Free Radical Polymerization
In this tutorial I want to talk about radical polymerization.
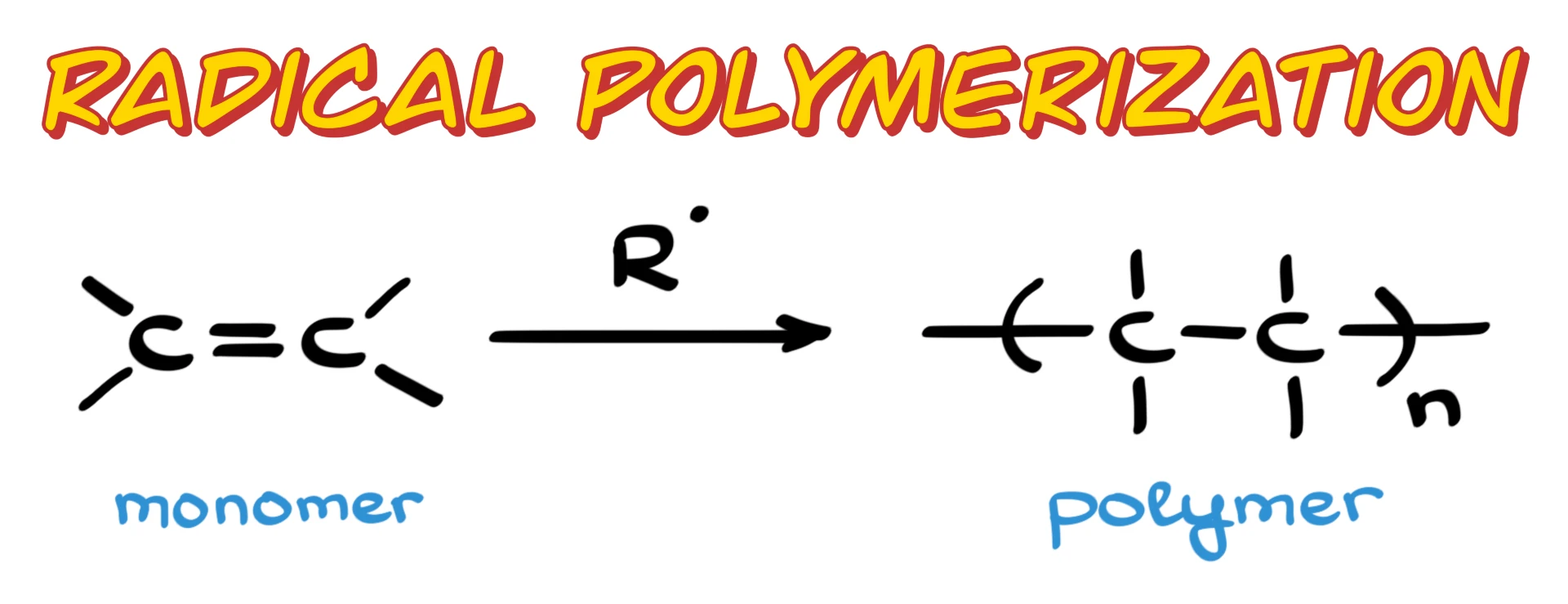
We’ll go through the full mechanism, starting with initiation, then propagation, and finally termination. I’ll also show you a unique feature of radical polymerization called branching, and we’ll wrap up with a few common examples you are very likely to see in class.
Initiation
Let’s start with the initiation step and the initiators commonly used in radical polymerization.
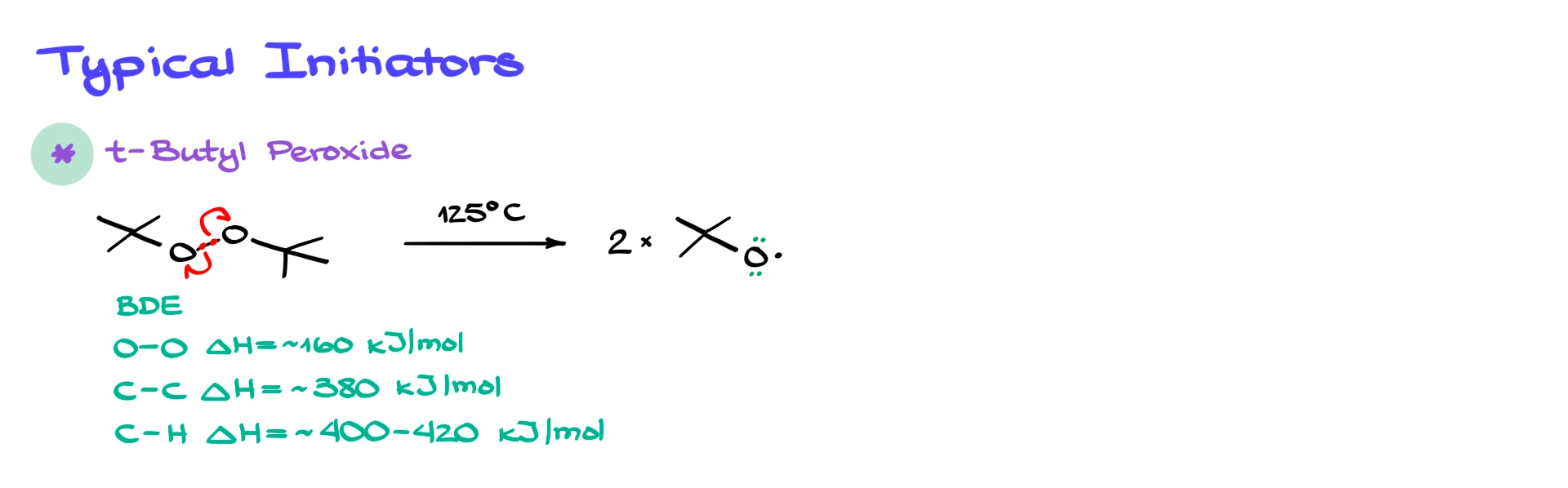
One of the most common initiators you will encounter is tert-butyl peroxide. Peroxides are popular initiators because the oxygen–oxygen bond is relatively weak. If you compare the O–O bond dissociation energy to a typical C–C or C–H bond, you’ll find that the O–O bond is roughly two to three times weaker. That means mild heating is enough to cause homolytic cleavage.
When tert-butyl peroxide is heated, the O–O bond breaks homolytically, and each oxygen takes one electron, giving two tert-butoxy radicals. Oxygen still has lone pairs, of course, but in radical mechanisms we usually ignore lone pairs unless they directly participate in bond making or breaking.
Since we are dealing with radical reactions, we use fishhook arrows. A full curved arrow represents movement of two electrons, while a fishhook represents movement of a single electron. I’m assuming you are already comfortable with this notation, so I won’t spend too much time on it.
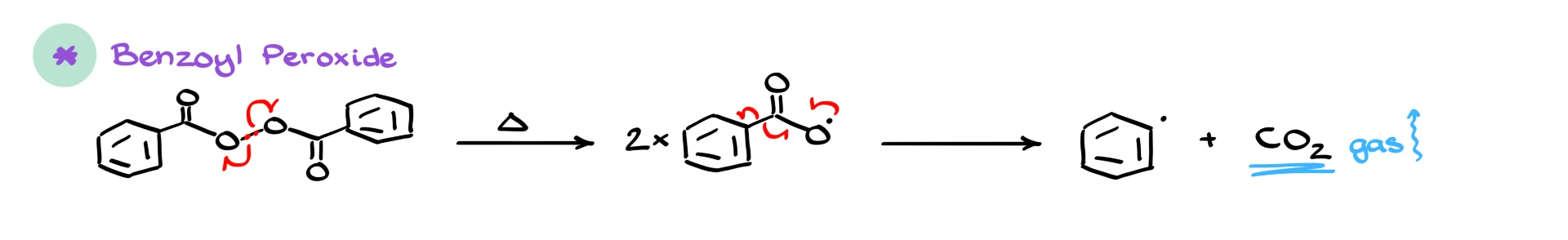
Another very common initiator is benzoyl peroxide. Just like tert-butyl peroxide, it contains an O–O bond that undergoes homolytic cleavage upon heating. This gives two benzoyloxy radicals. These radicals are not very stable, so each one quickly breaks apart further, releasing carbon dioxide and forming a phenyl radical. The formation of CO₂ gas makes this step extremely favorable thermodynamically, which is why benzoyl peroxide is such an effective radical initiator.
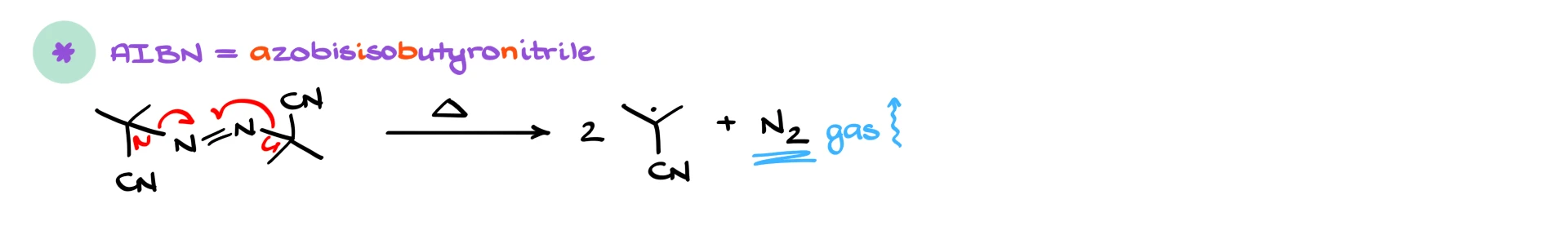
A third classic initiator is AIBN, short for azobisisobutyronitrile. Upon heating, AIBN breaks apart by cleaving two C–N bonds, releasing nitrogen gas and forming two carbon-centered radicals that contain nitrile groups. Again, loss of N₂ gas strongly drives this reaction forward.
So in practice, the radical that actually starts the polymer chain could be a tert-butoxy radical, a phenyl radical, a nitrile-substituted radical, or something similar. From a mechanistic standpoint, it does not matter which one it is. To keep things simple, I will just refer to all of them as R·, a generic radical initiator.
Propagation
Once initiation is complete, we move on to propagation.
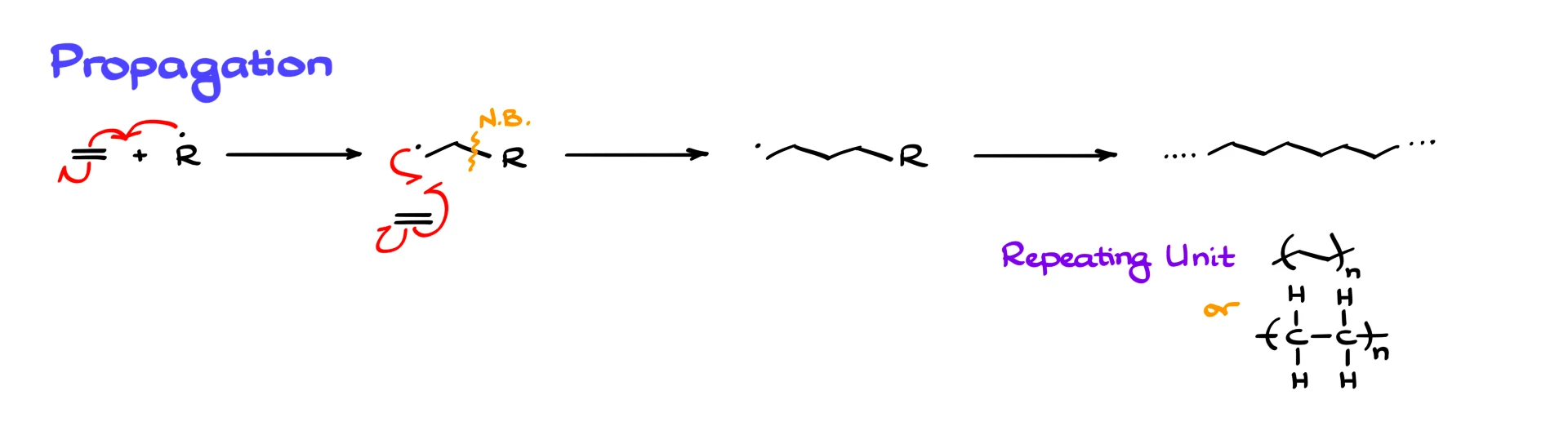
Let’s start with the simplest possible monomer, ethylene. The radical R· reacts with the double bond of ethylene. One electron comes from the radical, one comes from the π bond, and the remaining π electron stays on one of the carbons. This forms a new C–C bond and generates a new radical at the end of the chain.
At this point, there are no new reagents in the system except more ethylene, so the radical reacts with another ethylene molecule. The same three fishhook arrows appear again, and the chain grows longer, with the radical always ending up at the terminus of the growing polymer.
This process repeats over and over, producing a very long chain. In reality, we do not draw thousands of repeating units. Instead, we represent the polymer using a repeating unit in parentheses with a subscript n, or by drawing a shortened chain that implies repetition. You may also notice that we stop showing the original R group. Once the chain is very long, the identity of the initiator fragment is statistically insignificant and is ignored in polymer structures.
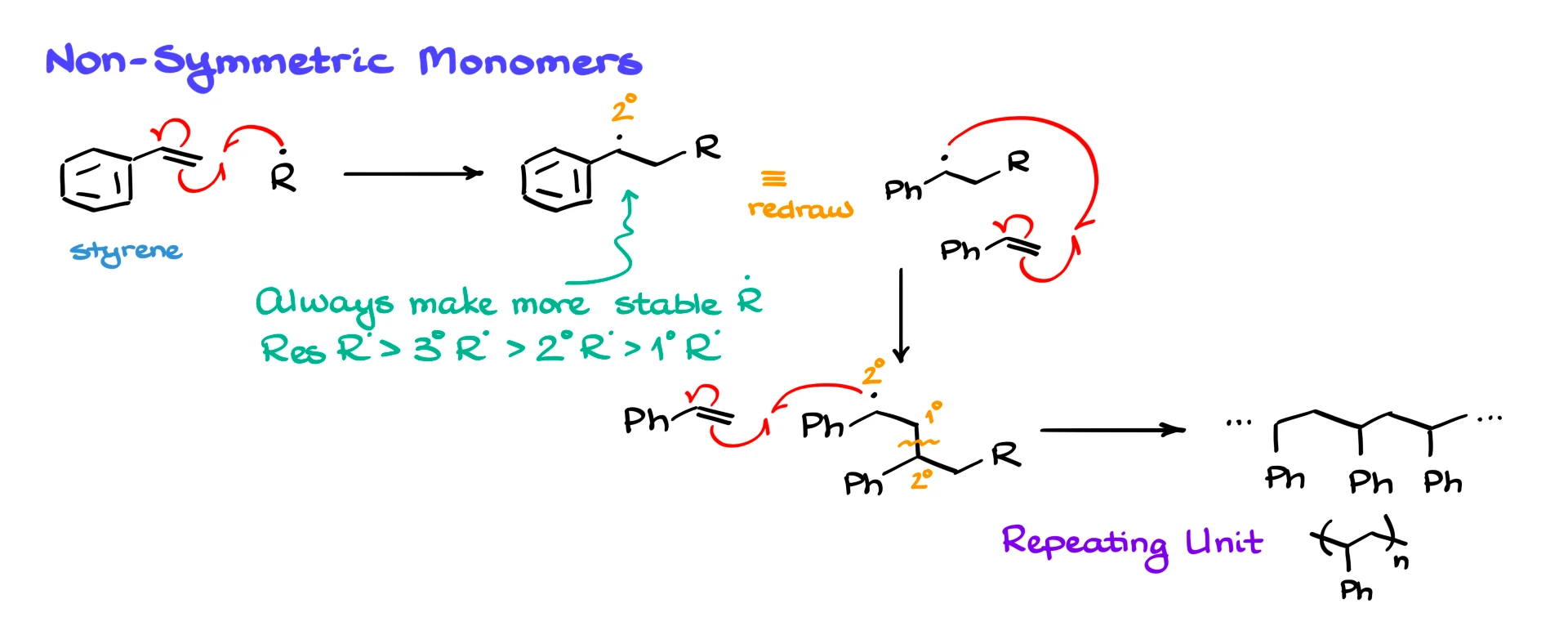
Now let’s look at what happens when the monomer is unsymmetrical, such as styrene. Mechanistically, the steps look the same, but there is one important rule you must follow. When a radical adds to a double bond, you must always draw the product that gives the most stable radical.
Radical stability follows the same trend as carbocation stability. Resonance-stabilized radicals are the most stable, followed by tertiary, secondary, and then primary radicals. In the case of styrene, addition occurs in a way that places the radical on the benzylic carbon, where it is stabilized by resonance with the aromatic ring.
Once that benzylic radical forms, it reacts with another equivalent of styrene in exactly the same way. Each propagation step creates a new bond and regenerates a benzylic radical at the end of the chain. Repeating this process gives polystyrene, whose repeating unit consists of a two-carbon backbone with a phenyl substituent.
Termination
Eventually, polymer growth must stop. This happens during the termination step.
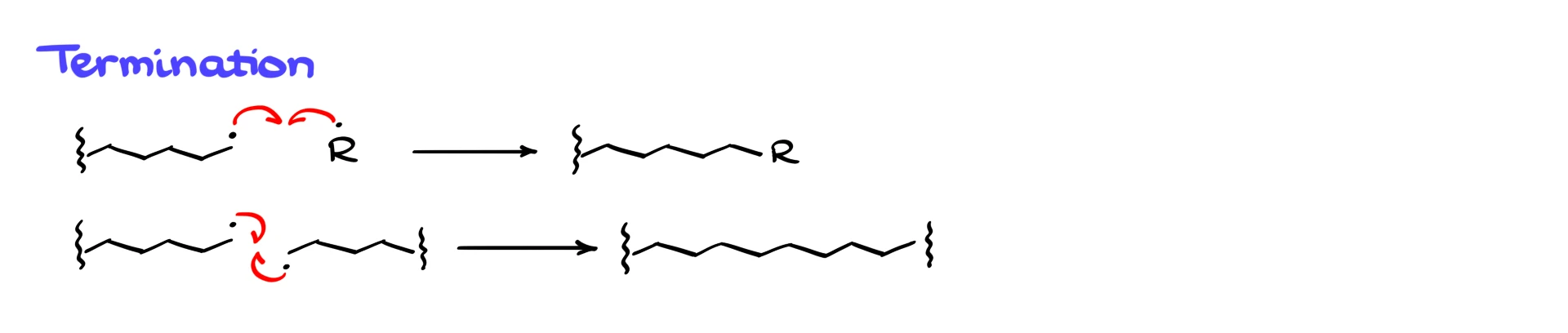
Termination can occur in several ways. A growing polymer radical can react with another radical initiator, or two growing polymer chains can combine, forming one longer chain. From a teaching standpoint, the exact termination mechanism is usually not critical, and many instructors are not very picky about how you draw it.
Polymer Branching in Radical Polymerization
Now let’s talk about a feature that is unique and extremely important in radical polymerization: branching.
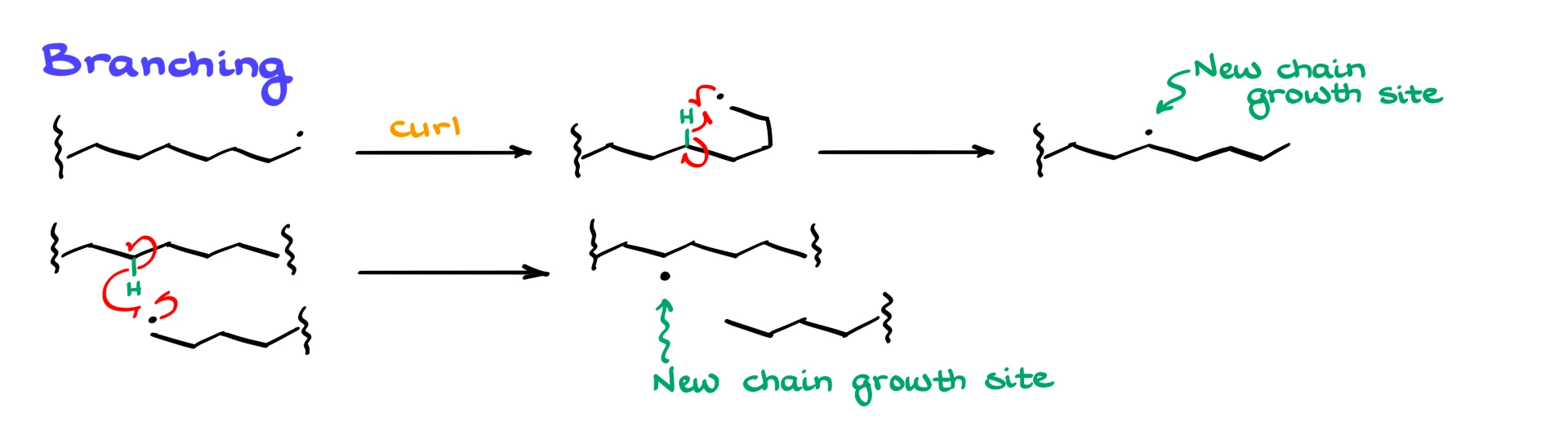
As polymer chains grow longer, they tend to coil and fold in space. That means a radical at the end of a chain can get close to a hydrogen atom somewhere along the same chain. Instead of reacting with another monomer, the radical can abstract that hydrogen. The original radical is quenched, but a new radical forms at the position where the hydrogen was removed.
Now the radical is no longer at the end of the chain. It is in the middle, and growth can continue from that point. This creates a branch. A similar process can happen between two different chains, where a radical abstracts a hydrogen from another polymer molecule, creating a new radical site and leading to branching.
By controlling reaction conditions, chemists can influence how much branching occurs. This is why polymers with the same molecular formula can have very different physical properties. A classic example is polyethylene. High-pressure polyethylene has lots of branching and is soft and flexible, while low-pressure polyethylene has much less branching and is stronger and more rigid.
Examples of Common Polymers
To wrap things up, here are a few common examples of polymers made by radical polymerization.
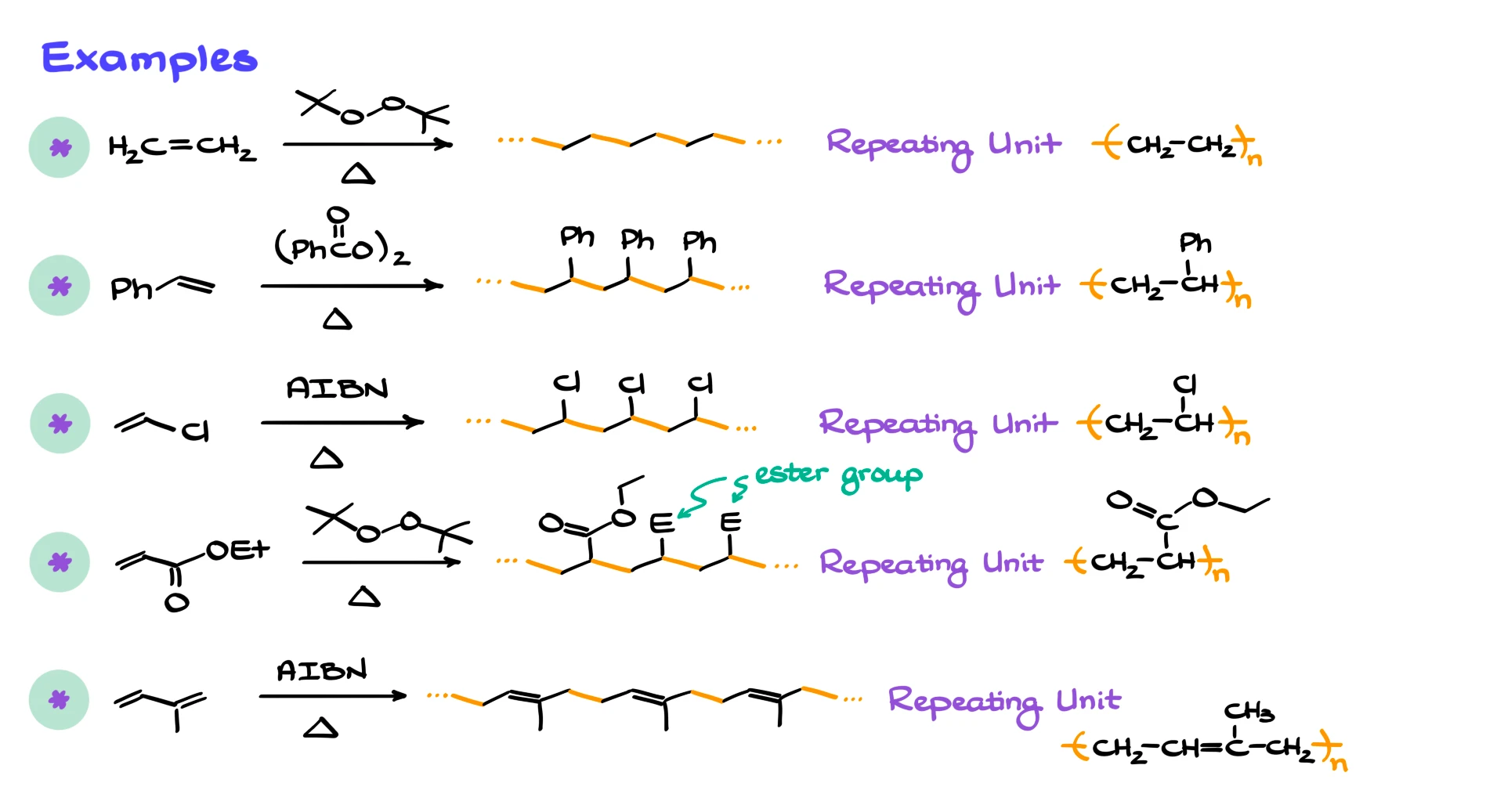
Polyethylene comes from ethylene. Polystyrene comes from styrene. Other very common examples include polyvinyl chloride, polyacrylates such as poly(ethyl acrylate), and polyisoprene, which is the main component of natural rubber.
As an exercise, I strongly encourage you to practice drawing the mechanisms for these polymerizations. One of them is very likely to show up on an exam.
