, a classic pattern for a para-substituted benzene ring.
The next striking signal is the set of peaks that form a septet. A septet means the hydrogen is coupled to six equivalent neighbors. Nearby we see a doublet, which is a typical pattern for the two CH3 groups in an isopropyl fragment. So we can identify an isopropyl group in our molecule. We also see a strong singlet at about 3.75 ppm. A singlet at that position is very commonly a CH3 group attached to an oxygen, so we can tentatively mark an OCH3 fragment.
At this point we list the fragments: the para-substituted aromatic ring, the methoxy group and the isopropyl group. We subtract these from the molecular formula to see what is left. Removing C6H4, then CH3O, and then C3H7 leaves one carbon and two oxygens that we have not placed. We also still have one degree of unsaturation unaccounted for. One additional degree and one carbon with two oxygens strongly suggest an ester. Putting all the building blocks together gives a structure that matches both the splitting patterns and the chemical shifts of the full spectrum.
Example 3
The next example follows the same method. From C12H17NO2 we again obtain a hydrogen deficiency index of 5, suggesting an aromatic ring. The aromatic region shows four distinct signals: a triplet, a doublet, a singlet and another doublet. That pattern is characteristic of a meta-substituted benzene ring. The spectrum also contains a quartet and a triplet, which is the unmistakable signature of an ethyl group. The quartet is shifted downfield to about 4 ppm, meaning that the CH2 group is attached to an oxygen. After removing the ring and the ethoxy fragment from the molecular formula, we examine the remaining signals. We find two singlets. One is small and the other is noticeably larger. The larger singlet must represent several equivalent hydrogens, most likely two identical CH3 groups. The smaller singlet can represent a CH2 fragment with no neighbors.
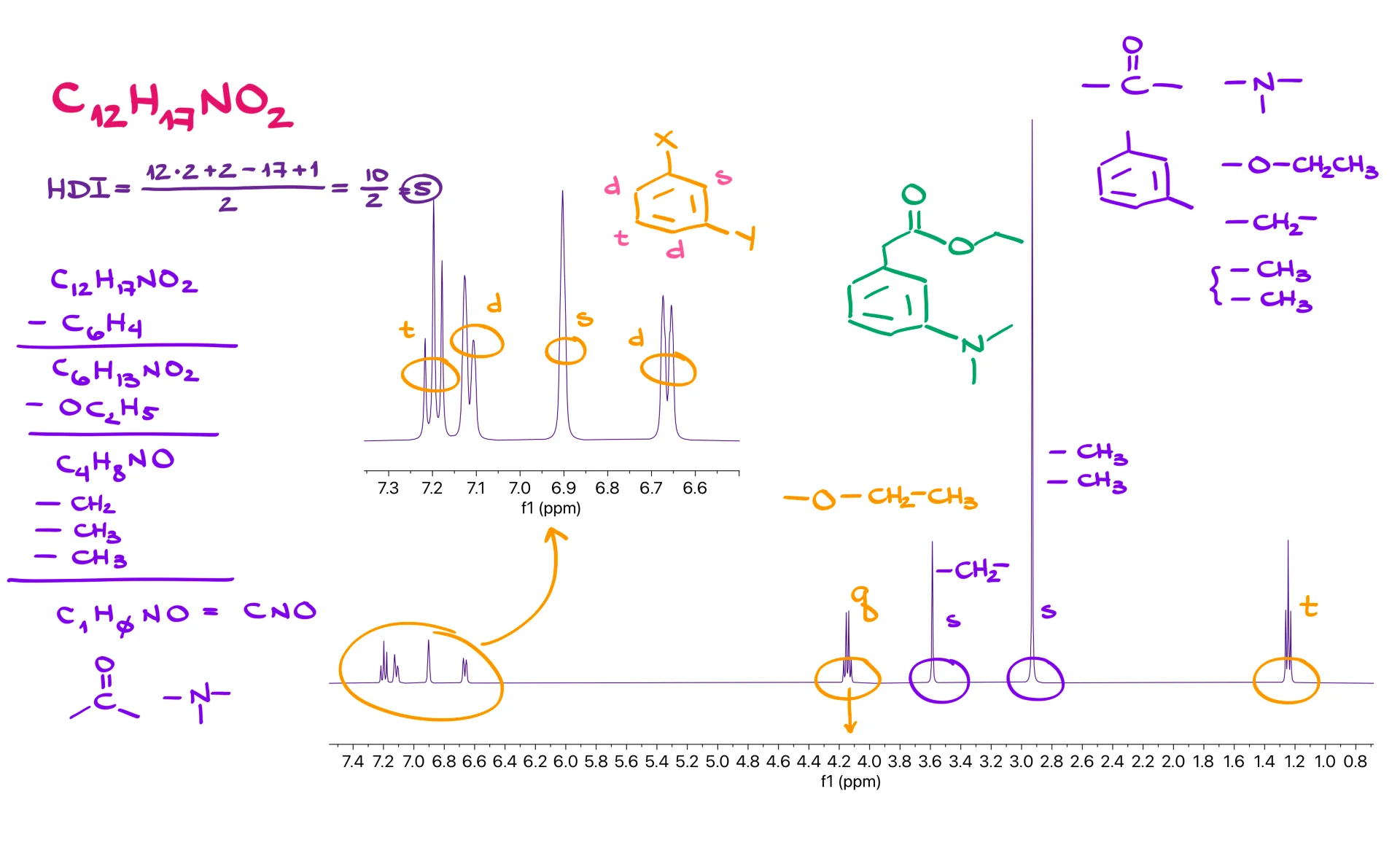
Subtracting CH2 and two CH3 groups leaves one carbon, one nitrogen and one oxygen. Combined with the remaining degree of unsaturation, we conclude that we must have a carbonyl group. When we assemble all the pieces, including the carbonyl and the nitrogen, we obtain a structure that agrees with both splitting patterns and chemical shifts.
These types of problems are challenging without integrations, but they become approachable once you break them down into fragments and test your assumptions. The more practice you get, the better your intuition becomes, and soon these puzzles start to feel more like enjoyable logic problems than roadblocks.
