41. Synthesis of an Oxime from an Alcohol
Can you solve this exam synthesis? Well, let’s work through it.
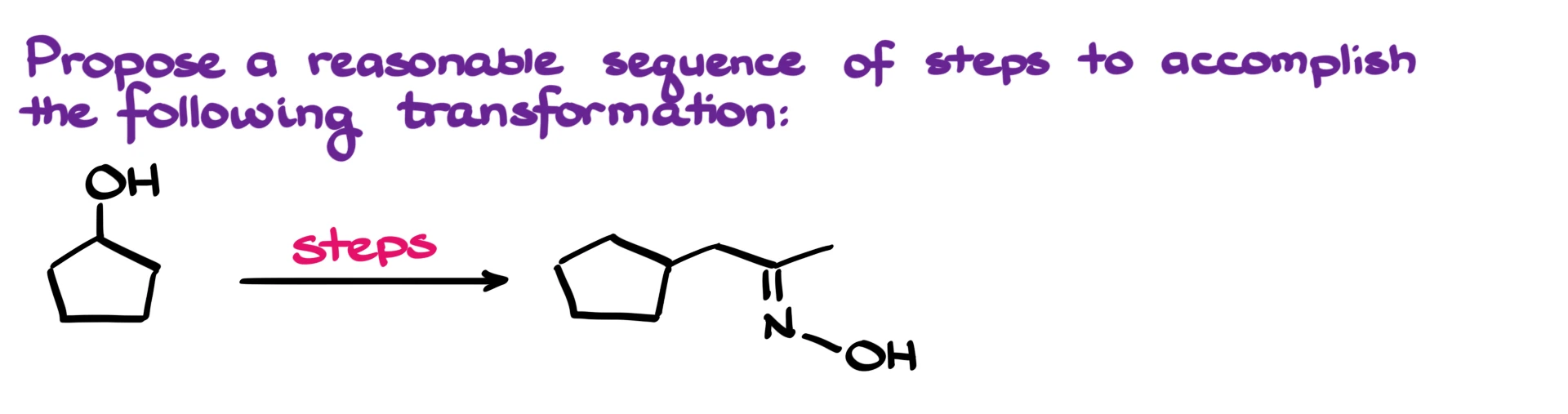
The very first thing that jumps out at me is the functional group over here, which is an oxime. That’s easily made by reacting the corresponding carbonyl compound with hydroxylamine, which means the direct precursor to our final product must have been the carbonyl.
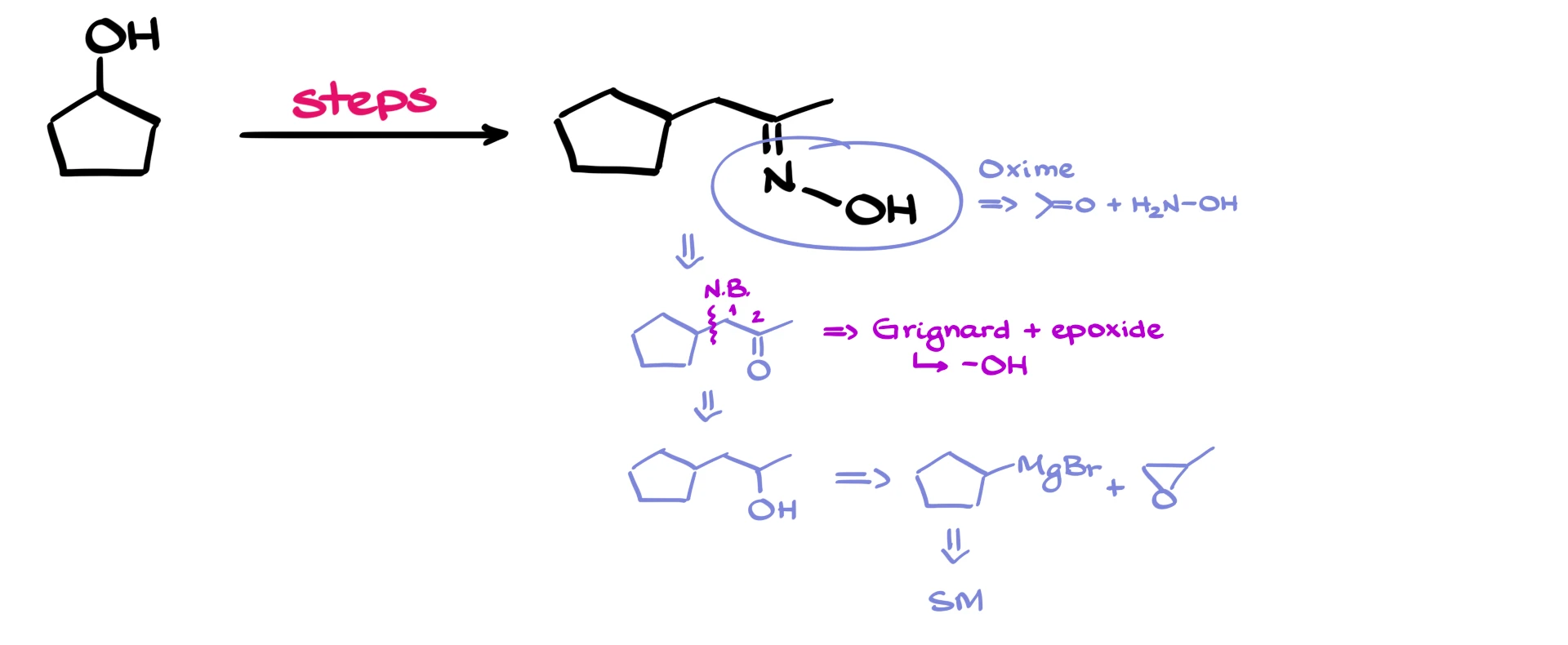
Now, the next thing I notice is that we’re going to be forming a new carbon–carbon bond, so I’m going to show that new bond. I’m also seeing that we’ll be forming the functional group at the second carbon, the kind we often get from a Grignard reaction where the Grignard reagent reacts with an epoxide.
We also know that a Grignard reaction typically gives an alcohol, which means that our ketone must have come from that alcohol. And that alcohol, in turn, can be made easily by reacting the appropriate Grignard reagent with an epoxide, just like I mentioned a moment ago.
And of course, we can make that Grignard reagent from our starting material in just a couple of simple steps.
So, now that we have a good idea of how we’re going to approach this synthesis, let’s outline our steps.
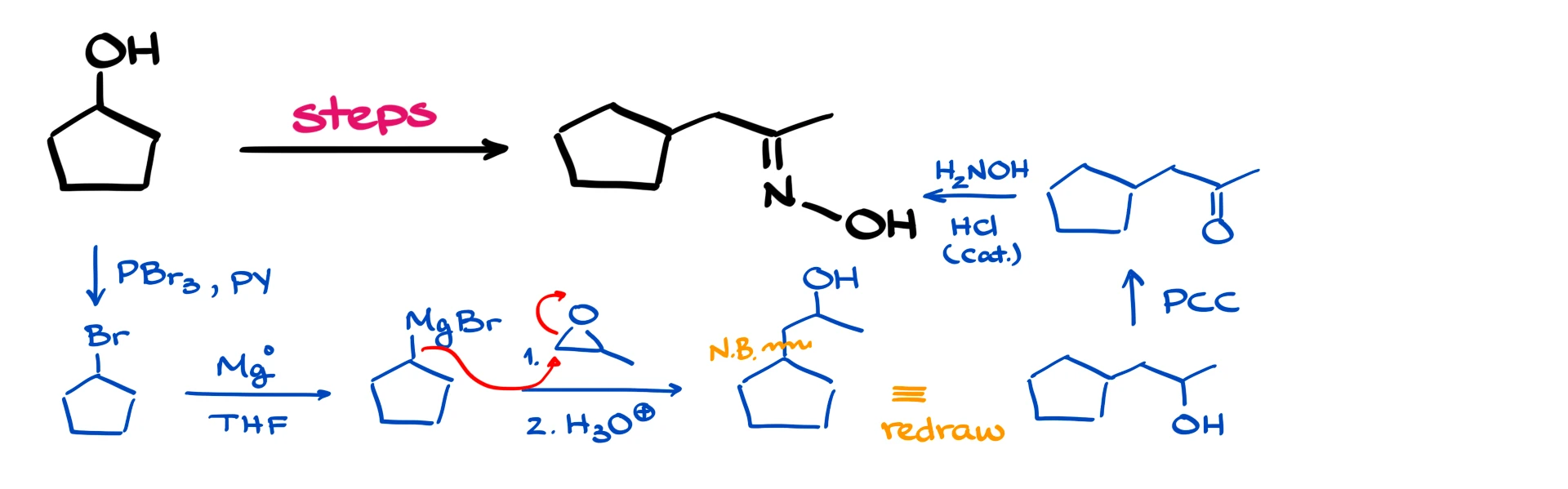
To convert the alcohol into the corresponding Grignard reagent, I’ll first turn the alcohol into a bromide using something like PBr₃ and pyridine. That gives us our bromide.
Then I’ll treat it with magnesium shavings in THF or another ether-type solvent, and that gives us our Grignard reagent.
Now we bring in our epoxide, and the Grignard is going to attack from the less substituted side. After the acidic workup, that gives us our alcohol, and of course the new carbon–carbon bond I just created is right here.
And now we only have a couple of steps left.
First, we need to oxidize the alcohol. Let’s use PCC, because why not? We’re dealing with a secondary alcohol, so it really doesn’t matter much which oxidation method we use; any standard oxidation technique will get us to the ketone.
And finally, to get to our target molecule, our final product, we’ll react the ketone with hydroxylamine in the presence of a trace amount of acid as a catalyst. And there we have it.
The only slightly tricky part you had to recognize here is that our functional group, the oxime, is basically just an imine analogue, and by now, hopefully you’re already familiar with how to make imines.
So, did you arrive at the same solution, or did you come up with something different? Let me know in the comments below.
