42. Synthesis of a Complex Ether from a Simple Alkene
So here is a really fun exam synthesis.
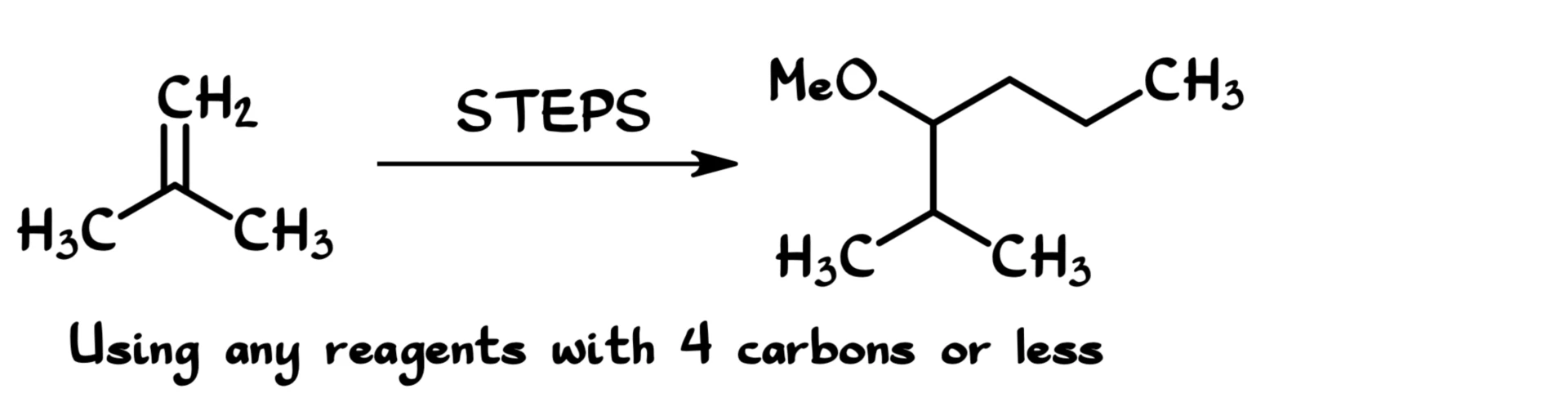
Looking at my starting material and my final product, I can see that my starting material maps nicely onto my final product. That tells me we’re going to be adding this methoxy group, but more importantly, we’re going to be forming a new carbon–carbon bond. I’ll mark that new bond with N.B.
Most likely, we’re going to form that bond using a Grignard reagent or a similar organometallic reaction.
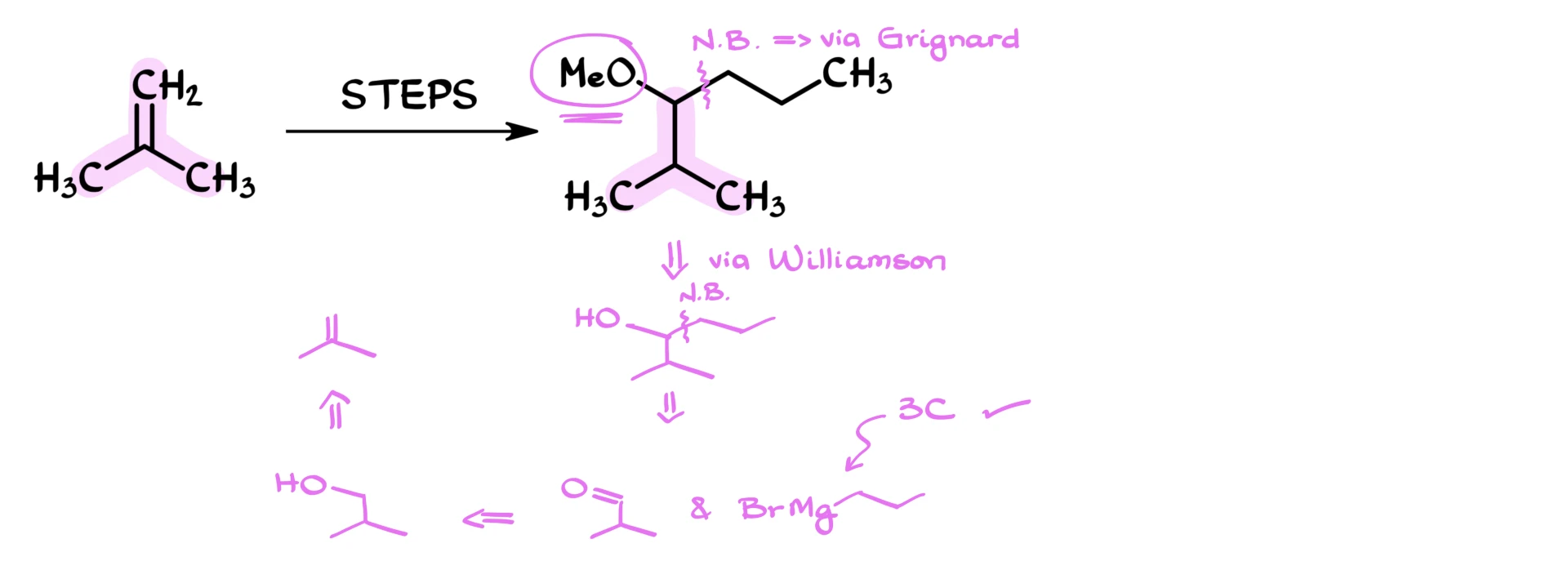
If I form this new carbon–carbon bond using a Grignard reagent, I know I’m going to generate an alcohol at the position where the methoxy group eventually ends up. That means the methoxy group will probably be introduced later using a Williamson ether synthesis. Most likely, that will be the final step of the synthesis.
So, a reasonable immediate precursor to my target molecule would be the corresponding secondary alcohol. Then I can convert that alcohol into the ether using the Williamson ether synthesis.
Now let’s think about the carbon–carbon bond formation. If I disconnect that bond, I get an aldehyde fragment and a propylmagnesium bromide fragment. The propyl group has three carbons, so that satisfies the “four carbons or fewer” requirement perfectly.
As for the aldehyde, we can obtain it from the corresponding alcohol, which in turn can be derived directly from our starting material.
Now that we have the retrosynthetic plan in place, let’s move forward with the actual synthesis.
Synthesis
First, I redraw my starting material. Step one is hydroboration–oxidation, which installs the alcohol on the less substituted carbon. That gives me the primary alcohol.
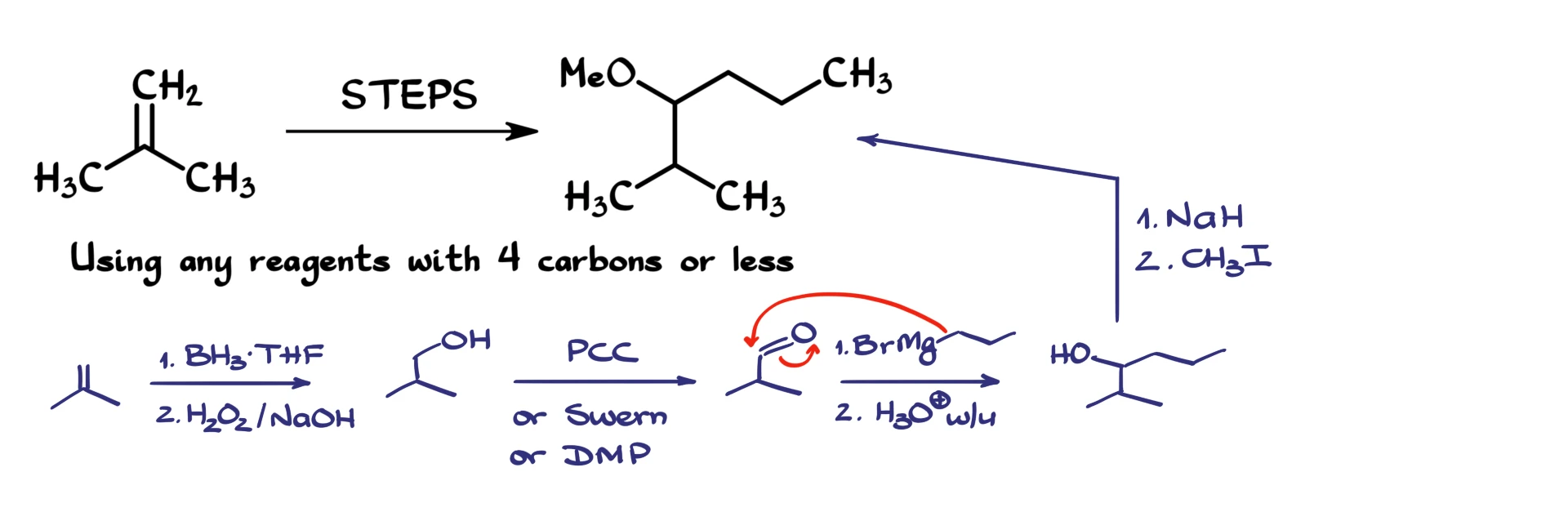
Next, I need to oxidize that alcohol to the corresponding aldehyde. I can use any oxidation method that stops at the aldehyde stage. PCC works, Swern oxidation works, Dess–Martin periodinane works. It doesn’t really matter, as long as we stop at the aldehyde. Let’s go with PCC for simplicity. That gives us the aldehyde.
Next, we treat the aldehyde with propylmagnesium bromide. Since this reagent contains only three carbons, it satisfies the reagent restriction. The Grignard reagent attacks the carbonyl carbon, and after acidic workup we obtain the secondary alcohol.
At this point, all that remains is converting the alcohol into the ether. As we discussed earlier, we can accomplish that using the Williamson ether synthesis. First, we treat the alcohol with sodium hydride to deprotonate it and form the alkoxide. Then we perform an SN2 reaction with methyl iodide. That installs the methoxy group and gives us the final product.
And that’s it.
Would you have approached this synthesis the same way, or would you have used a different strategy? Let me know in the comments below.
