Bond Length Ranking

In this tutorial I want to talk about bond length rankings, and as a little bonus at the end we’ll also look at bond strengths.
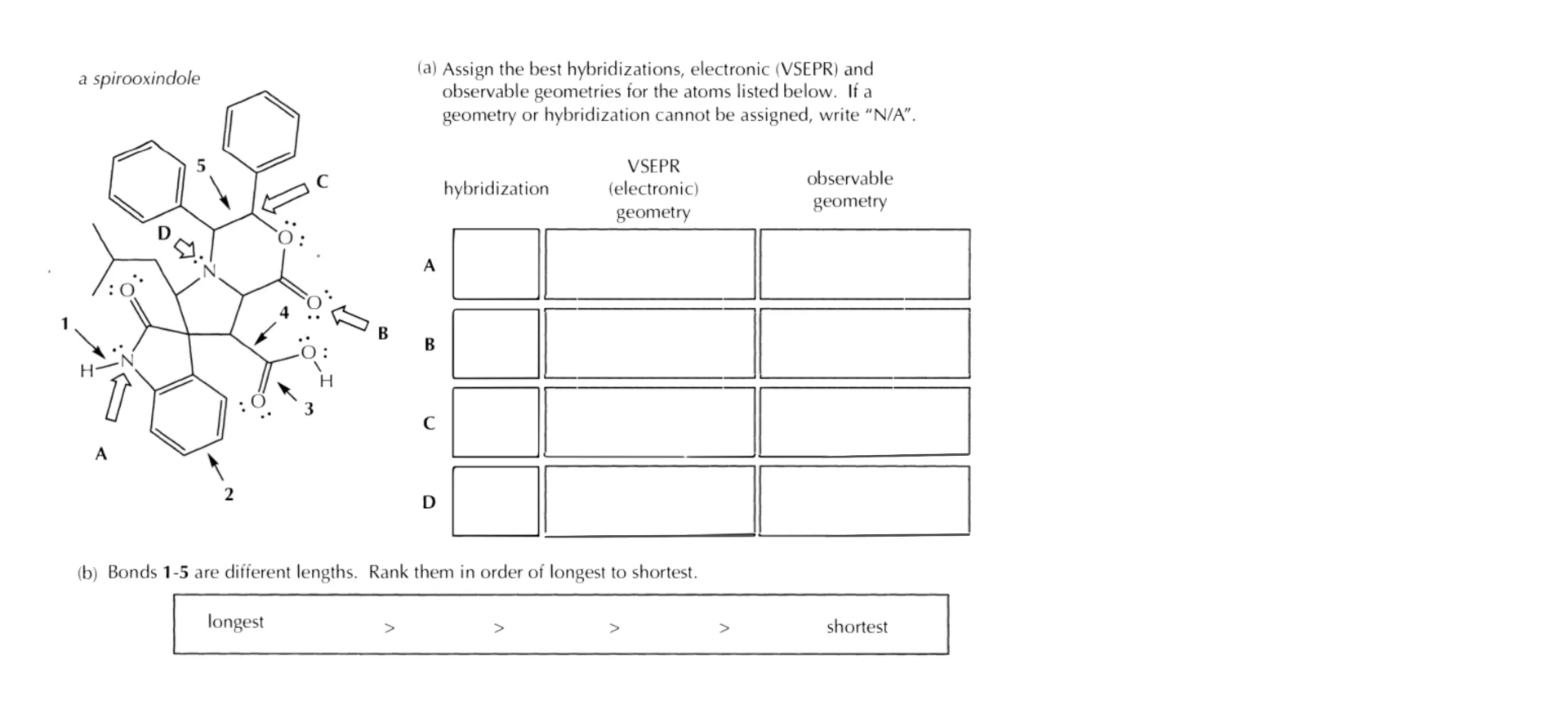
Here’s a fairly typical exam question: you’re given a molecule and asked to rank the indicated bonds by their length. Or, alternatively, you might see a series of molecules and each has a bond you would need to rank in terms of the bond length. It doesn’t matter how the question if phrased, the idea is always going to be the same: rank the bonds based on their length.
At first, this type of problem can look intimidating, but once you know the four core principles I’m about to show you, it becomes much more manageable.
Principle 1: Atomic Size
The first principle is atomic size. In organic chemistry, the size of the atom is the most important factor for bond length. Other factors like electronegativity, hybridization, or geometry do play a role, but atomic size dominates.

Hydrogen, for example, is the smallest atom. The H–H bond is very short, only 0.74 Å. (And just to remind you, 1 Å is 10-10 m, ten times smaller than a nanometer.)
Any bond with hydrogen is going to be short. Here are some examples:
- C–H = 1.09 Å
- N–H = 1.02 Å
- O–H = 0.96 Å
Compare that to a C–C bond, which is 1.53 Å. Since carbon is much larger than hydrogen, the bond is longer.
We can see similar patterns as we move through the periodic table. For example:
- C–F = 1.35 Å
- C–Cl = 1.77 Å
- C–Br = 1.93 Å
- C–I = 2.13 Å
Relevant Periodic Table
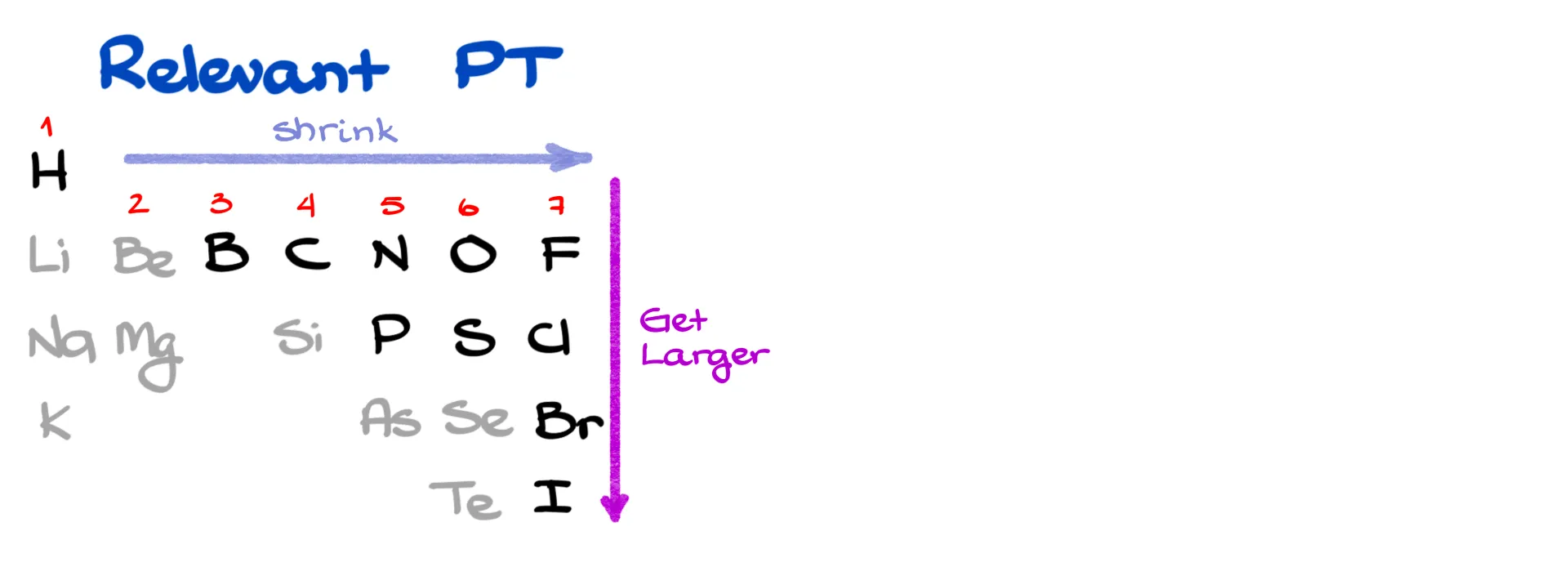
As you can see, bond length increases as the atoms get larger as you move down to the next period. Good news is, you don’t need to memorize these numbers! The periodic trends are what matters. Across a period, atom size decreases slightly from left to right, and down a group, atom size increases significantly.
Example 1
Let’s apply this.
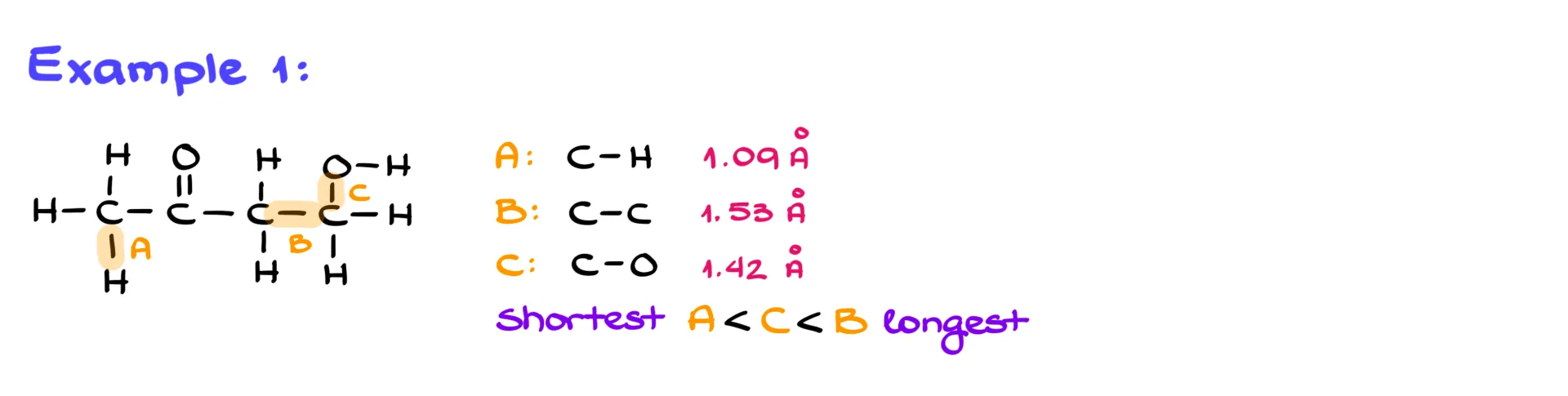
Suppose we need to rank C–H, C–C, and C–O bonds from the molecule above. Since hydrogen is smallest, C–H is shortest. Between C–O and C–C, oxygen is smaller than carbon, so C–O is shorter than C–C.
Principle 2: Bond Order
The second principle is bond order. Triple bonds are shorter than double bonds, and double bonds are shorter than single bonds.

For example, with carbon–nitrogen bonds, or nitrogen–nitrogen bonds, you’ll consistently see:
C≡N < C=N < C–N
But we still need to account for atomic size. Compare carbon–oxygen versus carbon–sulfur bonds. Even though a C=S double bond has higher bond order, it’s still longer than a single C–O bond because sulfur is much larger than oxygen.
Example 2
Here’s another example with several highlighted bonds:
C–H, C≡C, C=O, C=C, and C–C.
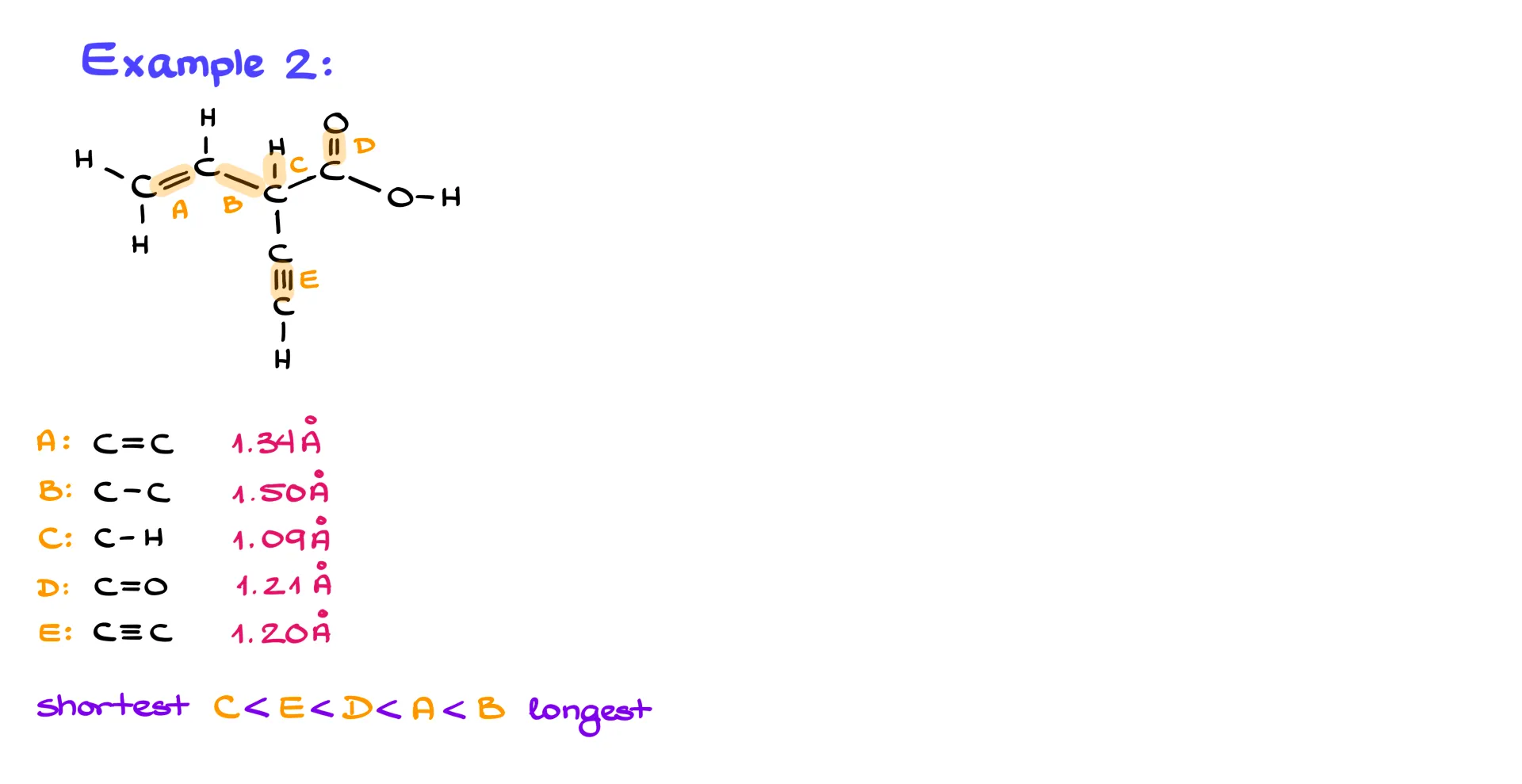
The shortest is C–H, followed by the triple bond, then the double bonds (C=O and C=C), and finally the longest is the single C–C bond.
So far, atomic size and bond order are the two major principles for ranking bond lengths. But there are two more to keep in mind.
Principle 3: Hybridization
The third principle is hybridization. The more s-character in a hybrid orbital, the shorter the bond.

- sp³ orbitals are largest, so sp³ bonds are the longest.
- sp² bonds are shorter.
- sp bonds are the shortest.
This effect isn’t as strong as atomic size or bond order, but it can still matter.
Principle 4: Resonance
The fourth principle is resonance. Resonance often creates partial bonds that fall in between single and double bond lengths.
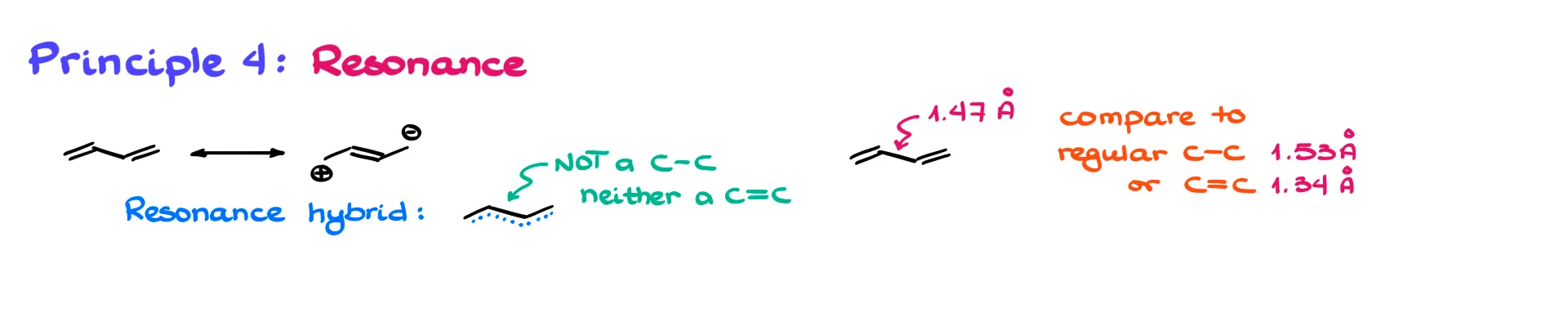
Take butadiene, for example. Resonance makes the central C–C bond not quite a single bond, but not quite a double either. Experimentally, it measures around 1.47 Å—shorter than a normal single bond but longer than a double bond.
So, once you’ve ranked bonds based on atomic size and bond order, double-check whether hybridization or resonance applies, since these can shift the bond lengths slightly.
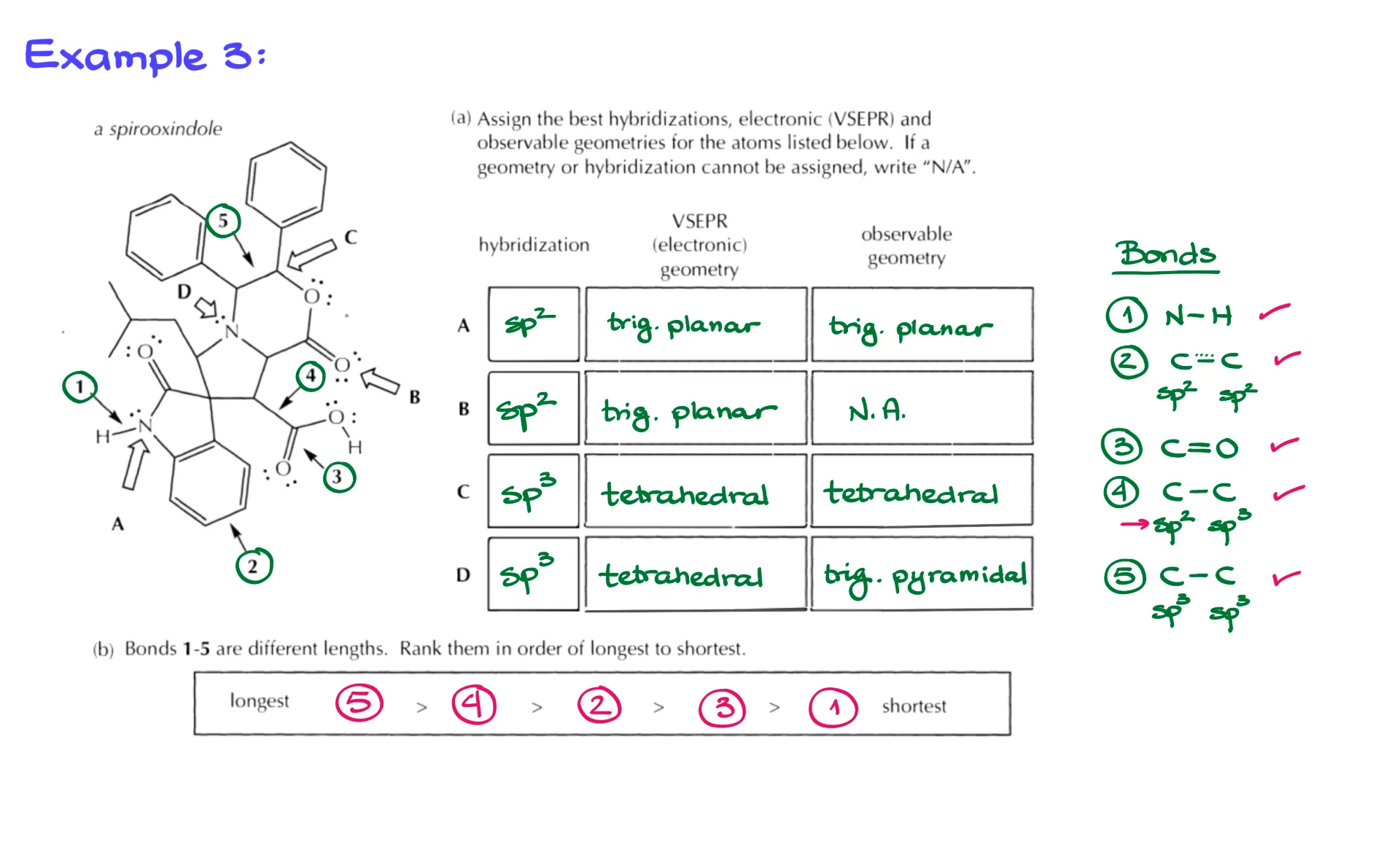
Complex Example
Now let’s return to the type of exam question we started with. Using these four principles: atomic size, bond order, hybridization, and resonance.
Bond Strength
And now for the bonus: bond strength. Bond strength and bond length are correlated: the shorter the bond, the stronger it is.

But there’s a catch: this only works when comparing bonds of the same type or between similar atoms.
For example, compare carbon–carbon single, double, and triple bonds. The single bond is longest and weakest, while the triple bond is shortest and strongest. The same trend holds for carbon–nitrogen bonds.
But if you compare very different bonds, the trend breaks down. A C–H bond is very short, but it’s not nearly as strong as other bonds of similar length. An H–H bond is even shorter, yet only about as strong as a C–H bond.
So, the bond length–bond strength correlation is useful, but only for comparing similar types of bonds.
The good news is, exam questions will usually stick to straightforward comparisons. If you master these four principles and practice a few examples, you’ll be ready to tackle bond length ranking problems with confidence.

