Beckmann Rearrangement
In this tutorial I want to talk about the Beckmann rearrangement.
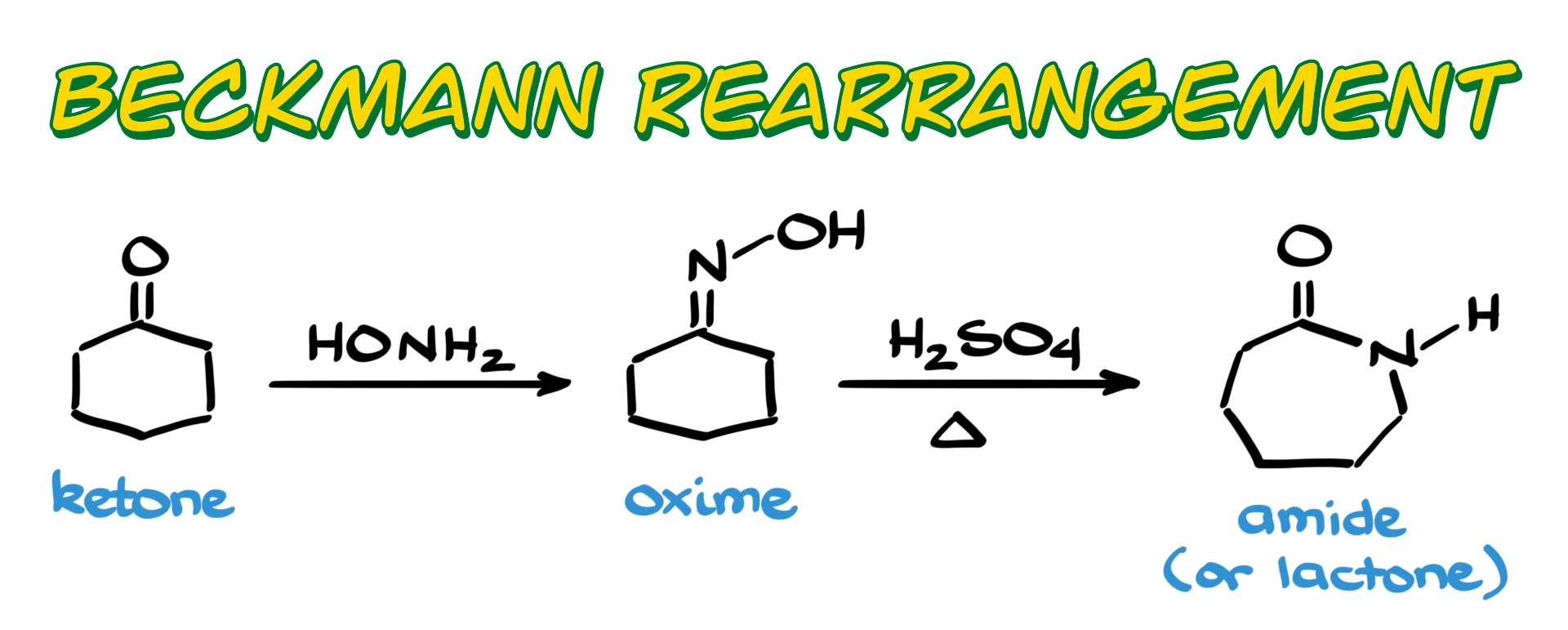
This is a reaction where we typically start with a ketone or, more commonly, an oxime and convert it into the corresponding amide, or a lactam in the case of cyclic systems.
Oxime Formation
Since ketones are very common starting materials for Beckmann rearrangements, let’s begin with a ketone example, benzophenone.
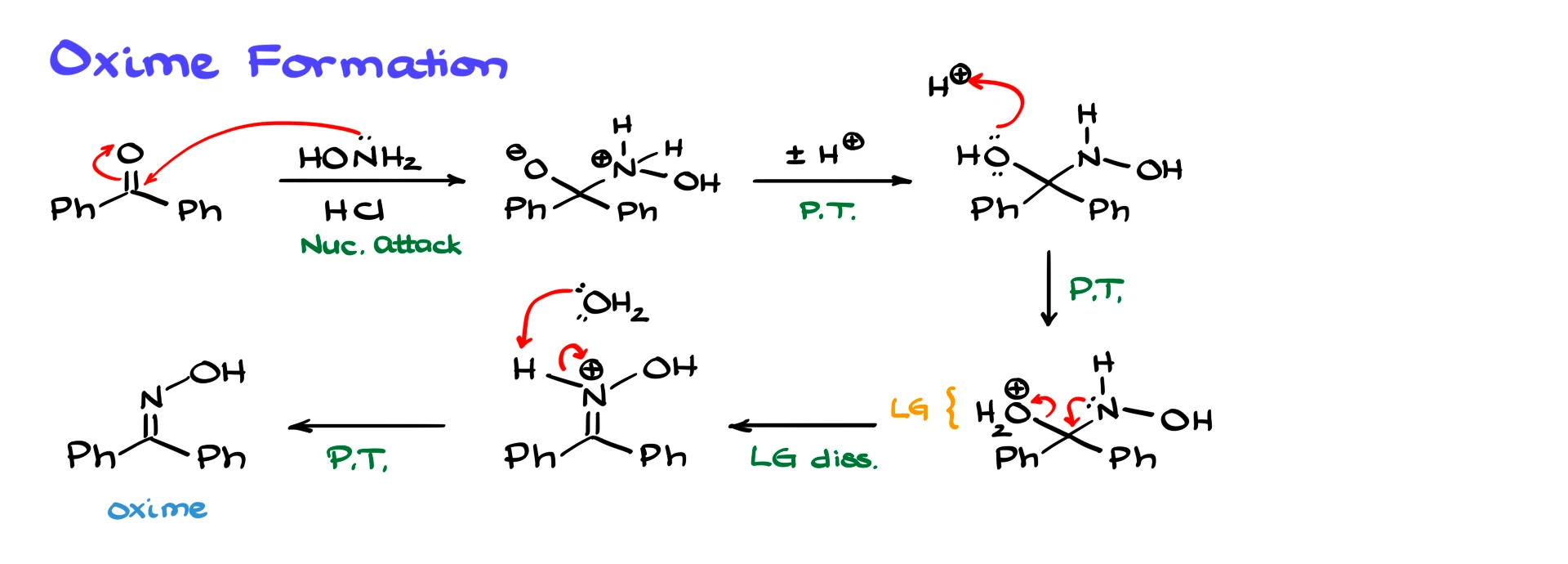
The first step in the overall process is oxime formation. Mechanistically, oxime formation follows the same logic as imine formation, and I cover that in detail in a separate tutorial, so here I will only go through it briefly.
We start with hydroxylamine. The nitrogen acts as a nucleophile and attacks the carbonyl carbon of the ketone, giving a tetrahedral intermediate. A series of proton transfers then occurs, which I will combine into one step for simplicity. We then protonate the OH group using an acid such as HCl, converting it into a good leaving group. Once that happens, the nitrogen lone pair helps push the leaving group out, forming the C=N bond. Finally, water or another base removes a proton, giving us the oxime. Up to this point, this is essentially an imine formation mechanism.
Beckmann Rearrangement Mechanism
Now that we have the oxime, we can move on to the Beckmann rearrangement itself.
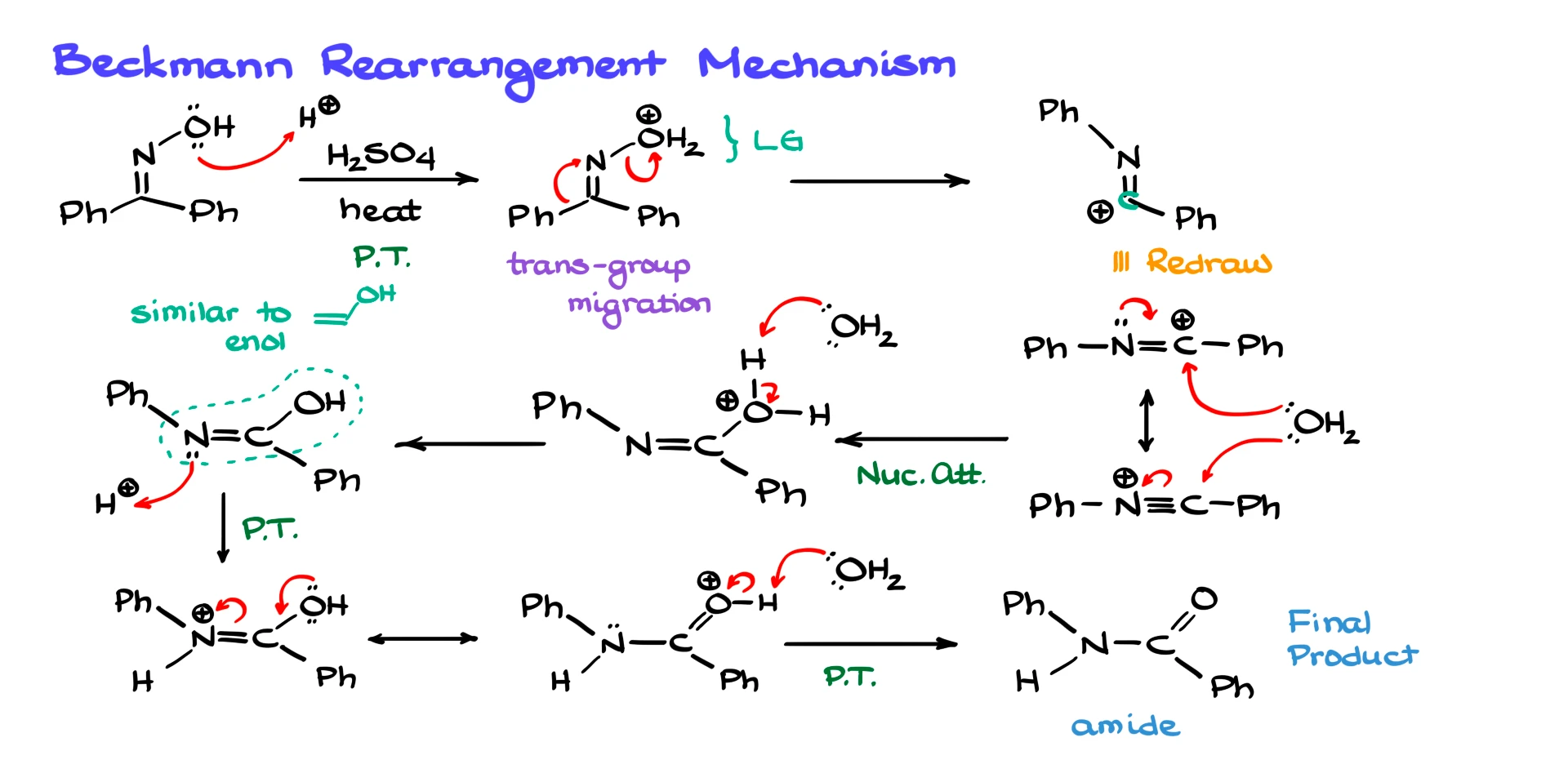
This step is typically carried out in strongly acidic conditions, most commonly concentrated sulfuric acid, although other options exist. Under these acidic conditions, the first thing that happens is protonation of the oxime oxygen, giving us an oxonium species. At this point we have H₂O⁺ attached to nitrogen, which is an excellent leaving group.
Next comes the key rearrangement step. As the leaving group departs, one of the substituents on the carbon migrates onto the nitrogen. A very important rule here is that the group trans to the leaving group is the one that migrates. In this particular example we have two phenyl groups, so it does not matter which one moves. However, if we had two different groups, X and Y, the group trans to the leaving group would be the one that migrates. If the leaving group were oriented on the opposite side, then the other substituent would migrate instead.
This selectivity is closely tied to oxime geometry. Oxime formation is usually under thermodynamic control, and as a result the leaving group often ends up trans to the bulkier substituent, though that is not a hard rule in every system.
To visualize the rearrangement, we can redraw the unchanged part of the molecule and then show the migrating phenyl group forming a new bond to nitrogen. The carbon that lost the phenyl group now bears a positive charge. This carbocation is directly adjacent to a nitrogen atom with a lone pair, so it is strongly resonance-stabilized.
Because we are dealing with a highly electrophilic intermediate, the nucleophile we just expelled, water, immediately attacks. It does not matter which resonance form you use to draw this step, both representations lead to the same outcome. After nucleophilic attack, we obtain a protonated intermediate, which is quickly deprotonated by water or another base in the system.
At this stage the structure may look a bit like an enol, except now it involves a C=N bond with an OH group attached. Just like enols, this form is not very stable, so the molecule undergoes tautomerization. Protonation followed by electron rearrangement restores the carbonyl, and a final deprotonation gives the amide as the product.
So let’s recap the Beckmann rearrangement mechanism. First, the oxime OH group is converted into a good leaving group under acidic conditions. Then the leaving group departs and the substituent trans to it migrates onto nitrogen. This generates a resonance-stabilized cationic intermediate, which is immediately attacked by water. A series of proton transfers and a tautomerization then lead to the amide. This mechanism pulls together ideas from both first- and second-semester organic chemistry, so it is a favorite topic for exams.
Beckmann Rearrangement Variations
There is, however, a practical problem with the classical Beckmann rearrangement. It usually requires concentrated sulfuric acid and elevated temperatures. Heat is often manageable, but concentrated sulfuric acid can be harsh and incompatible with many functional groups. Because of this, several useful variations of the Beckmann rearrangement have been developed.
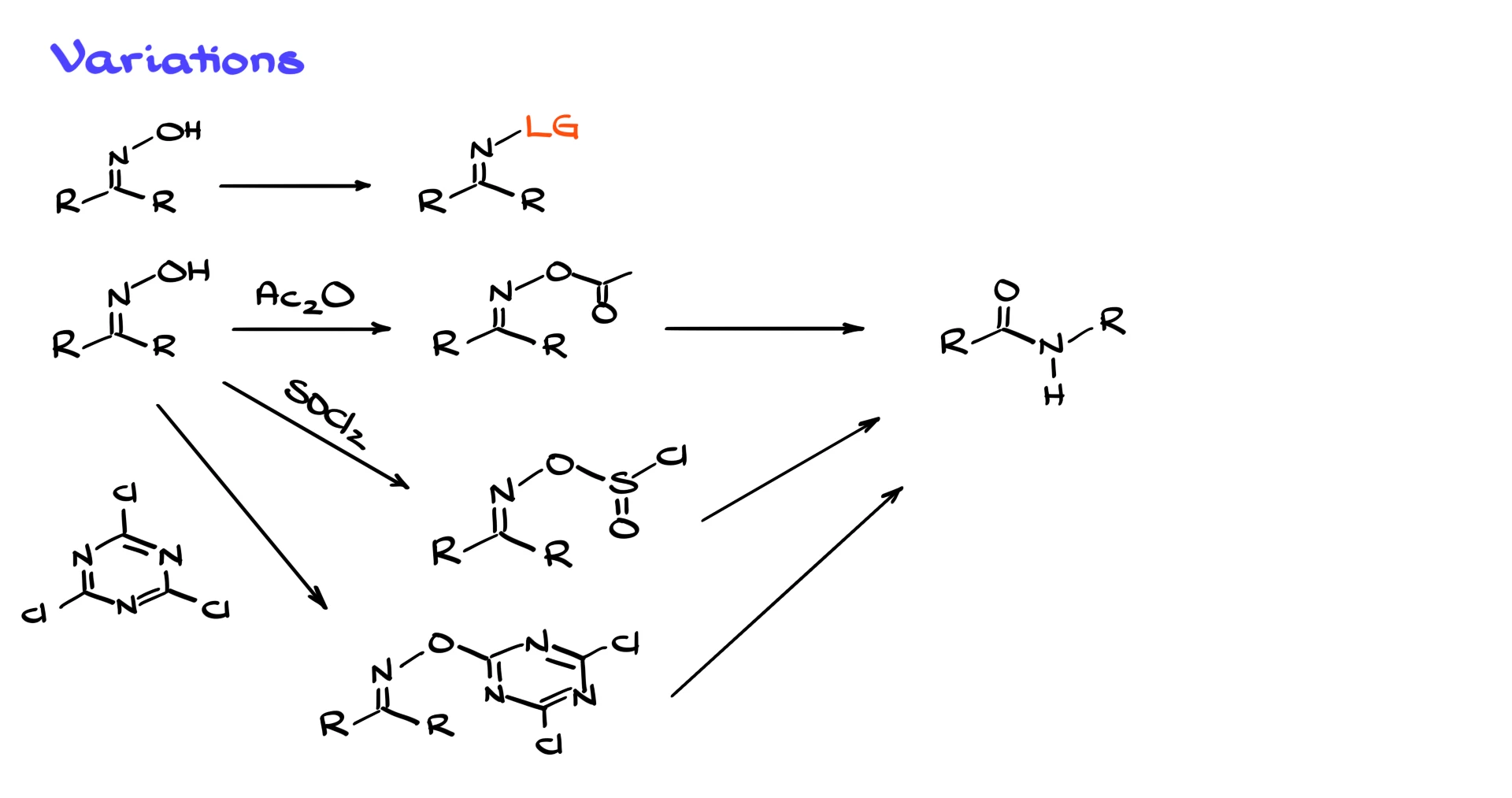
The key requirement is converting the oxime OH into a good leaving group. One classic alternative is to use an acid anhydride, which converts the OH into an acetate, a much better leaving group. Another option is thionyl chloride, which is very effective but also unpleasant to work with. A friendlier alternative is cyanuric chloride, which accomplishes the same goal without the eye-watering side effects. Regardless of the method used to activate the OH group, these reactions proceed through the same Beckmann-style rearrangement and give the corresponding amide after aqueous workup.
Beckmann Fragmentation
While the Beckmann rearrangement is generally reliable and often gives good yields, there is one major complication to be aware of, known as Beckmann fragmentation.
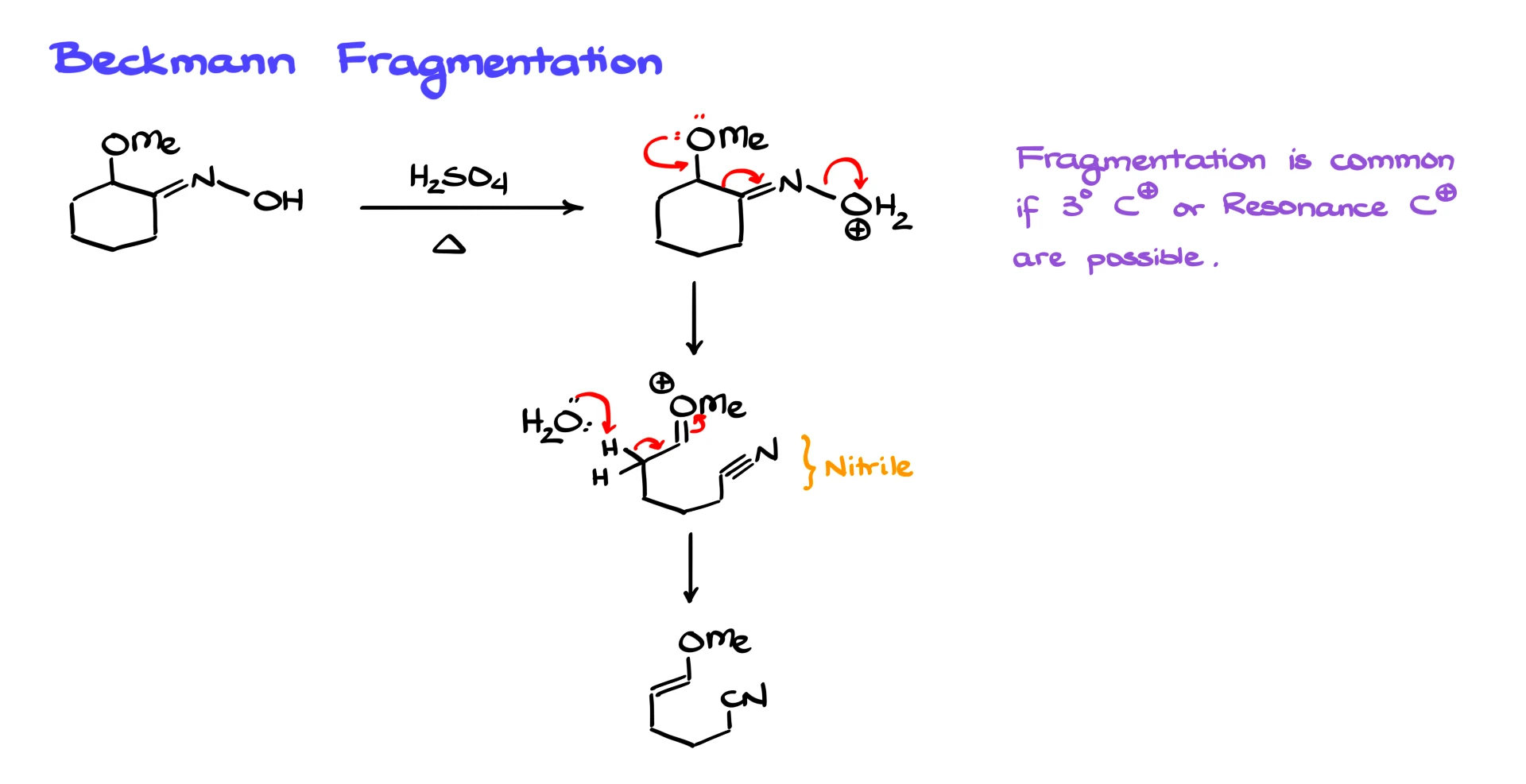
Consider an oxime where a methoxy group sits next to the C=N bond. If this compound is subjected to Beckmann conditions, either with sulfuric acid and heat or via an activated leaving group, the reaction can take a different path. Instead of a substituent migrating onto nitrogen, the molecule fragments. The bond breaks, forming a stable nitrile. The carbocation that forms alongside it is stabilized, and water does not attack the nitrile. Instead, water removes a proton from the carbocationic intermediate, leading to alkene formation.
Beckmann fragmentation is especially common when formation of a tertiary carbocation or a highly resonance-stabilized carbocation is possible. In those cases, fragmentation can easily outcompete rearrangement. This is why careful substrate analysis is crucial when planning a Beckmann rearrangement in synthesis.
