40. Synthesis of a Complex Tri-Substituted Aromatic Compound

The first thing that stands out in our target molecule is the enamine functional group. Enamines come from carbonyl compounds and secondary amines, so the very last step in this synthesis will be an enamine formation. That lets us identify the corresponding carbonyl precursor immediately.
Next, the aromatic ring carries three different substituents: a chlorine, a nitro group, and an acyl group that is the carbonyl needed for the enamine step. Chlorine can be introduced through a halogenation, the nitro group through nitration with concentrated nitric and sulfuric acids, and the acyl group through a Friedel–Crafts acylation using an acyl chloride and a Lewis acid such as AlCl3.
All three substituents influence the reactivity of the ring in different ways. Chlorine is a deactivating ortho-para director. The nitro group is a strongly deactivating meta director. The acyl group is also a deactivating meta director. And this is where reaction limitations matter. Friedel–Crafts reactions fail on rings that are already deactivated, so the acyl group must be installed first while the benzene ring is still fully active.
Once the acyl group is on the ring, we choose between nitration and chlorination as the next step. Because the nitro group is a meta director and chlorine is an ortho-para director, the nitration must come second. Introducing the nitro group now ensures that all remaining ortho-para positions are deactivated, leaving only the intended position available for the final chlorination. Chlorination then becomes step three.
Synthesis
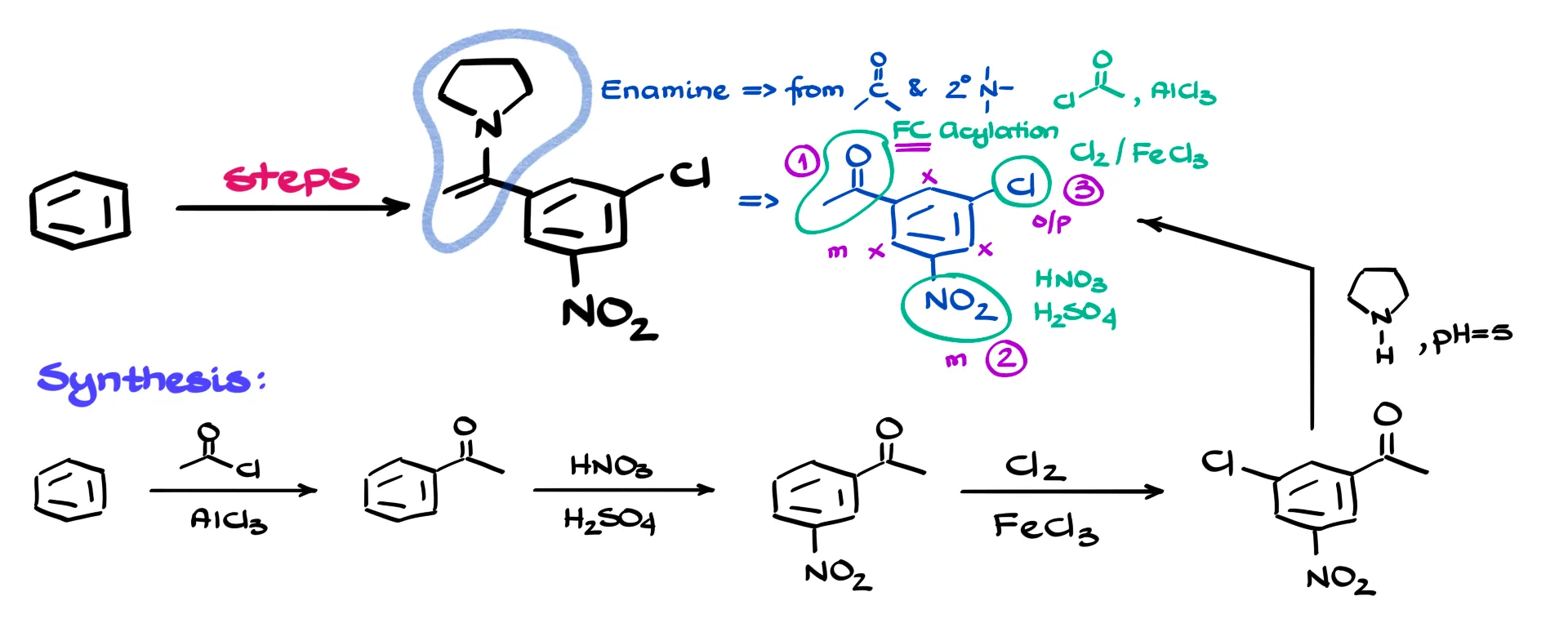
So let’s put the full synthesis together. We start with benzene. The first transformation is the Friedel–Crafts acylation, producing acetophenone. Next we carry out nitration by treating the compound with concentrated nitric and sulfuric acids. This gives the nitro-substituted acetophenone. After that, we perform the final electrophilic aromatic substitution: chlorination with Cl2 in the presence of a Lewis acid such as FeCl3 or AlCl3. That installs the chlorine at the correct position.
With all three substituents on the ring, the last step is forming the enamine. We react the carbonyl compound with the secondary amine pyrrolidine. Enamine formation works best under mildly acidic conditions, so a pH of about five is ideal. This gives the target molecule.
