Aryl Diazonium Ions
In this tutorial I want to talk about everything you need to know about aryl diazonium ions: their synthesis, their reactions, and everything in between.
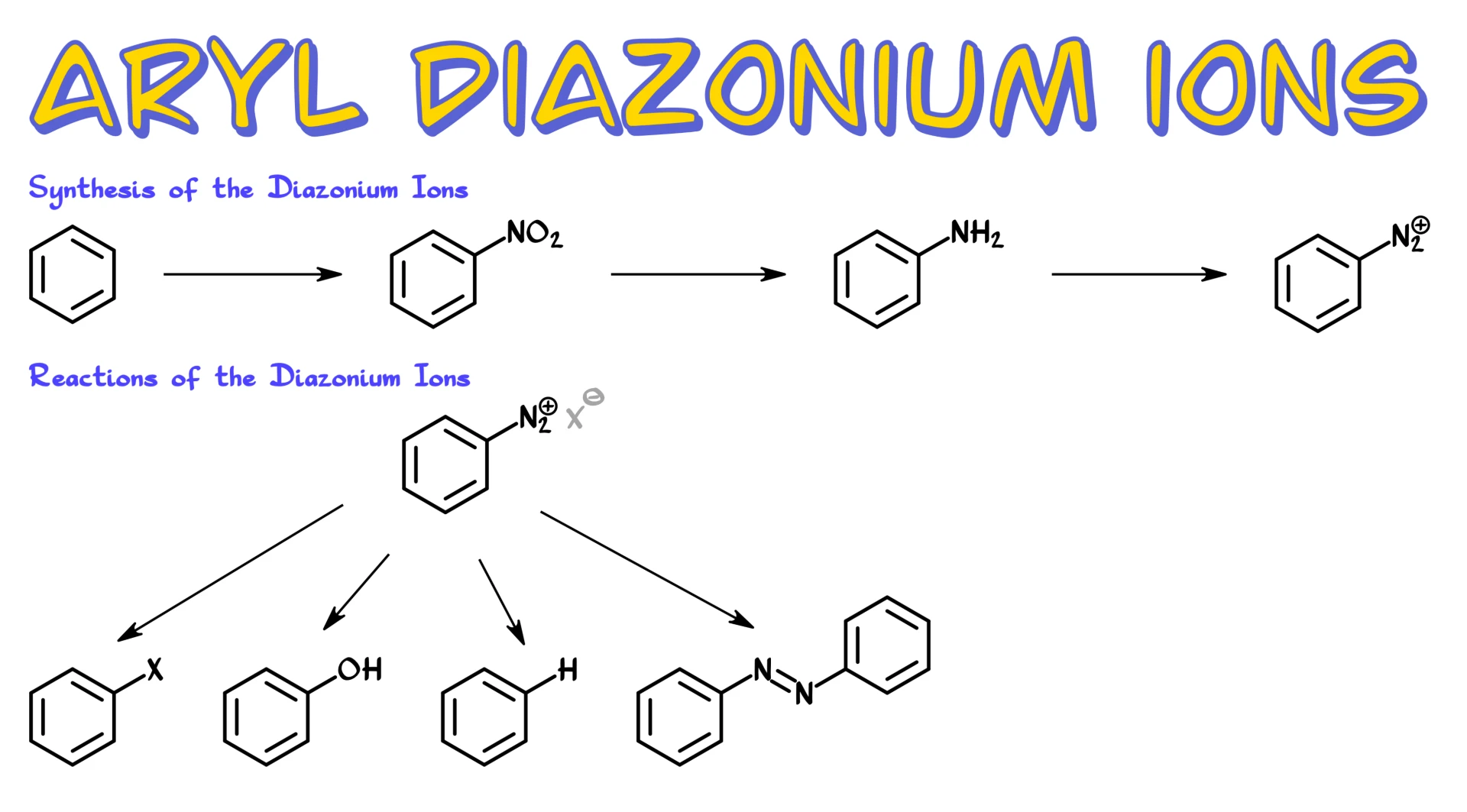
Synthesis od Aryl Diazonium Ions
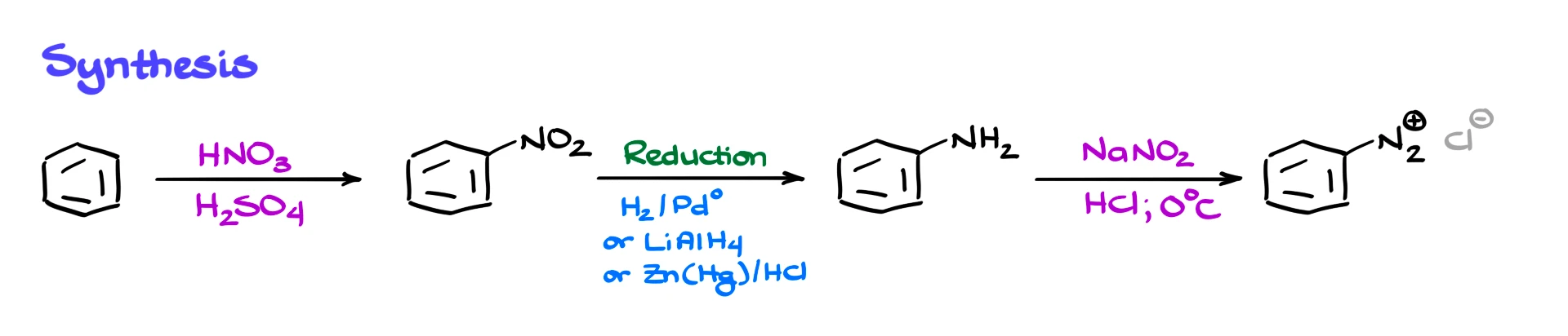
Typically, we begin with an aromatic compound, and the first thing we do is a nitration reaction. I do have a dedicated tutorial on nitration, so if you need a refresher, go ahead and check that out. Once we have our nitro compound, we reduce it. Reduction of nitro groups is an extremely easy task. Virtually any reduction method you know will work. You can use hydrogen with a heterogeneous catalyst such as platinum or palladium, you can use complex hydrides like lithium aluminum hydride, or you can use a metal and acid system such as zinc and acid. Take your pick. The good news is that you are typically not responsible for the mechanism of that reduction.
After reduction, we obtain our aromatic amine.
Once we have the aromatic amine, we treat it with sodium nitrite under acidic conditions at low temperature. That gives us the diazonium compound, specifically an aryl diazonium salt.
A couple of important points here. First, we are using sodium nitrite, NaNO₂, not sodium nitrate, NaNO₃. Don’t mix those up. Second, the diazonium species is positively charged, so it must have a counterion. Often that counterion is Cl⁻ if we are using HCl, but it could also be Br⁻, a sulfate ion, or something else. In most cases, the exact identity of the counterion is not particularly important, so we usually do not emphasize it.
Mechanism of Diazotization
Now, unlike the nitro reduction, the mechanism of diazonium formation is something you are often expected to know. So let’s go through it.
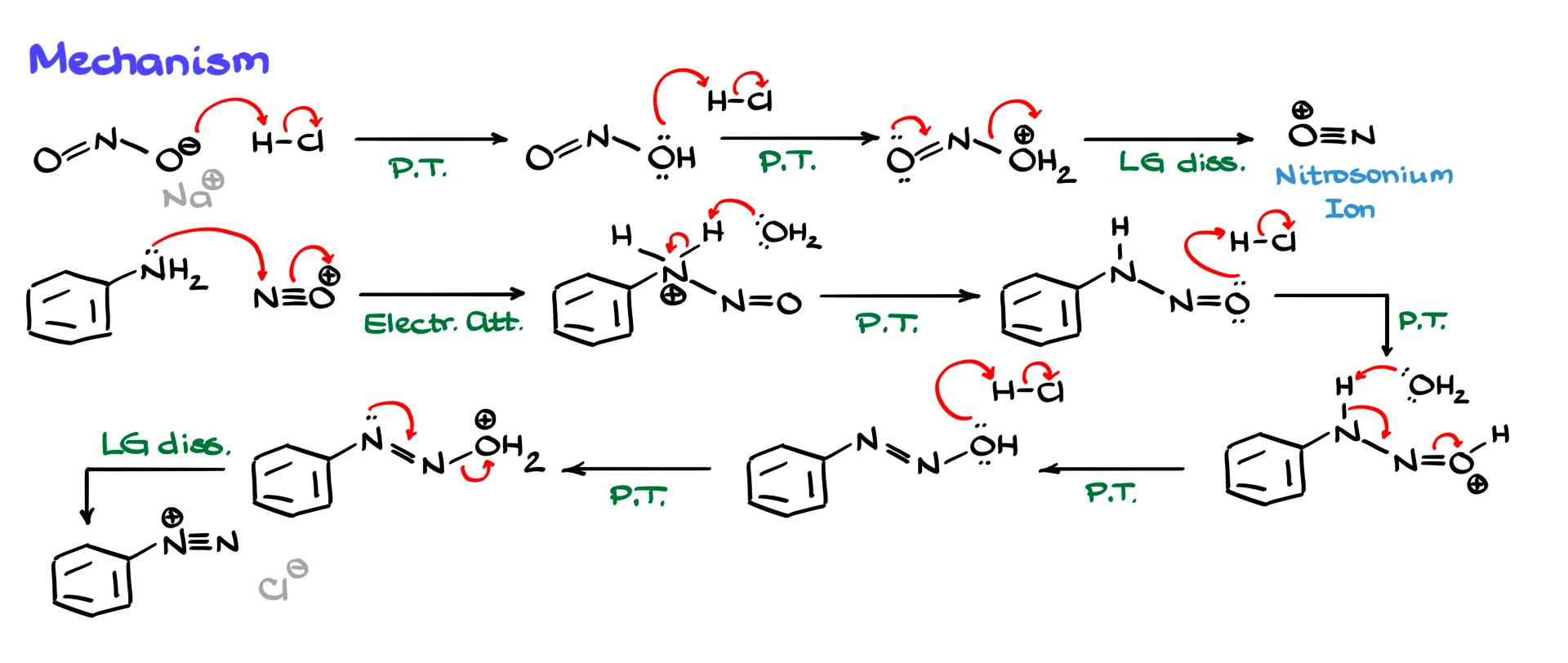
We start by reacting sodium nitrite with our acid, for example HCl. This is a simple proton transfer, forming nitrous acid, HNO₂. We cannot just use nitrous acid directly because it is unstable and decomposes easily, so we generate it in situ during the reaction.
Next, we protonate nitrous acid again with another equivalent of acid, forming a species where water becomes a good leaving group. Loss of water then gives us the nitrosonium ion, NO⁺. This nitrosonium ion is the key electrophile in the reaction.
Now we bring in our aromatic amine, for this example, I use aniline. The amine acts as the nucleophile, attacking the nitrosonium ion and forming a nitrogen–nitrogen bond. That gives us a protonated intermediate.
From here, we go through several proton transfers. First, we remove a proton from nitrogen to form a neutral intermediate. Then we protonate the oxygen, setting up another good leaving group. Another proton transfer and rearrangement of electron density leads us to a species with a nitrogen–nitrogen double bond. Finally, we protonate the –OH group, convert it into a good leaving group, and eliminate water. The electron pair on nitrogen helps expel the leaving group, and we end up with the aryl diazonium ion. The counterion, such as Cl⁻, balances the charge.
It is a long mechanism with many proton transfers, but it is one you should know for the test.
Now that we have our diazonium ion, what can we do with it?
Sandmeyer Reaction
Probably the most iconic reaction is the Sandmeyer reaction.

In this reaction, we replace the N₂ group with another substituent, typically Cl, Br, I, or CN. The source of the nucleophile is usually a sodium or potassium salt, such as NaCl, NaBr, KI, or KCN. Crucially, we also use a cuprous salt as a catalyst, such as CuCl, CuBr, CuI, or CuCN.
It is very important that the copper is in the +1 oxidation state. This is not Cu²⁺, it is Cu⁺. That detail matters for the mechanism.
Sandmeyer Reaction Mechanism
The mechanism of the Sandmeyer reaction involves radical chemistry.
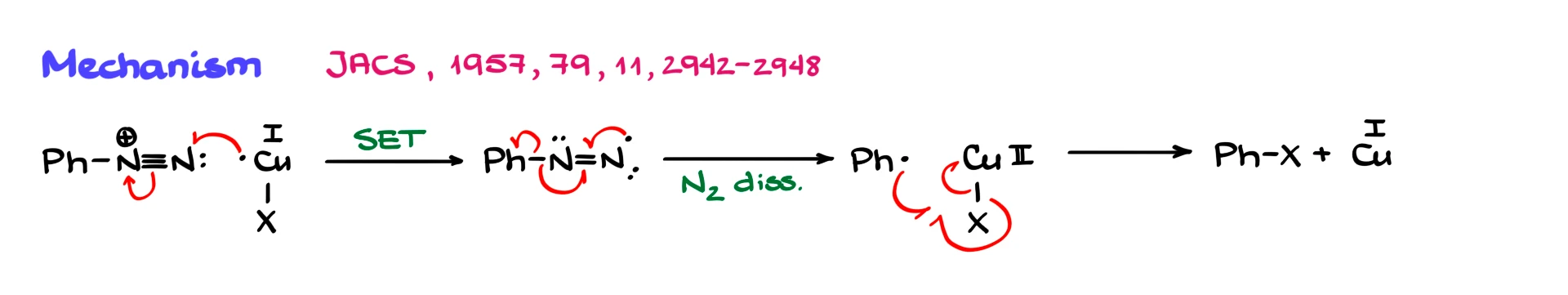
The first step is a single electron transfer from Cu⁺ to the diazonium ion. This generates a radical intermediate and converts copper to Cu²⁺. The radical intermediate then loses nitrogen gas, forming a phenyl radical. There is strong experimental evidence that this radical is indeed formed.
Next, the phenyl radical reacts with the copper(II) species bearing the halide or cyanide. In the pathway that makes the most sense, the radical abstracts the X group, forming the aryl halide or aryl nitrile and regenerating Cu⁺. Since we regenerate Cu⁺ at the end, copper truly acts as a catalyst.
Examples of the Sandmeyer Reaction
For example, starting from a 2-methoxyaniline, we first convert it into the corresponding diazonium salt using NaNO₂ and HCl at low temperature. Then, using KI and CuI at slightly elevated temperature, we replace the diazonium group with iodine.
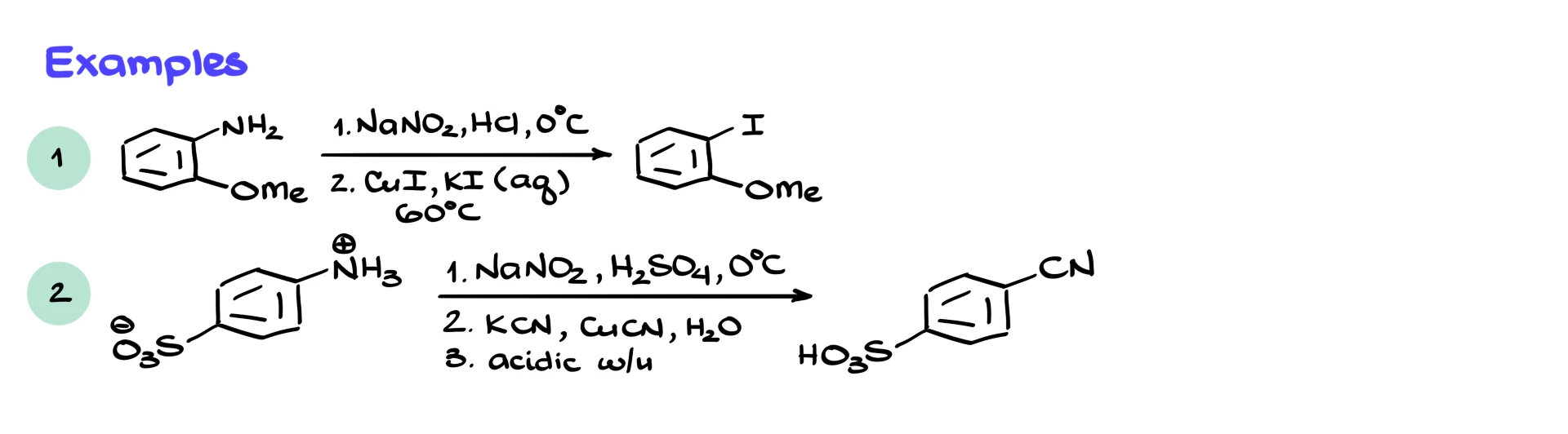
Another example involves an aromatic compound containing both an amine and a sulfonic acid group. These groups form a zwitterionic structure under normal conditions, similar to amino acids. We first generate the diazonium salt under the usual conditions. Then, using KCN and CuCN, we replace the diazonium group with a nitrile. After acidic workup, we obtain the corresponding cyanosulfonic acid. Installing a nitrile para to a sulfonic acid group would be very difficult by other methods, so this reaction is extremely useful.
The Sandmeyer reaction is not the only transformation available.
Fluorination (Schiemann Reaction)
We can also introduce fluorine using what is often called the Schiemann reaction.

In this case, we use tetrafluoroboric acid, HBF₄, or sodium tetrafluoroborate to form the corresponding diazonium tetrafluoroborate salt. Upon heating, it decomposes to give the aryl fluoride.
Mechanistically, this reaction is different from the Sandmeyer reaction. There is strong evidence that it proceeds through a phenyl carbocation intermediate. The tetrafluoroborate ion then delivers fluoride to the carbocation. Because phenyl carbocations are highly unstable, these reactions often produce significant side products. Even so, this remains one of the most reliable methods for introducing fluorine onto an aromatic ring.
Hydroxylation of Diazonium Ions
Another useful transformation is conversion of diazonium salts into phenols.

This hydroxylation is typically done in aqueous conditions using cuprous oxide, Cu₂O, as a catalyst. Some textbooks describe this as simple thermal decomposition, but without the copper catalyst you tend to get many side products. Using Cu⁺ makes the reaction much cleaner.
Reduction of Diazonium Ions
We can also reduce diazonium salts to simply replace the N₂ group with hydrogen.
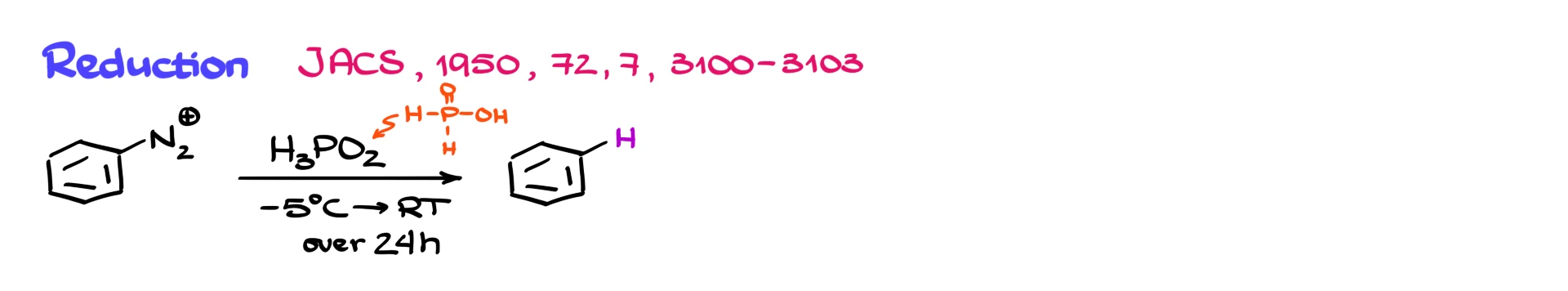
This reduction is done using hypophosphorous acid, H₃PO₂. Be very careful here. It is not H₃PO₄ and not H₃PO₃. The correct reagent is H₃PO₂. This is a common mistake. You usually do not need to know the mechanism, but you do need to know that this reaction exists and that H₃PO₂ is the reagent.
Azo Coupling Reaction
Finally, we have azo coupling reactions.
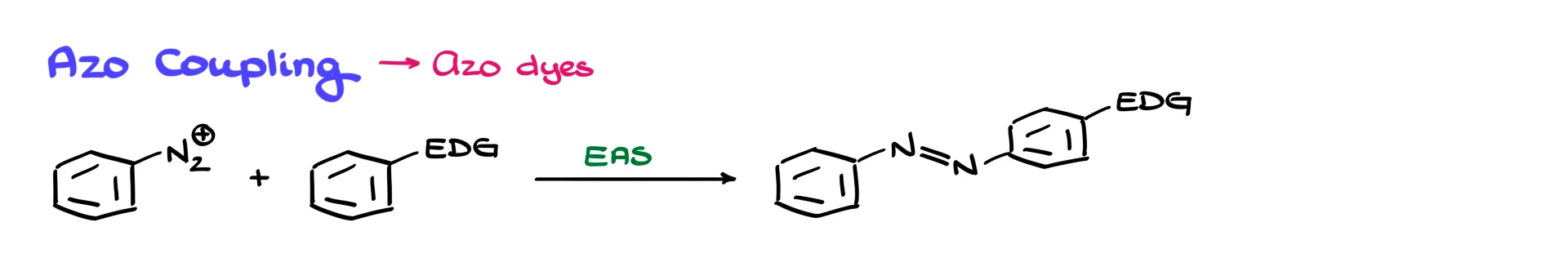
Azo coupling is an electrophilic aromatic substitution reaction used extensively in industry to produce azo dyes. Although many azo dyes have fallen out of favor, this reaction is still performed on a large scale.
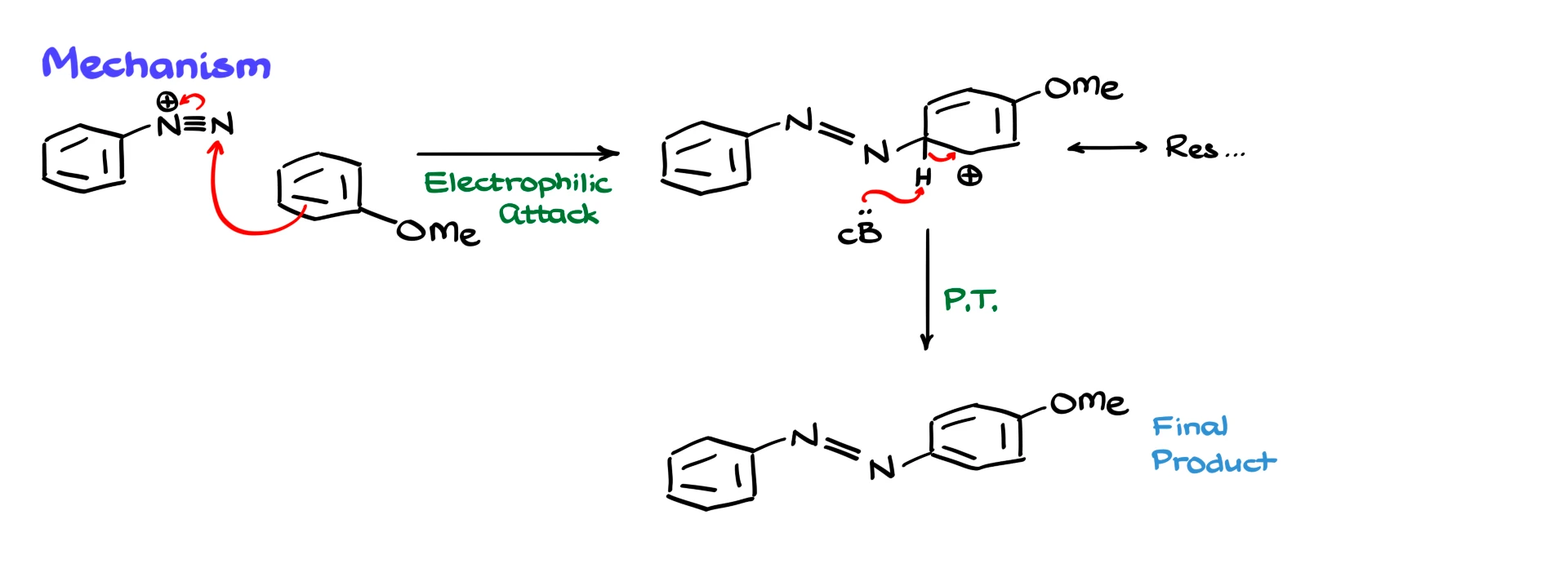
In an azo coupling reaction, the diazonium ion acts as the electrophile. For example, if we react a phenyldiazonium ion with anisole, the aromatic ring of anisole attacks the diazonium ion, forming a sigma complex. After formation of the carbocation intermediate, a base removes a proton, restoring aromaticity and giving the azo compound.
The mechanism follows the same general pattern as any electrophilic aromatic substitution: formation of a sigma complex, resonance stabilization, and deprotonation to restore aromaticity.
And that is essentially what you need to know about aryl diazonium ions: how to make them and how they react.
Which reaction surprised you the most? Let me know in the comments below.
