Mitsunobu Reaction
When it comes to activating alcohols, the first thing most people think about is sulfonate esters like tosylates, mesylates, or triflates. They work, but for a bunch of practical reasons they are not always the best option. Luckily, we have a powerful alternative called the Mitsunobu reaction, first reported by Oyo Mitsunobu in 1967. It quickly became one of the most versatile alcohol activation methods in synthesis, especially in total synthesis.

In this tutorial I will go over what the Mitsunobu reaction does, how the mechanism works, the major benefits and drawbacks, and then I will show a few useful synthetic applications.
Mitsunobu Reaction Components
The Mitsunobu reaction is a multicomponent reaction.
You combine an alcohol, an acidic pronucleophile, and two reagents, DEAD and triphenylphosphine. The alcohol is typically primary or secondary. Tertiary alcohol examples exist, but they are exceptions rather than the rule.
The pro-nucleophile is something that becomes nucleophilic after it loses a proton. For example, a carboxylic acid becomes a carboxylate, and that carboxylate is the nucleophile. In general, Mitsunobu works best when the pronucleophile has a pKa of about 11 or lower, and the reason for that becomes clear once we look at the key acid–base steps in the mechanism.
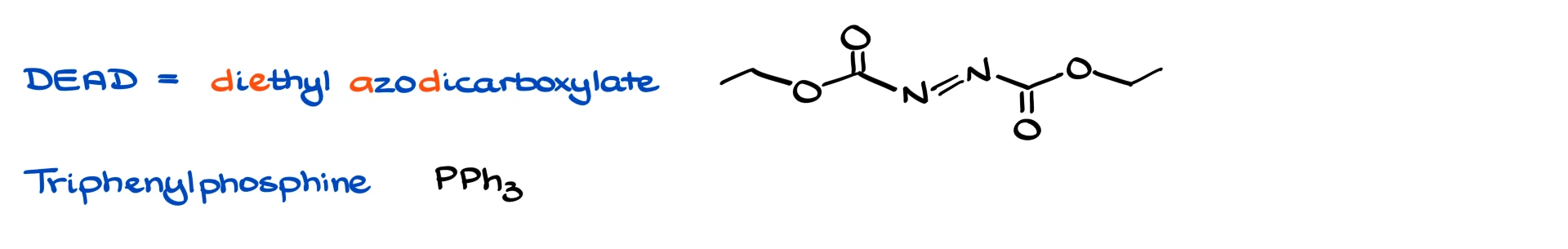
The two classic Mitsunobu reagents are DEAD and triphenylphosphine. DEAD stands for diethyl azodicarboxylate. There are many variations of this reagent family, but DEAD is the iconic one you will see most often. Triphenylphosphine is the other key component, and you have almost certainly seen it before in other contexts.
Mitsunobu Reaction Mechanism
Now let’s go through the mechanism.
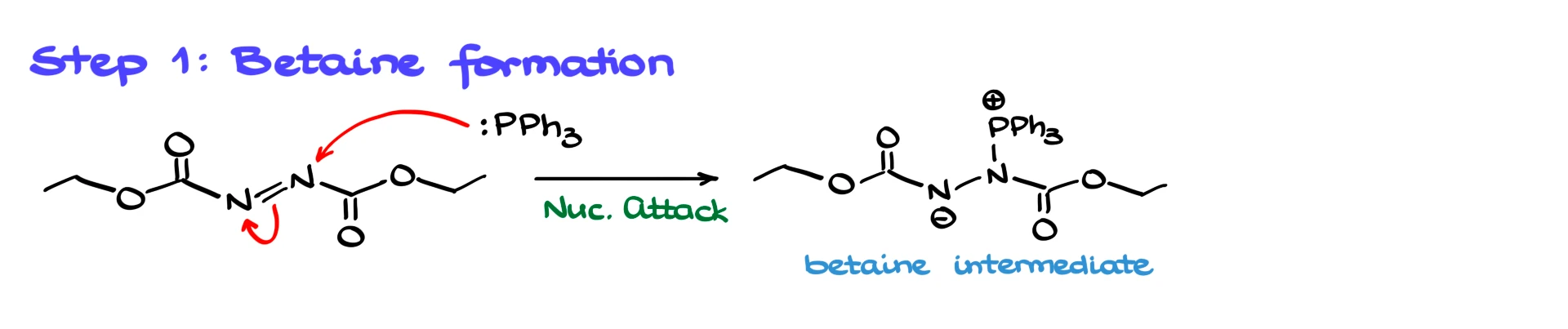
The first step is formation of a betaine intermediate. Triphenylphosphine is a strong nucleophile. DEAD has an electrophilic N=N system that is activated by the two electron-withdrawing carbonyl groups. Triphenylphosphine attacks one of the nitrogens, pushing electron density onto the other nitrogen and forming the betaine. The negative charge on nitrogen is resonance stabilized by the adjacent carbonyl group, which is why this step happens readily.
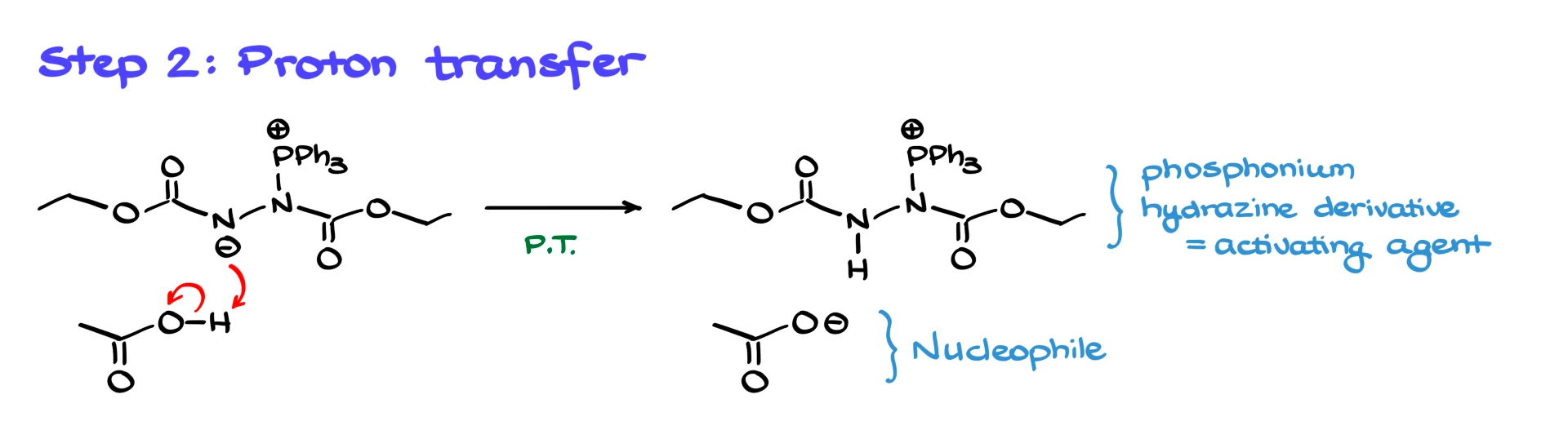
Next we bring in the pro-nucleophile, and I will use acetic acid as the example. The betaine deprotonates the acid. That gives us two important species. We generate the nucleophile, the acetate anion, and we also form a hydrazine-derived intermediate, sometimes described as the key “activating agent” in Mitsunobu chemistry. The nucleophile sits in the background for a moment, but it will come back in the final substitution step.
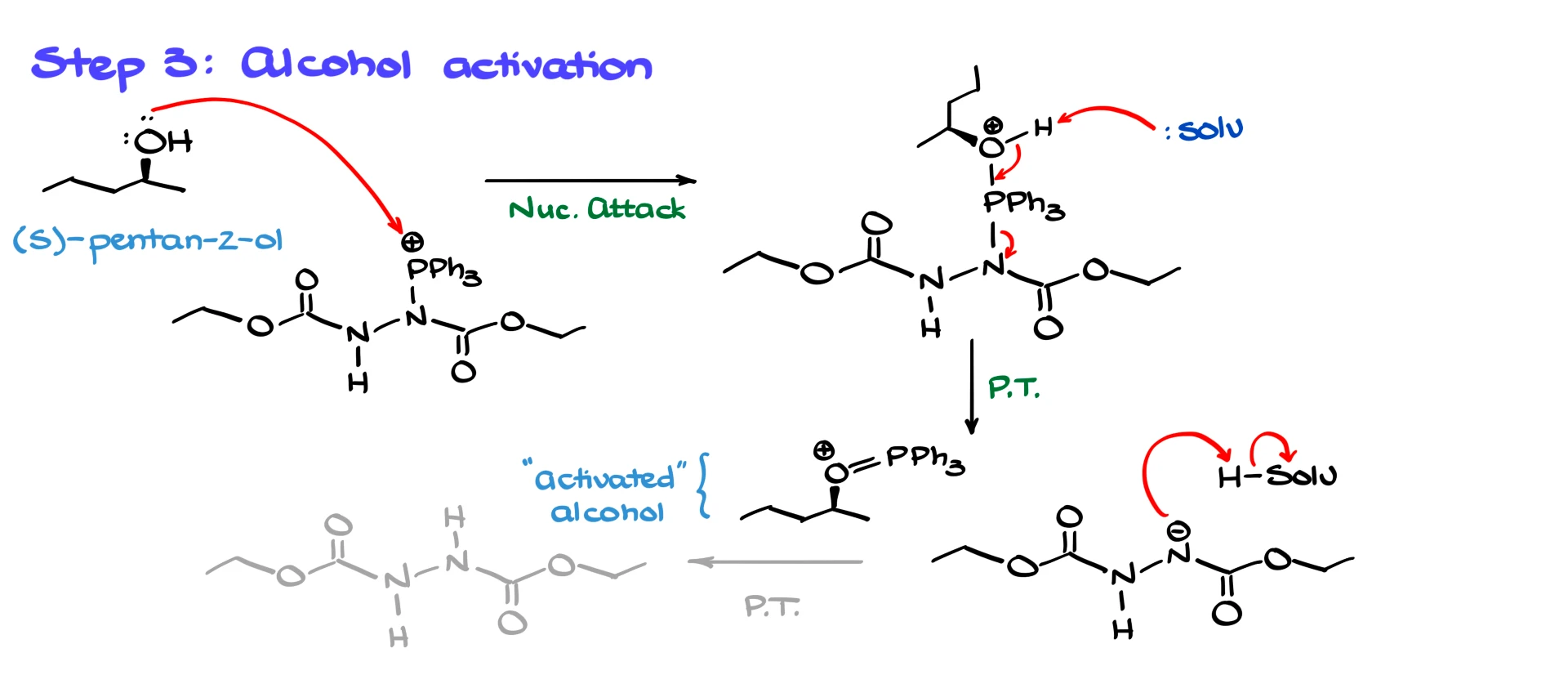
Now we move to alcohol activation. The activating agent is electrophilic, and the alcohol acts as a nucleophile. The alcohol attacks phosphorus, giving an alkoxyphosphonium-type intermediate. Then a base, usually the conjugate base present in the mixture or the solvent acting as a proton acceptor, removes the proton. That triggers a cascade that breaks the intermediate apart and produces the activated alcohol species that will undergo substitution.
At this point there is a critical detail. One of the nitrogen-containing intermediates can be nucleophilic if the medium is not acidic enough. If it is allowed to compete as a nucleophile, it can attack the activated alcohol and create side products. This is one major reason the pro-nucleophile needs to be acidic enough in the first place. We want proton transfers to happen efficiently so that the “wrong” nucleophile is kept under control.
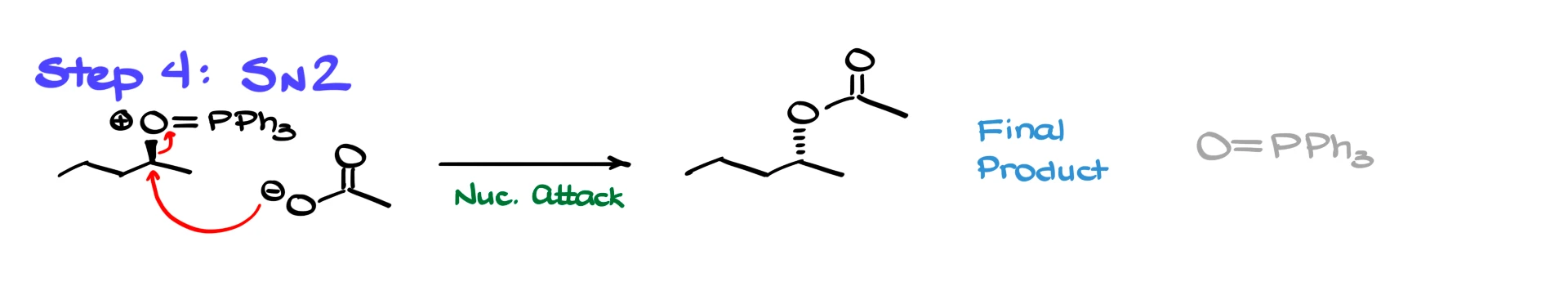
Now for the last step, the part everyone likes. The nucleophile we generated earlier, in this example acetate, performs a clean SN2 reaction on the activated alcohol. It kicks out the leaving group and forms the substituted product, which in this case is an ester. The phosphorus-containing side product ends up as triphenylphosphine oxide, which is one of the thermodynamic drivers of the reaction because forming the P=O bond is highly favorable.
If we recap the flow, we first form the betaine, then deprotonate the pro-nucleophile to generate the nucleophile and the activating agent. The activating agent converts the alcohol into a great electrophile, and then the nucleophile does SN2 to give the product.
Mitsunobu Reaction Benefits
Even though the mechanism looks complicated on paper, the actual experimental setup is often simple. It is usually run as a one-pot procedure where you combine all four components in one flask. Sometimes you have to be careful about the order of addition depending on the substrates, but the overall workflow is still straightforward.
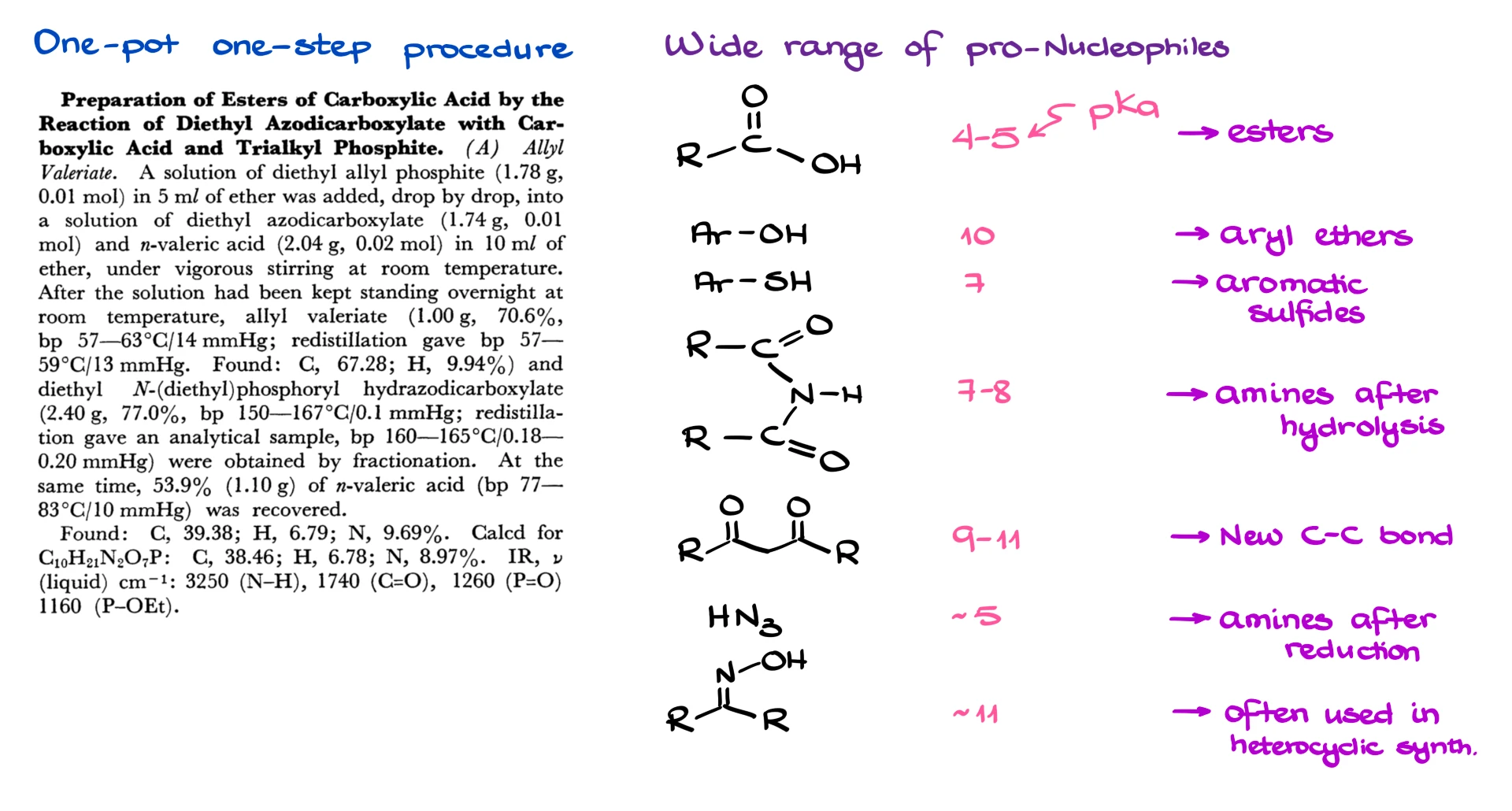
One of the biggest advantages of the Mitsunobu reaction is how broad the pronucleophile scope can be. Carboxylic acids work, phenols work, thiophenols work, imides work, β-dicarbonyl compounds can work, and the list goes on. Depending on what pro-nucleophile you choose, you can build esters, ethers, thioethers, and in many cases you can even create new carbon–carbon bonds.
Mitsunobu Reaction Drawbacks
Of course, there are drawbacks. One major issue is side reactions involving the nucleophilic intermediates that form during activation.
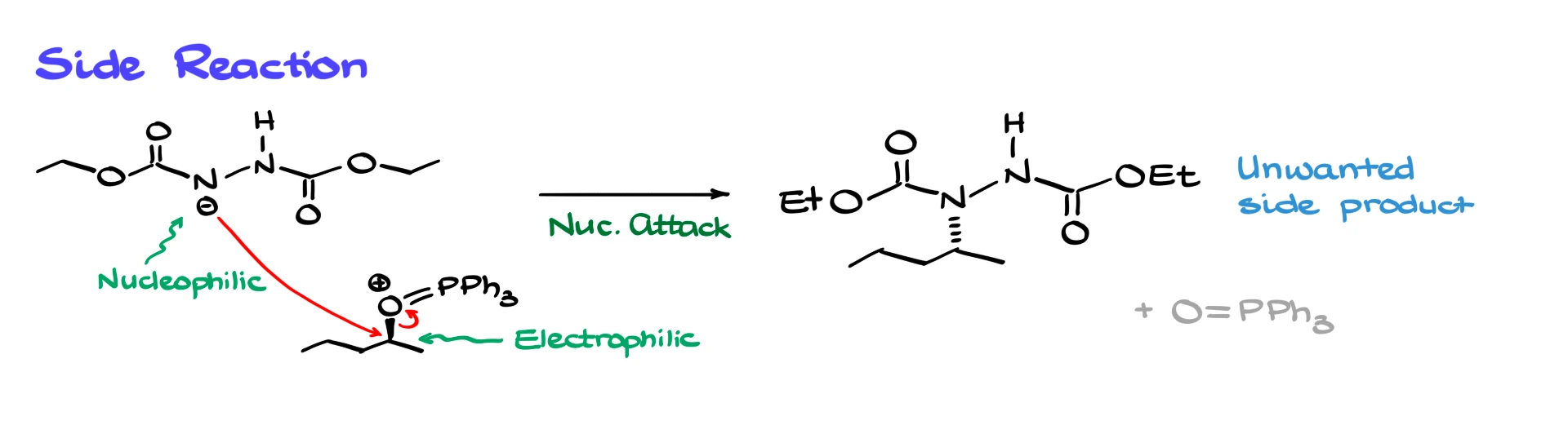
The activated alcohol is strongly electrophilic, and if the undesired nucleophile attacks it, you lose yield and form messy byproducts. Because of this, Mitsunobu yields are often not amazing. Depending on the substrates you might see yields from the high 30s to the mid 80s, with many reactions landing around 60 to 70 percent. Not terrible, but not stellar either.
Synthetic Applications of Mitsunobu Reaction
Despite that, Mitsunobu is still widely used because it solves problems that are difficult to solve cleanly in other ways.
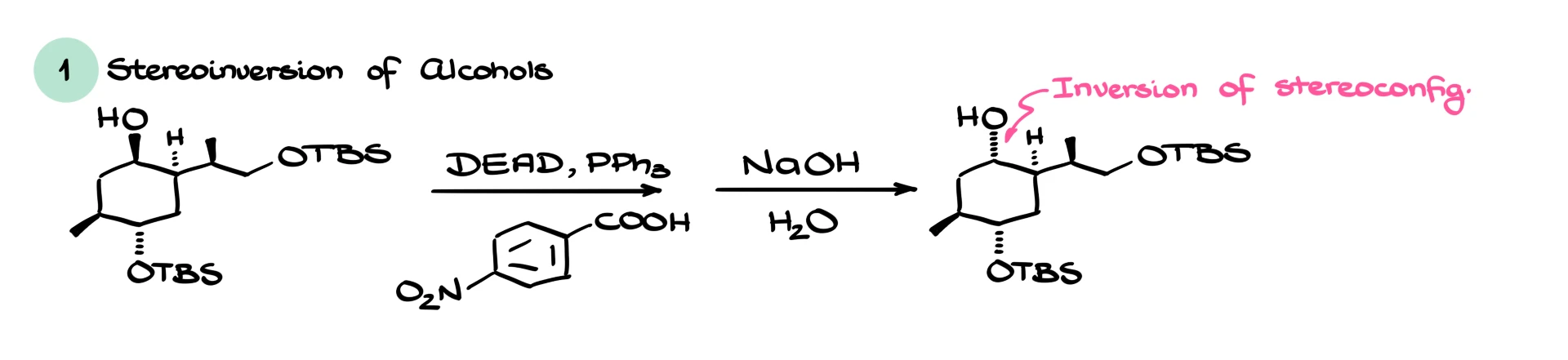
One classic application is stereochemical inversion of an alcohol. If you start with a chiral alcohol and you want the opposite stereochemistry, you can run a Mitsunobu reaction with a carboxylic acid to form an ester with inversion, then hydrolyze that ester under basic conditions to regenerate the alcohol. You end up with the same alcohol skeleton but inverted configuration at the stereocenter.
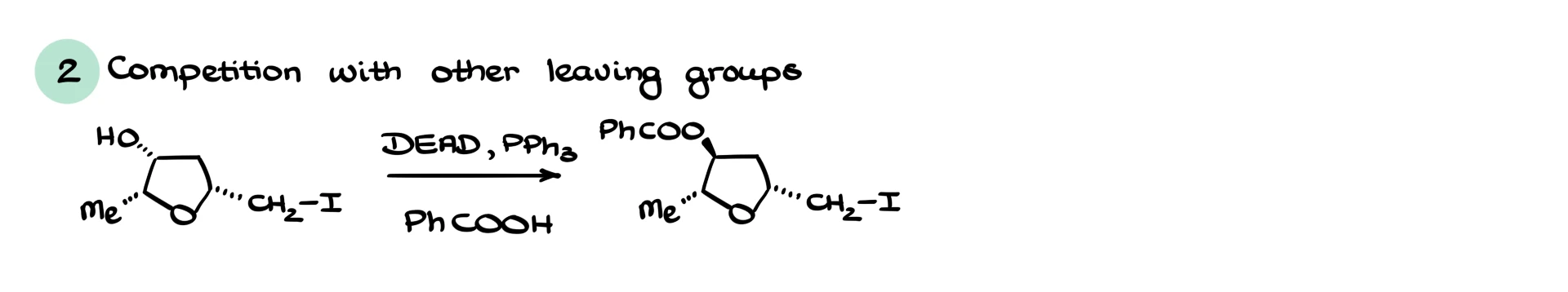
Another major advantage is chemoselectivity. The leaving group generated in Mitsunobu chemistry is exceptionally good. You can sometimes substitute a secondary alcohol through Mitsunobu even in the presence of other leaving groups, including a primary iodide. That is impressive because iodide is normally one of the best leaving groups around. Mitsunobu often targets the alcohol you want, leaving other functional groups intact for later steps.
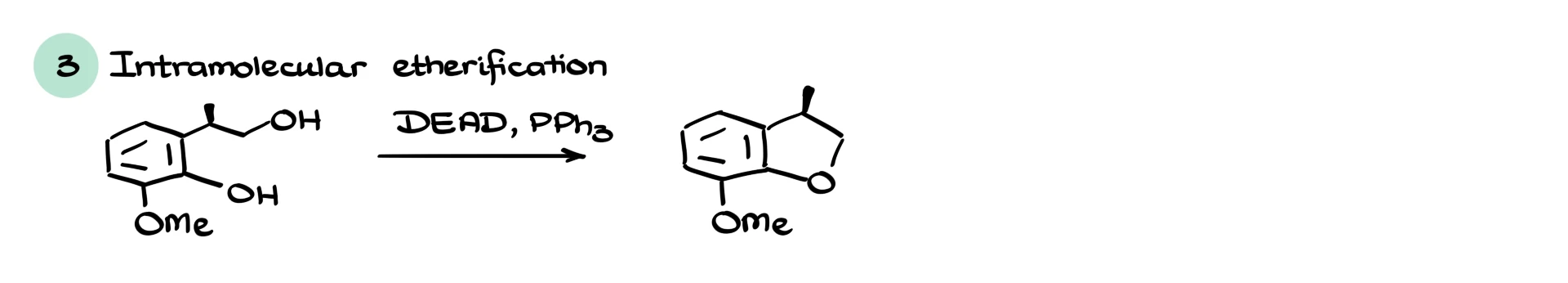
A final application that shows up in synthesis is intramolecular ether formation. Making cyclic ethers, especially non-aromatic ones, can be challenging. While Mitsunobu generally does not couple two separate alcohols together efficiently, intramolecular versions can work very well, which makes it a useful ring-closing strategy in the right substrate.
So that is the Mitsunobu reaction. It is a flexible, often mild way to activate alcohols and replace OH with a wide variety of nucleophiles, usually with inversion of configuration. It has quirks, it can be moody, and the yields are not always stunning, but when it is the right tool, it is hard to beat.
