Structure and Properties of Alcohols
In this tutorial, I want to talk about the structure and properties of alcohols.
So, first of all, what exactly is an alcohol?
We define an alcohol as a molecule that has an –OH group connected to an sp³ hybridized carbon, and that carbon shouldn’t have any other heteroatoms or functional groups attached. For example, these three molecules are all alcohols:
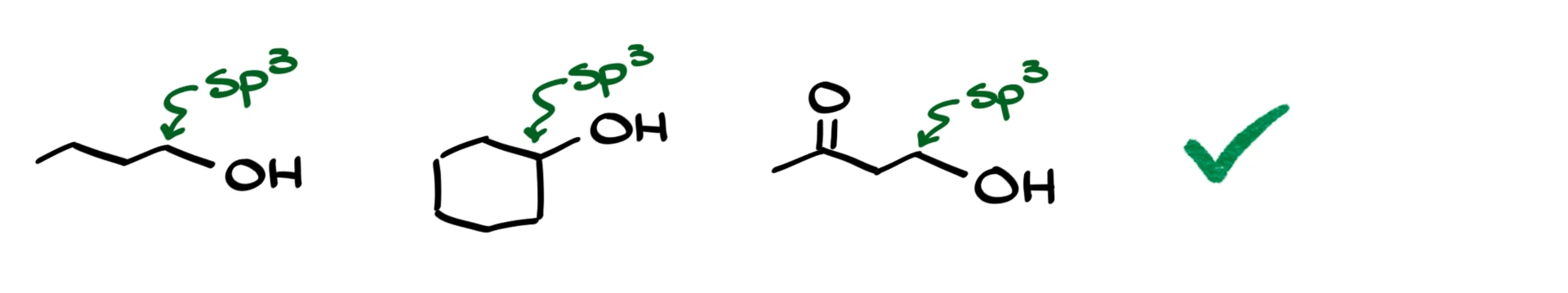
We have butanol as the first one, where the –OH is bonded to an sp³ hybridized carbon. Next is cyclohexanol, which also fits the definition. Then there’s a hydroxy ketone, which still counts as an alcohol because the –OH is connected to an sp³ hybridized carbon, and the ketone group is further down the molecule, it’s not attached to the same carbon as the –OH.
That last detail is really important. Just because a molecule contains an –OH group doesn’t automatically make it an alcohol. Let’s look at three more examples.

These next molecules all have an –OH group, but none of them are alcohols. In the first one, the –OH is connected to an sp² hybridized carbon in an aromatic ring—that makes it a phenol. The second one has the –OH next to a carbonyl group, making it a carboxylic acid. In the third example, the –OH is part of a molecule with a double bond, so it’s an enol.
Remember, a functional group is a specific arrangement of atoms that gives a molecule its unique chemical behavior. So, not every compound with an –OH group behaves the same. For example, phenols and alcohols both contain –OH groups, and they do share some chemical similarities, but each also has unique reactivity. Phenols can undergo some reactions that alcohols can’t, and the reverse is true as well. The same goes for carboxylic acids and enols.
Bottom line: not everything with an –OH group is an alcohol. For a molecule to qualify, that –OH must be attached to an sp³ hybridized carbon, and that carbon can’t have any other functional groups on it.
Physical Properties of Alcohols
Compared to many other organic molecules, alcohols are highly polar and have unusually high boiling points. That’s all thanks to the –OH group, which can participate in hydrogen bonding.

To show how drastic this effect is, look at these three molecules. Ethane, a simple alkane, boils at –89 °C, so it’s a gas at room temperature. Chloroethane boils at 12 °C—also a gas. But ethanol, which has that –OH group, has a boiling point of 78 °C. That’s a liquid at room temperature.
Even though I have a full tutorial on hydrogen bonding, here’s a quick reminder: hydrogen bonding happens when hydrogen is sandwiched between two electronegative elements like fluorine, oxygen, or nitrogen. Alcohols can act both as hydrogen bond donors and acceptors.
And because of that, alcohols are also pretty soluble in water. You’ve probably heard of the “four-carbon rule.” That just means that for every –OH group in a molecule, it can typically keep up to four carbon atoms soluble in water.

Take this lineup of alcohols—from methanol to pentanol. Methanol (1 carbon), ethanol (2 carbons), and propanol (3 carbons) are all completely miscible with water. But once we hit butanol (4 carbons), things change. Butanol isn’t fully miscible anymore, you can only dissolve about 73 grams per liter. Then with pentanol (5 carbons), the solubility drops even more.
The structure of the carbon chain also matters. A bulkier, more branched group makes it harder for molecules to interact with each other. That affects both solubility and boiling points.
Take the four isomers of butanol: we go from the straight-chain 1-butanol to the most branched isomer, tert-butanol. As branching increases, the boiling point drops—from 118 °C for 1-butanol down to just 82 °C for tert-butanol.
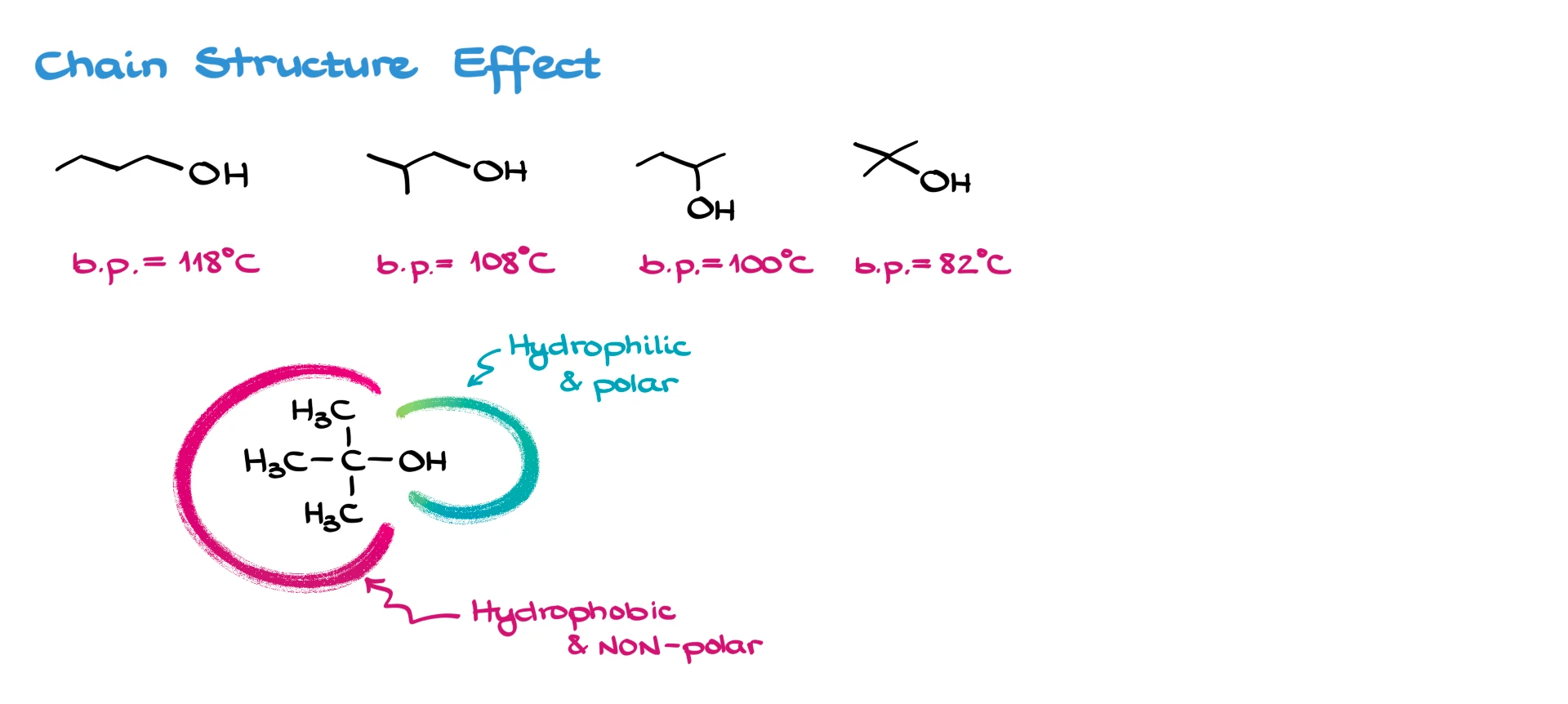
Why? Because that bulky hydrophobic (non-polar) region makes it harder for the molecules to form strong interactions with each other. I’ll redraw it here: the red section represents the hydrophobic part, and the blue section is the hydrophilic (polar) –OH. The bulkier that red part gets, the less the blue parts can interact. Weaker interactions mean lower boiling points.
Acid-Base Properties of Alcohols
Alcohols can act as weak acids. The typical pKa values for alcohols fall between 16 and 18. That’s weak, but if you use a strong enough base, you can deprotonate them.

For example, in reactions like the Williamson ether synthesis, you’ll want full deprotonation. A strong base like sodium hydride (NaH) works well here. The equilibrium constant for that deprotonation is about 10¹⁹, so it heavily favors the products. Plus, H₂ gas is released, which escapes the system and drives the reaction forward.
You can also use alkali metals like sodium, lithium, or potassium instead of their hydrides. The reaction will still produce an alkoxide and hydrogen gas, but now you’re looking at a redox reaction, not an acid–base one. In this case, the metal gets oxidized from an oxidation state of 0 to +1.
Factors Affecting Acidity of Alcohols
When it comes to alcohol acidity, let’s review the main factors that affect it.
Normally, things like resonance, atomic size, hybridization, or electronegativity can impact acidity. But for alcohols, where the –OH is on an sp³ hybridized carbon, those factors don’t apply.

The only major factor that affects alcohol acidity is inductive effects. Let me give you an example.
Compare ethanol with trifluoroethanol. Both are similar, but trifluoroethanol has three fluorine atoms on the second carbon. Fluorine is highly electronegative and pulls electron density away from the rest of the molecule. That stabilizes the negative charge on the conjugate base after deprotonation, making trifluoroethanol a stronger acid.
Ethanol has a pKa around 16. Trifluoroethanol is closer to 12.5. Since pKa is logarithmic, that’s more than a 1000-fold difference in acidity!
Of course, other functional groups with –OH like phenols and carboxylic acids can be affected by resonance or atomic size. But for simple alcohols, inductive effects are the main player.
Solvation Effect
Remember that diagram from earlier with the polar and non-polar regions of alcohols? Imagine removing the proton from tert-butanol. Now the oxygen has a negative charge, and it’s surrounded by solvent molecules—usually more alcohol molecules. That creates a solvation shell.
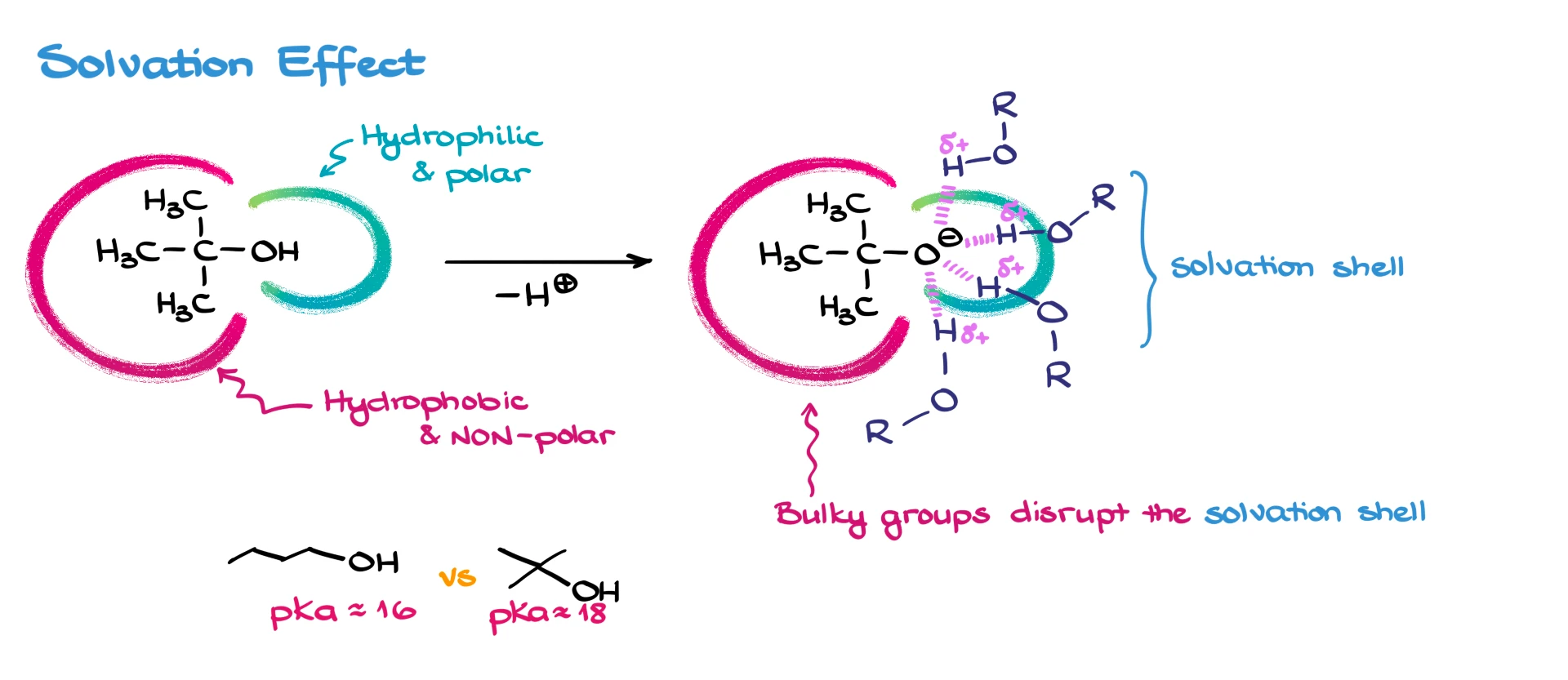
But here’s the catch: the bulkier that hydrophobic group is, the less effective solvation becomes. That means the negative charge on the oxygen isn’t stabilized as well, and the molecule becomes less acidic.
So, bulky alcohols like tert-butanol tend to have higher pKₐ values. For example, linear 1-butanol has a pKa around 16, while tert-butanol can be closer to 17 or 18 depending on the source.
So if there are no obvious inductive effects, think about solvation. The bulkier the alcohol, the less acidic it’s likely to be.
