39. Synthesis of an Ester from 2-chlorotoluene

At first glance this looks easy. We start with 2-chlorotoluene and we want to replace the chlorine with an ester. The catch is that doing a direct substitution will not work. An SN2 reaction is impossible on an sp²-hybridized carbon, and nucleophilic aromatic substitution is also off the table. We have no electron-withdrawing group to stabilize the intermediate. The CH₃ group is electron donating, which actually makes the aromatic ring even less cooperative. If we tried any direct substitution via benzyne chemistry, we would lose regioselectivity and waste a good portion of the starting material. And best of luck purifying the product…
So we look at the product instead. We need to build an ester. One classical route would be to generate a phenoxide and react it with an acid chloride or some other carboxylic acid derivative. The problem is getting an O⁻ into the ortho position of chlorotoluene. That is not a realistic move, as I’ve explained a moment ago.
There is, however, a reliable alternative: the Baeyer–Villiger oxidation. This reaction allows us to insert an oxygen between the aromatic ring and the carbonyl. That points us toward building the ketone. And since we want to introduce a two-carbon fragment, forming a new carbon–carbon bond with a Grignard reagent is a straightforward way to do it. After that, oxidation of the secondary alcohol will give the ketone.
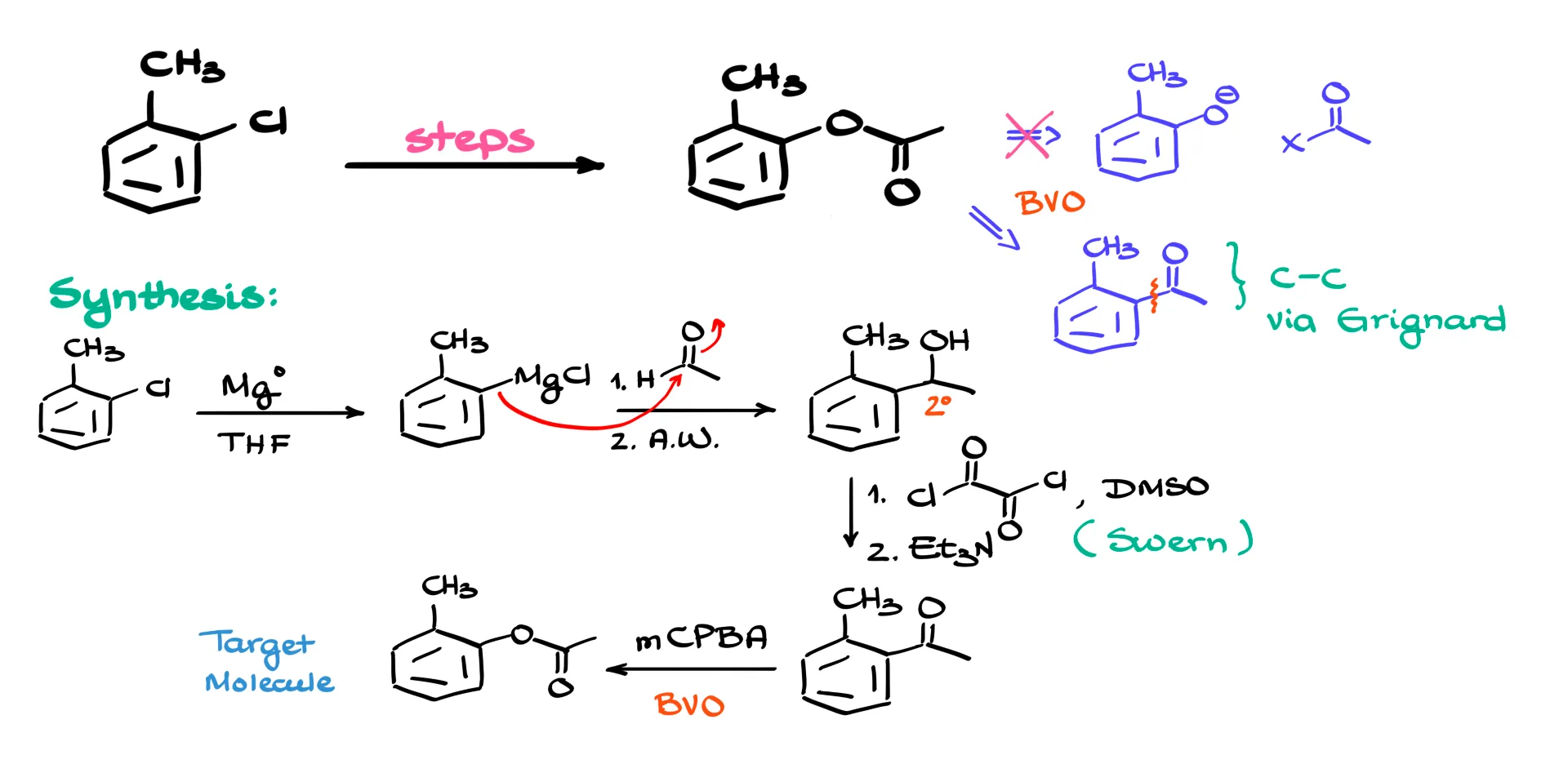
Let’s assemble the full sequence. We begin with 2-chlorotoluene and convert it into the organometallic reagent by treating it with magnesium in THF or another ether-like solvent. That gives the corresponding arylmagnesium chloride. To add the two-carbon unit, we react this Grignard reagent with acetaldehyde, then carry out the aqueous workup. The nucleophilic carbon of the Grignard reagent attacks the electrophilic carbonyl carbon of the aldehyde, forming the new carbon–carbon bond. After workup we obtain the corresponding secondary alcohol.
Next we oxidize the alcohol to the ketone. Because this is already a secondary alcohol, many oxidizing agents will work. I prefer Swern oxidation here so that I avoid the risk of overoxidation. Jones oxidation would be dangerous because chromium-based reagents will happily oxidize benzylic carbons that have at least one hydrogen. That means the toluene methyl group would oxidize to a carboxylic acid, and even the newly added fragment would be chopped down to its carboxylic acid. We definitely do not want that, so Swern oxidation keeps everything under control.
Once the ketone is in place, the final step is the Baeyer–Villiger oxidation. Treating the ketone with m-chloroperbenzoic acid, mCPBA, or any similar peroxy acid inserts an oxygen and transforms the ketone into the ester. EZPZ!
