Weinreb Amides in Ketone and Aldehyde Synthesis
A Weinreb amide is a special type of amide that serves as a very important intermediate in organic synthesis. In this tutorial, we are going to look at what makes Weinreb amides unique and how we can use them to make aldehydes and ketones.
Synthesis of Weinreb Amides
Let’s start with how we make Weinreb amides.
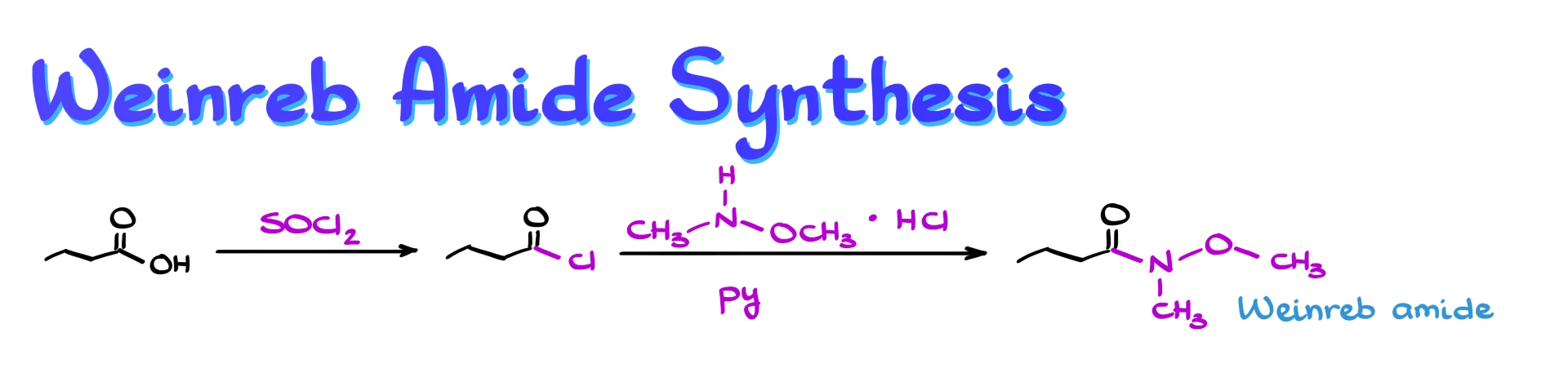
Typically, we begin with a carboxylic acid and convert it into the corresponding acid chloride, usually using a reagent like thionyl chloride. For example, if we start with butanoic acid, we would form butanoyl chloride in the first step.
Next, we treat the acid chloride with a special amine, N-methyl-O-methylhydroxylamine (or N-methoxy-N-methylamine, if you like), where the nitrogen is bonded to both a methyl group and a methoxy group. This reagent is often commercially available as its hydrochloride salt, so we usually add a base like pyridine to neutralize it during the reaction.
As a result, we obtain the Weinreb amide.
Now, let’s briefly look at the mechanism of this last step, converting the acid chloride into the Weinreb amide.
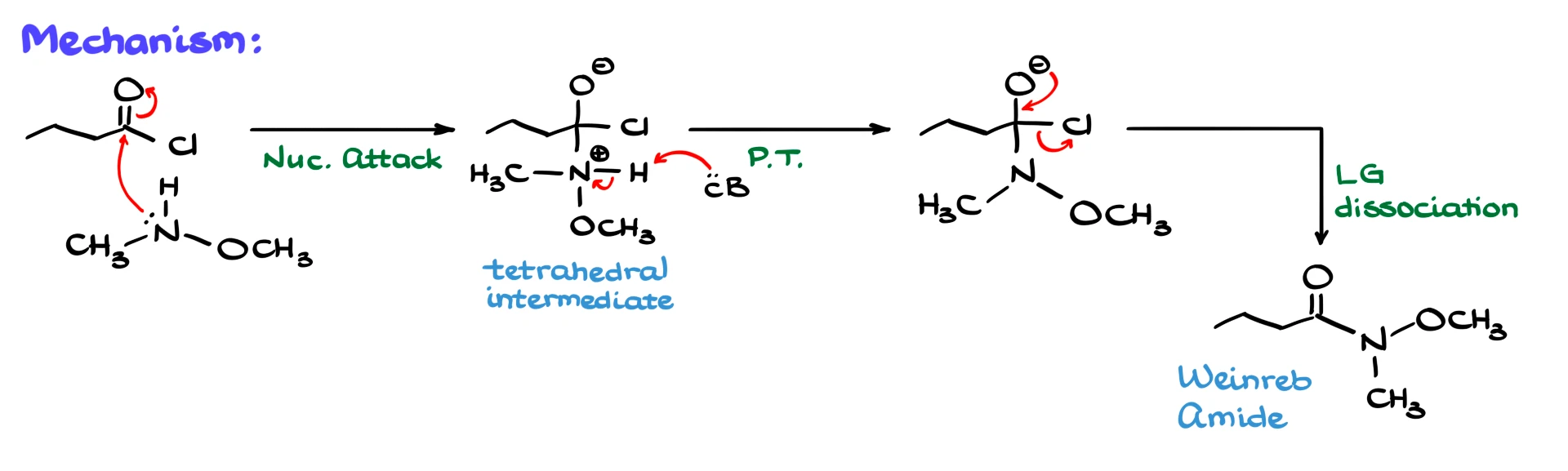
We start with the acid chloride and the neutral amine. Since nitrogen is nucleophilic, the first step is a nucleophilic attack on the carbonyl carbon, forming a tetrahedral intermediate.
From here, two things need to happen. We need to remove the proton from nitrogen, and we need to expel the chloride. A base, such as pyridine or another equivalent of the amine, removes the proton from nitrogen, giving a neutral intermediate.
Then, the electrons collapse back down, kicking out the chloride as a leaving group and forming the Weinreb amide.
As with many reactions involving carboxylic acid derivatives, the exact order of proton transfer versus leaving group departure can vary depending on how the mechanism is presented. I typically show proton transfer first, but you should always follow your instructor’s preferred version.
Now that we have our Weinreb amide, what can we do with it?
Ketone Synthesis Using Weinreb Amides
One of the most important applications is ketone synthesis.
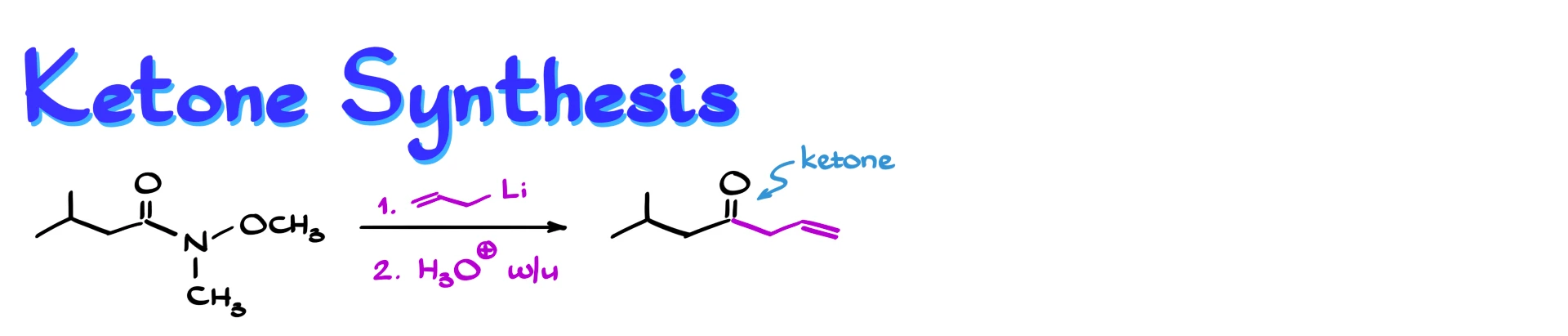
If we treat a Weinreb amide with an organometallic reagent, such as an organolithium compound or a Grignard reagent, we obtain a ketone. This is a major advantage over other carboxylic acid derivatives. For example, if we use an acid chloride or an ester with organometallic reagents, we often get overalkylation, meaning the reaction continues past the ketone stage to give a tertiary alcohol.
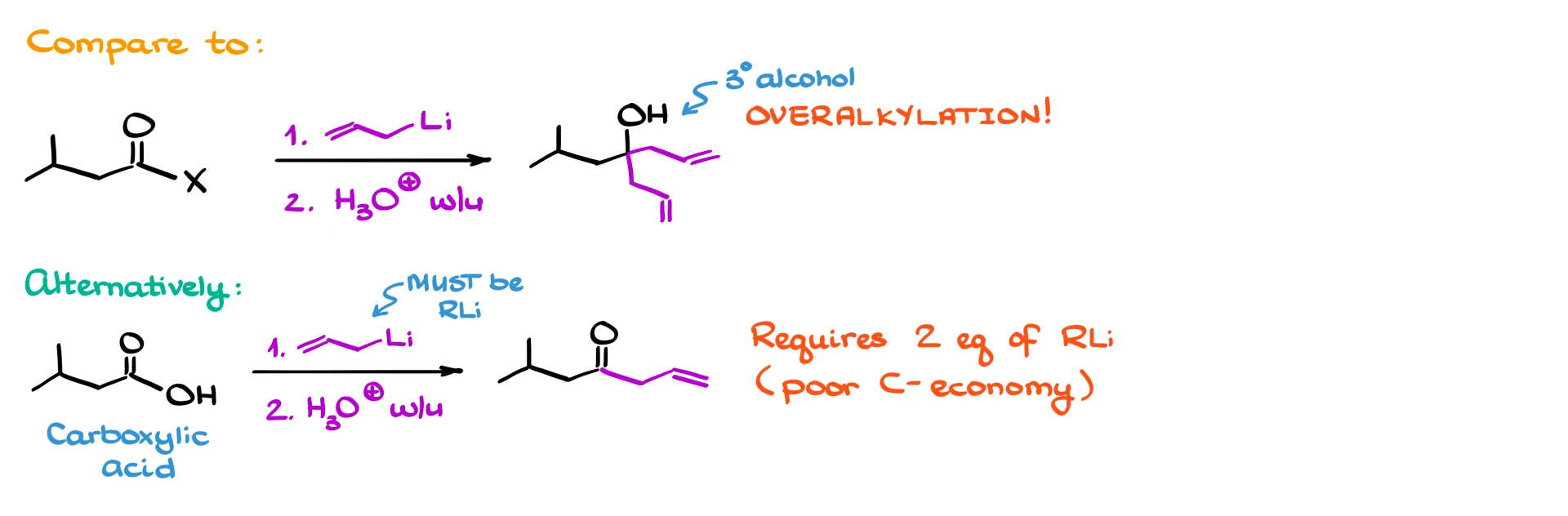
We could also form ketones from carboxylic acids using organolithium reagents, but that typically requires two equivalents of the reagent, which is wasteful, especially if the reagent is expensive. This is where Weinreb amides really shine.
Let’s look at why this works mechanistically.
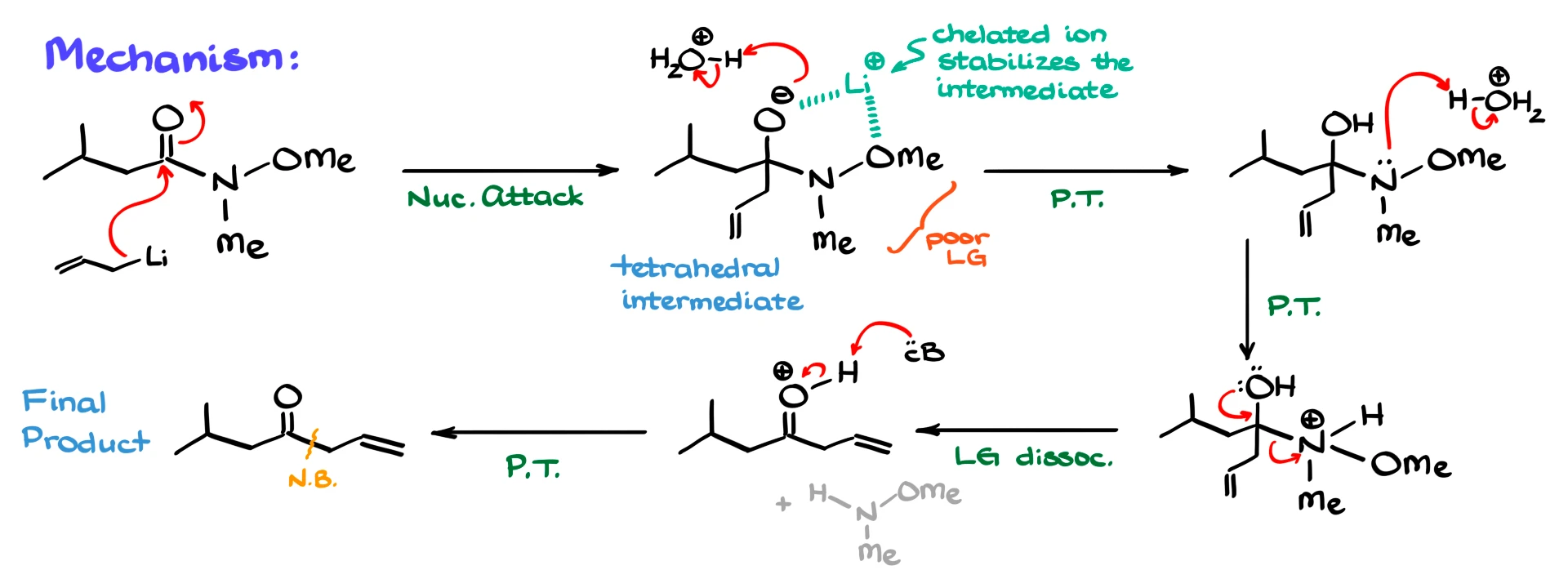
First, the organometallic reagent attacks the carbonyl carbon of the Weinreb amide, forming a tetrahedral intermediate. Normally, we would expect this intermediate to collapse and expel the leaving group, but in this case, that does not happen.
There are two key reasons. First, the amide fragment is a poor leaving group, making it difficult to expel. Second, and more importantly, the metal ion, lithium or magnesium, coordinates with both oxygen atoms, the former carbonyl oxygen and the methoxy oxygen. This creates a chelated complex that stabilizes the tetrahedral intermediate.
Because of this stabilization, the intermediate does not collapse further, and the reaction effectively stops at this stage.
During the acidic workup, the oxygen is protonated, and then the nitrogen is protonated to turn it into a good leaving group. The intermediate then collapses, expelling the amine fragment and forming a protonated ketone. Finally, deprotonation gives the neutral ketone.
So the key idea is that the unique stabilization of the intermediate prevents overreaction, allowing us to stop cleanly at the ketone stage.
Aldehyde Synthesis Using Weinreb Amides
We can also use Weinreb amides to make aldehydes.
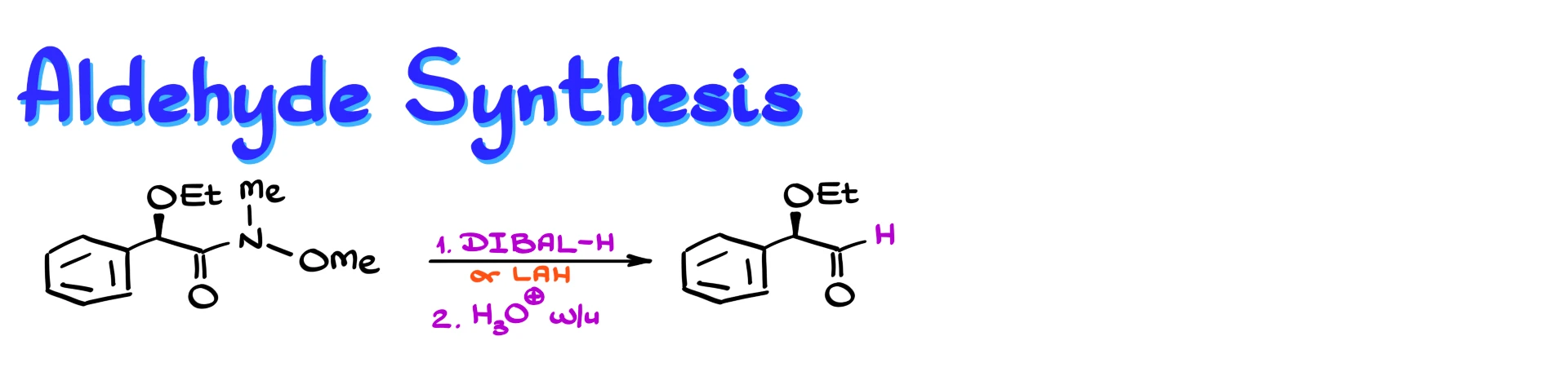
If we treat a Weinreb amide with a hydride reagent such as DIBAL-H or even lithium aluminum hydride, followed by acidic workup, we obtain an aldehyde.
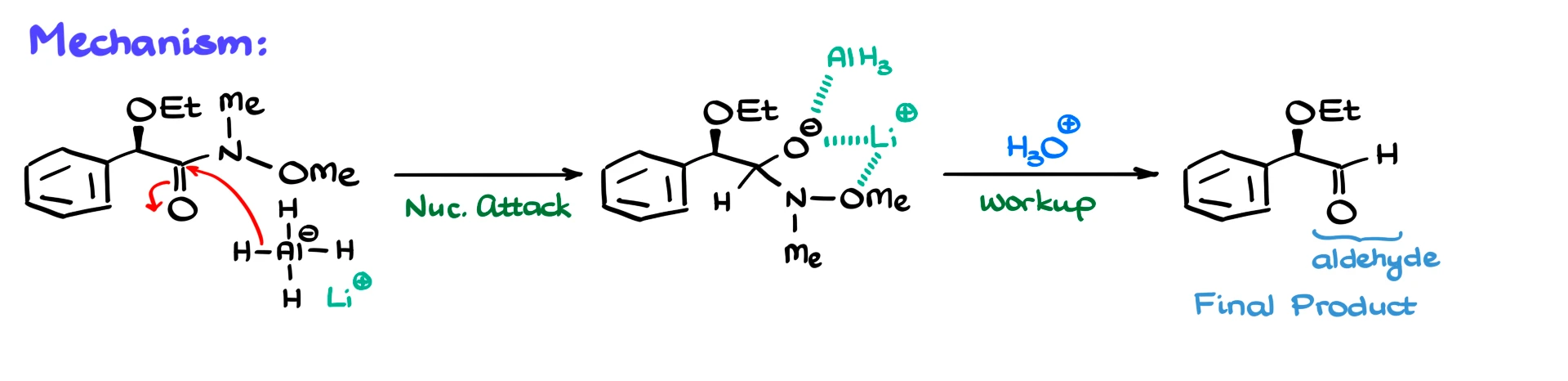
The mechanism starts with hydride attack on the carbonyl carbon, forming a tetrahedral intermediate. Just like before, lithium and aluminum coordinate with the oxygen atoms, stabilizing the intermediate and preventing further reduction.
Because of this stabilization, we do not get overreduction to the alcohol, which would normally happen with strong hydride reagents.
After acidic workup, the intermediate collapses, and we obtain the aldehyde.
So even though Weinreb amides are not a universal tool for every reaction, they are incredibly valuable for selectively forming ketones and aldehydes from carboxylic acid derivatives.
Because of this unique reactivity and control, Weinreb amides are an essential tool in synthetic organic chemistry, especially when planning multistep syntheses.
