Why You Keep Hitting A Wall With Organic Chemistry
If you’re spending hours studying organic chemistry and still not getting your A, this post is for you.
Let me start with an example.
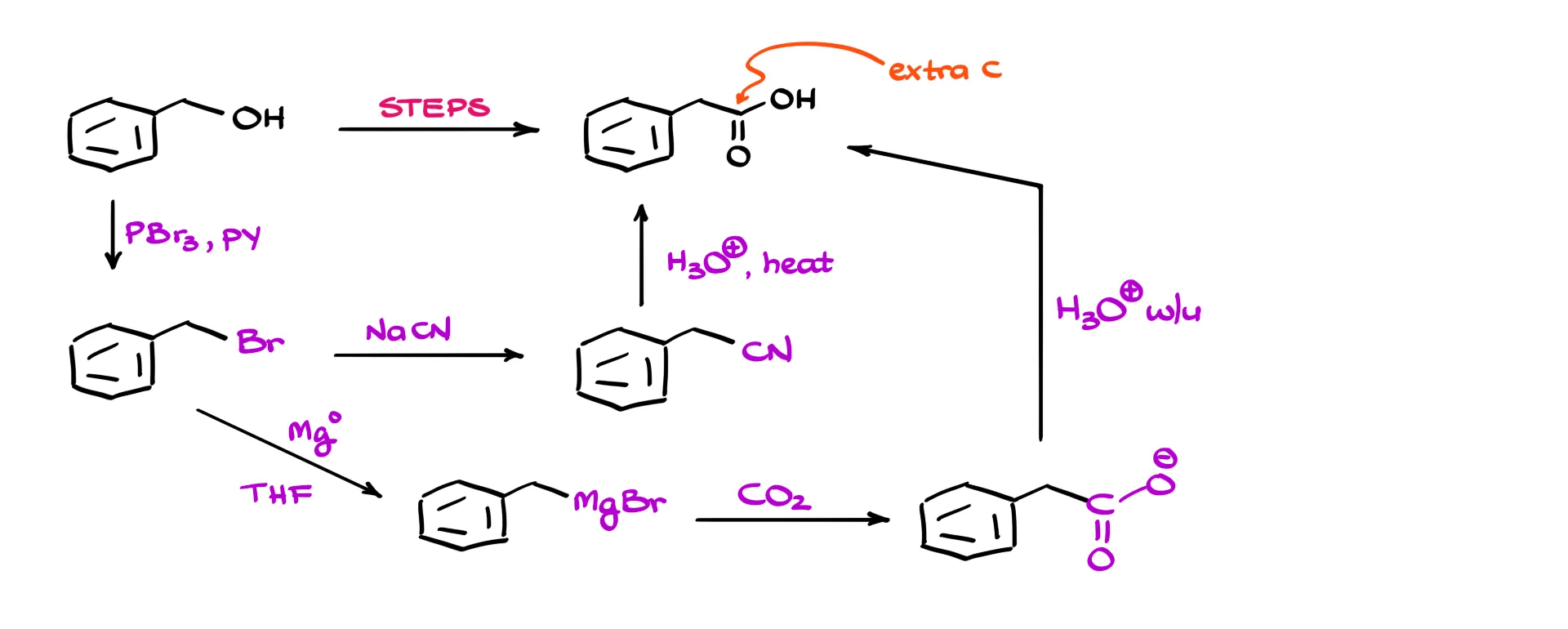
Here is a fairly typical second-semester organic chemistry question: we start with benzyl alcohol and need to convert it into a carboxylic acid. The first thing I notice is that the product has an extra carbon. That suggests an approach where we convert the alcohol into the corresponding bromide, then treat it with sodium or potassium cyanide to form the nitrile, and finally hydrolyze it to get the carboxylic acid. Alternatively, we could use a Grignard reagent and perform a carboxylation with carbon dioxide.
If you’ve seen questions like this before, it probably felt familiar. If you haven’t, it likely felt like you just learned something useful. And if that’s what you thought—congratulations, you’re in the majority. And I also just tricked you. I’ve been teaching organic chemistry for over 20 years, and I see the same issue over and over again: most students are stuck in a recognition and memorization loop. That little “oh, I get it” moment you just felt isn’t learning—it’s recognition. It’s a quick dopamine hit because your brain recognized something familiar. But I didn’t actually teach you anything. I gave you a very specific recipe for a very specific problem.
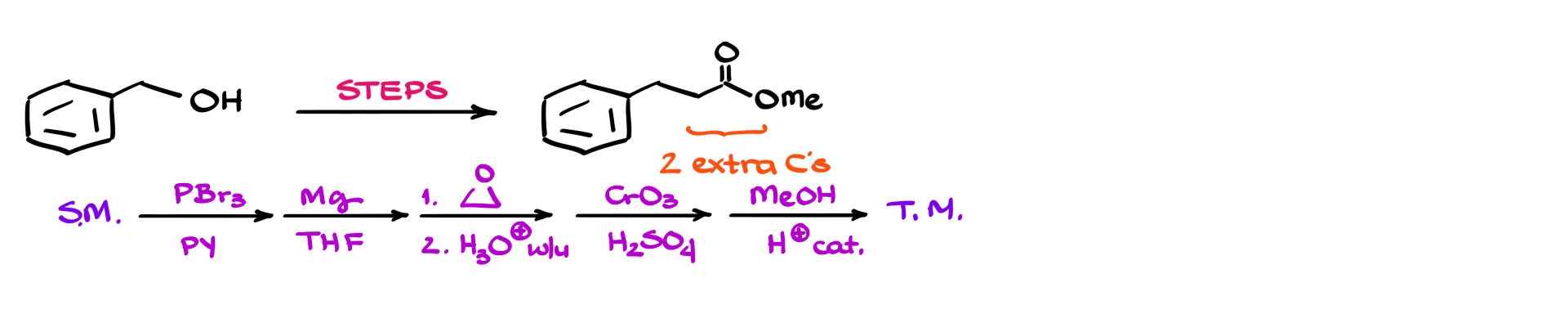
Now consider a slightly different example. If the product has two extra carbons instead of one, that previous approach no longer works. The idea is similar, but that small structural change forces a completely new sequence. So now you feel like you need another trick, another rule, another recipe—and that’s the trap. Organic chemistry is not your grandma’s recipe book. You can’t collect enough recipes to succeed on exams.
Recognize Building Blocks!
Instead, you need to focus on the building blocks. Every reagent can be understood in terms of a few roles: acid, base, nucleophile, or electrophile. Solvents and catalysts matter too, you need to recognize those as part of the system. Once you start thinking in these terms, reactions stop being memorized facts and start becoming logical patterns. A strong base means something will get deprotonated. A nucleophile and an electrophile mean there will be an attack. Now you’re not recalling—you’re reasoning.

I saw this clearly in a recent tutoring session.
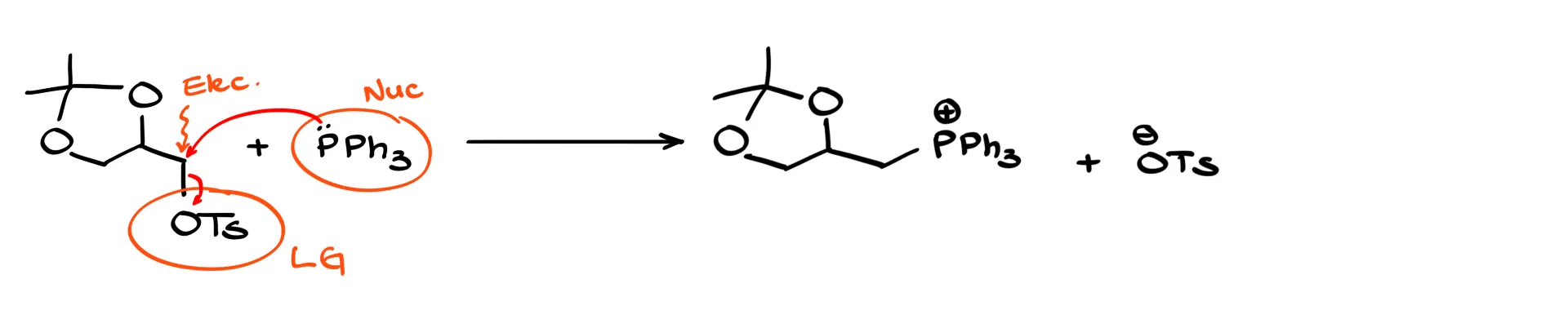
A student recognized a tosylate but didn’t know what to do because she associated it only with a specific reaction—making bromides from alcohols. She was thinking in terms of a recipe. But if you understand that tosylate is a good leaving group, then that carbon is an electrophile. Triphenylphosphine is a nucleophile. Once you identify those roles, the reaction becomes straightforward: nucleophile attacks electrophile, the leaving group leaves, and the product forms. If you rely on recipes, one small change breaks everything. If you understand patterns, you can adapt.
How DO I Know … ?
This is also where many students sabotage themselves. I constantly hear, “How do I know which double bond reacts?” or “How do I know which atom gets protonated?” The answer is: you don’t. You’re not supposed to know ahead of time. That question is your brain looking for a shortcut, a rule to memorize. But that’s not how organic chemistry works. Instead of asking “how do I know?”, try both options. Compare the intermediates. Evaluate their stability. Then decide.

Think of it like choosing ice cream. You don’t ask, “What’s the correct flavor?” You try a few and make a decision. The same idea applies here. You don’t magically know which bond reacts. You explore possibilities, run a mental experiment, and draw a conclusion. You explore first and conclude second.
If there’s one thing I want you to take from this, it’s that organic chemistry is a science of patterns, not memorization. It’s about reasoning, not tricks. If your studying feels like collecting facts and shortcuts, you’re going to keep hitting a wall. But if you shift your focus to roles, patterns, and mechanisms, that’s when things start to click. And that’s how you actually get good at organic chemistry.

