Cyanohydrins
Formation of cyanohydrins from aldehydes and ketones is probably one of the first reactions we introduce in the carbonyl chapter. In a nutshell, this is a reaction of a carbonyl compound, either an aldehyde or a ketone, with cyanide, giving the corresponding cyanohydrin.

A cyanohydrin is a combination of two functional groups in which we have a nitrile and an alcohol, an OH group, both attached to the same carbon atom.
Synthesis of Cyanohydrins
Making cyanohydrins is fairly easy. For example, if I take acetone and react it with sodium cyanide and aqueous sulfuric acid, I get the corresponding cyanohydrin in roughly 80% yield, which is not bad at all.
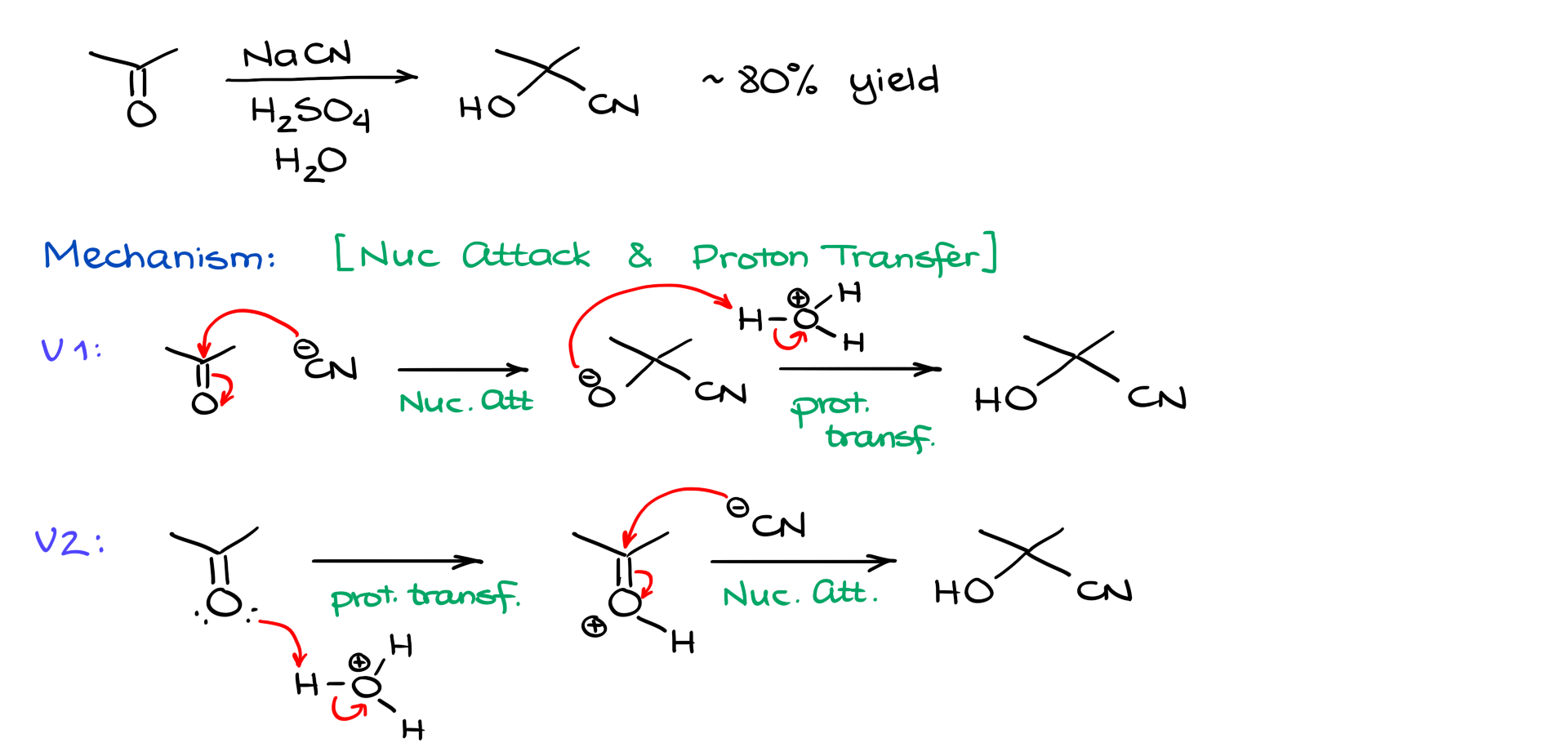
Mechanistically, this reaction is a sequence of a nucleophilic attack and a proton transfer. However, depending on your textbook and your instructor, you may see those two steps shown in different orders.
What I mean is that some textbooks and instructors show the nucleophilic attack first. In that version, cyanide attacks the carbonyl carbon to give a negatively charged alkoxide intermediate, and then a proton transfer gives the final cyanohydrin.
Other books and instructors show the proton transfer first, giving a protonated carbonyl intermediate, and then cyanide attacks that activated carbonyl to form the cyanohydrin.
So which mechanism is more correct? That is a very good question. As far as I know, there are no direct mechanistic studies that definitively prove one version over the other. If you happen to know of any, let me know. Personally, I lean toward the second version, the one where protonation comes first, simply because forming a negatively charged intermediate in strongly acidic conditions, after all we are working with sulfuric acid, feels a little questionable.
Also, in many other reactions of carbonyl compounds under acidic conditions, the first step is typically protonation of the carbonyl oxygen. So the protonated intermediate shown in the second version seems very plausible.
That said, I am not the one assigning your final grade, so you should always check with your textbook or your instructor to make sure you know which version is expected on your test.
Cyanohydrin Formation Equilibrium
One really cool thing about this reaction is that it is an equilibrium. That means I can take the cyanohydrin and reverse the process to regenerate the original carbonyl compound and cyanide.

Since we normally make cyanohydrins under acidic conditions, we can often push the reverse process by switching to basic conditions instead. So if I take a cyanohydrin and treat it with sodium hydroxide, I can get back the original carbonyl, acetone in this case, along with cyanide.
In this reverse process, cyanide acts as a leaving group. That may seem a little unusual because cyanide is not one of the leaving groups we normally focus on, but under basic conditions it can actually leave relatively easily. There are several interesting reactions in organic chemistry where cyanide first acts as a nucleophile, then later serves as a leaving group and disappears once it is no longer needed.
Cyanohydrin Applications

Now, why do we care about cyanohydrins? Outside of the fact that they are kind of a fun combination of functional groups, cyanohydrins have some very useful synthetic applications.

First, we can hydrolyze the nitrile group of a cyanohydrin to a corresponding carboxylic acid. Since nitriles can be hydrolyzed under acidic conditions to carboxylic acids, cyanohydrins can serve as convenient precursors to α-hydroxy carboxylic acids.
For instance, if I start with cyclohexanone and convert it into the corresponding cyanohydrin, I can then hydrolyze that nitrile group to make 1-hydroxycyclohexanecarboxylic acid. That type of molecule would be significantly more challenging to make using many other synthetic routes.
So whenever we need to make a carboxylic acid with a functional group at the α position, cyanohydrins can be a very useful synthetic intermediate.

Another cool use for cyanohydrins is their ability to give amino alcohols after reduction of the nitrile group. For example, if I take a carbonyl compound and convert it into the corresponding cyanohydrin, I can then reduce the nitrile all the way to a primary amine.
That gives me a molecule containing both an alcohol and an amine, an amino alcohol. There are other methods that can make the same combination of functional groups, but this one is very clean, simple, and straightforward.
And since simple aldehydes and ketones are usually commercially available and fairly inexpensive, cyanohydrin chemistry provides a very practical starting point for many more complex synthetic sequences.
So while cyanohydrin formation is one of those reactions that often gets brushed aside as boring or insignificant, it actually offers a lot of versatility in synthesis of more complex targets. This is definitely a reaction worth keeping in your synthetic toolkit because it can save you a lot of time and effort on exams or homework problems.
