Benzilic Rearrangement
In this tutorial I want to talk about the benzilic acid rearrangement.
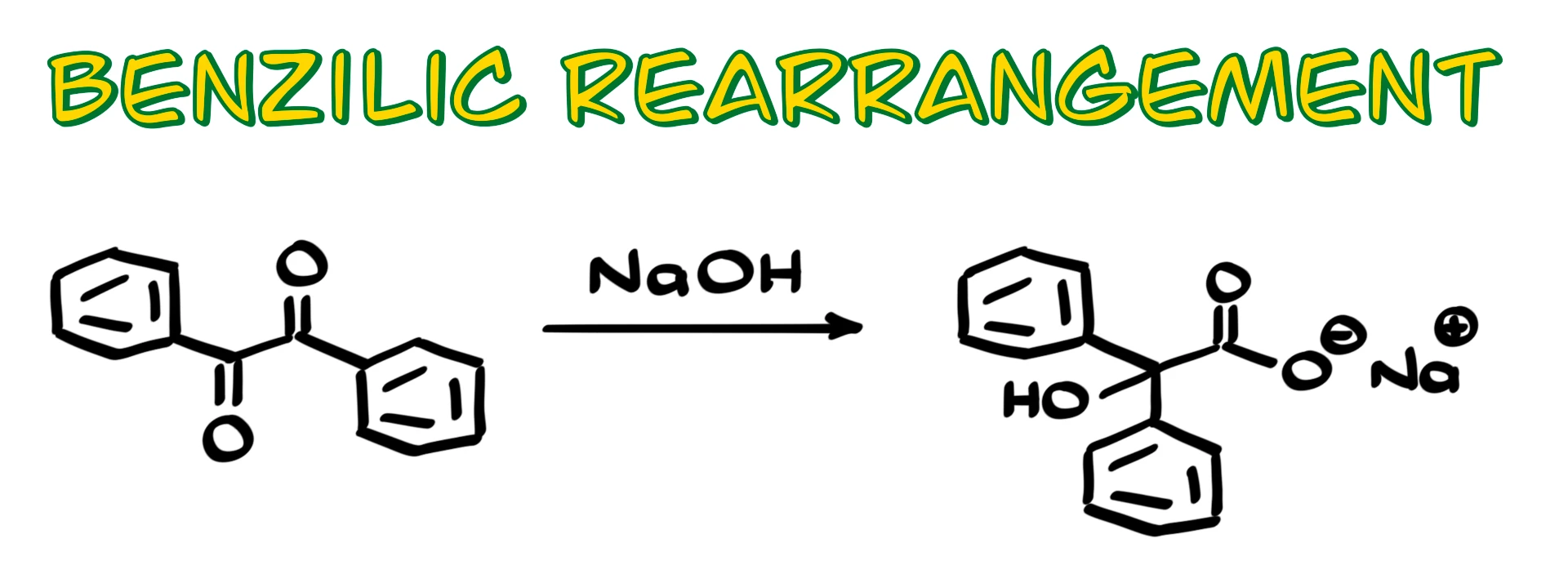
This reaction is a very specific rearrangement of α-dicarbonyl compounds, like the one shown here, which under basic conditions give carboxylates. I am saying carboxylates on purpose because in basic media the carboxylic acid product will be deprotonated.
Mechanism of the Benzilic Rearrangement
Let’s walk through the mechanism step by step. I’ve redrawn the starting material and I am showing hydroxide as the base. The metal counterion, whether it is sodium or something else, is not important for the mechanism.
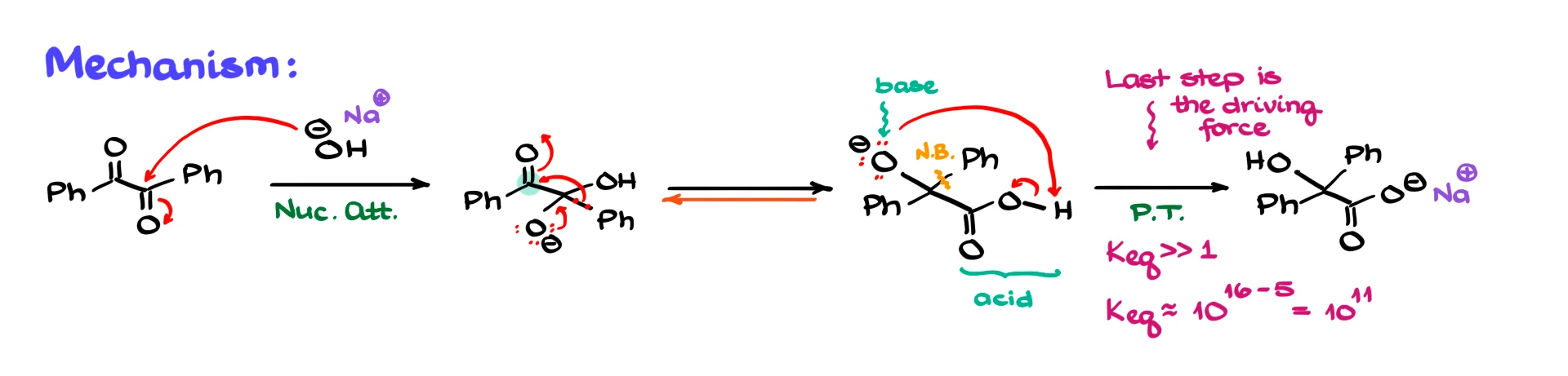
The first step is a nucleophilic attack by OH⁻ on one of the carbonyl carbons. In this particular example the molecule is fully symmetrical, so it does not matter which carbonyl we attack. As a result of this nucleophilic addition, we form a tetrahedral intermediate. This makes sense because carbonyl carbons are electron deficient and hydroxide is a strong nucleophile.
In the next step the key rearrangement happens. One of the phenyl groups migrates from its original carbon to the adjacent carbonyl carbon. Mechanistically, the electrons on the alkoxide oxygen collapse back down to reform the carbonyl, and at the same time the C–Ph bond breaks and the phenyl group migrates to the neighboring carbon. This creates a new carbon–carbon bond and gives us a carboxylic acid functionality on one side of the molecule and an alkoxide on the other.
At this point we have a strongly basic O⁻ and an acidic carboxylic acid in the same molecule. These two cannot coexist for very long, so a proton transfer happens immediately. The carboxylic acid loses its proton, which is transferred to the alkoxide, giving the carboxylate as the final product. You can draw this proton transfer intramolecularly, or you can show it with a chaperone mechanism where one species protonates the alkoxide and another base deprotonates the acid. Either representation is acceptable.
There are two important mechanistic details worth pointing out here. First, the phenyl migration step is technically an equilibrium step and should be drawn as reversible. Second, the final proton transfer is the true driving force of the reaction. A typical alcohol has a pKₐ of around 16, while a typical carboxylic acid has a pKₐ of about 5. That corresponds to an equilibrium constant on the order of 1011 or larger. Once that proton transfer happens, there is no going back.
Interesting Examples
Although this reaction might look a bit obscure at first, it has some very useful applications. One of the most important is ring contraction. If you start with a cyclic 1,2-dicarbonyl compound, you can use the benzilic acid rearrangement to shrink the ring by one carbon. For example, starting from a six-membered ring, the rearrangement gives a five-membered ring carboxylate.
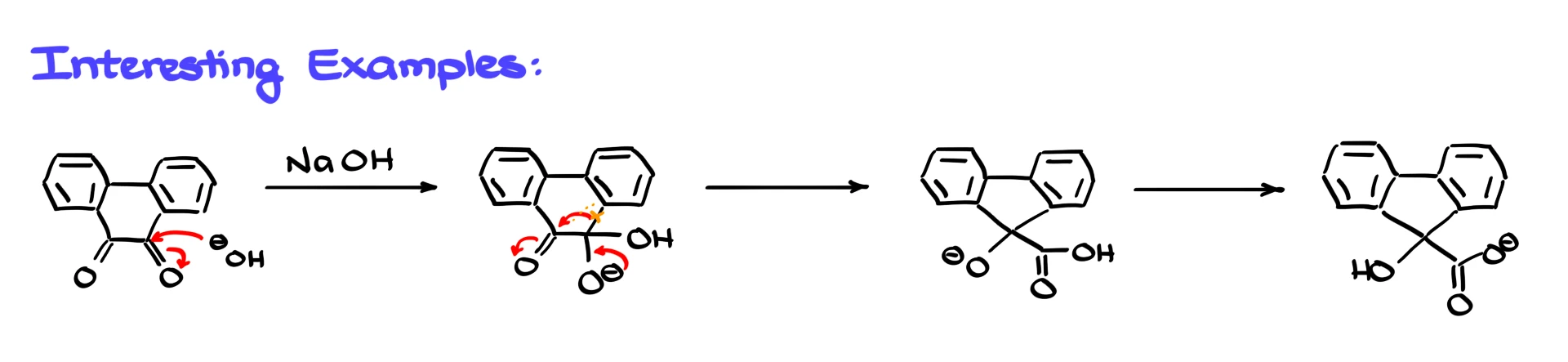
Mechanistically this follows the same logic. Hydroxide attacks one of the carbonyls, giving a tetrahedral intermediate. When the oxygen collapses back down, the aryl group migrates, a new carbon–carbon bond forms, and the ring contracts. A final proton transfer then gives the carboxylate product.
One of the really cool things about this reaction is how effective it is at shrinking rings of many different sizes. Six-membered rings go to five-membered rings very easily.

Five-membered rings can contract to four-membered rings, and even four-membered rings can contract to three-membered rings under the right conditions. There is extensive literature showing examples of all of these transformations.

Limitations of the Benzilic Rearrangement
There is, however, an important caveat. If the ring system is too strained, the rearrangement will not give a smaller ring. Instead, the ring will open. A four-membered ring is already quite strained, but if all of the atoms in that ring are sp² hybridized, the strain becomes extreme. In such a case, shrinking the ring to three members would be even more unfavorable.
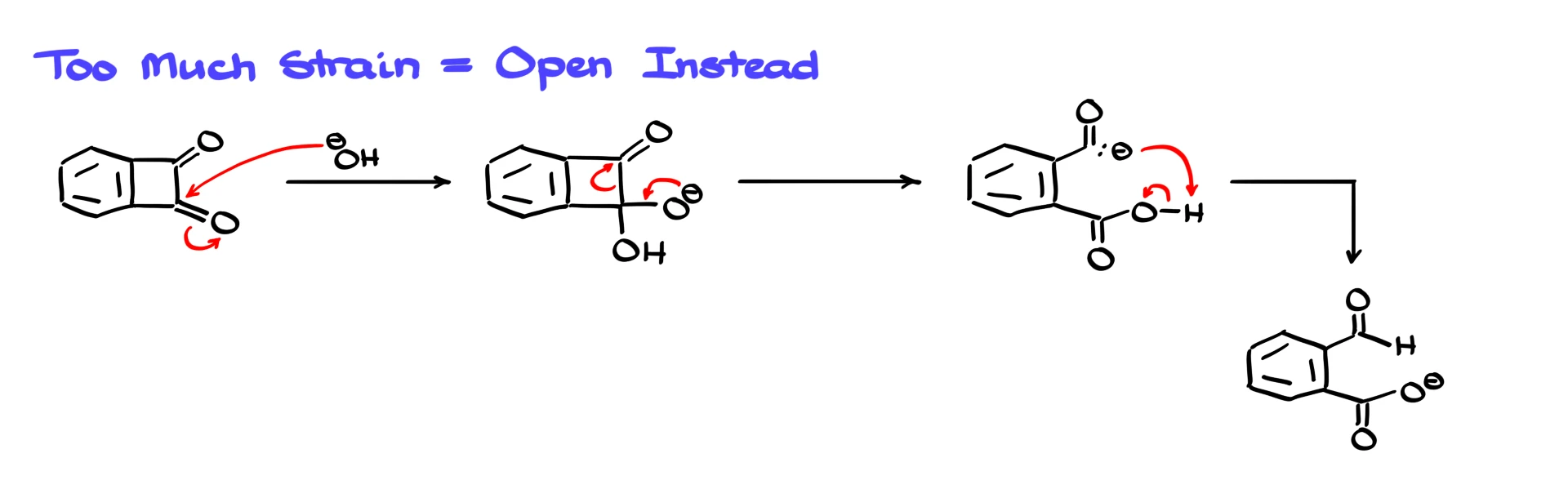
If we treat a highly strained system like that with base, hydroxide will still attack the carbonyl very efficiently, giving the familiar tetrahedral intermediate. However, when the electrons collapse back down, phenyl migration to form a three-membered ring is not feasible. Instead, a carbon–carbon bond breaks and the ring opens, producing a carbanion. This anion is not particularly stable, but it is still more stable than a hypothetical three-membered ring in such a strained system. A rapid proton transfer then occurs, giving a product that contains a carboxylate and an aldehyde.
Quantum chemical calculations support this pathway and show that formation of the three-membered ring intermediate is prohibitively high in energy. In these systems, ring opening is the preferred outcome.
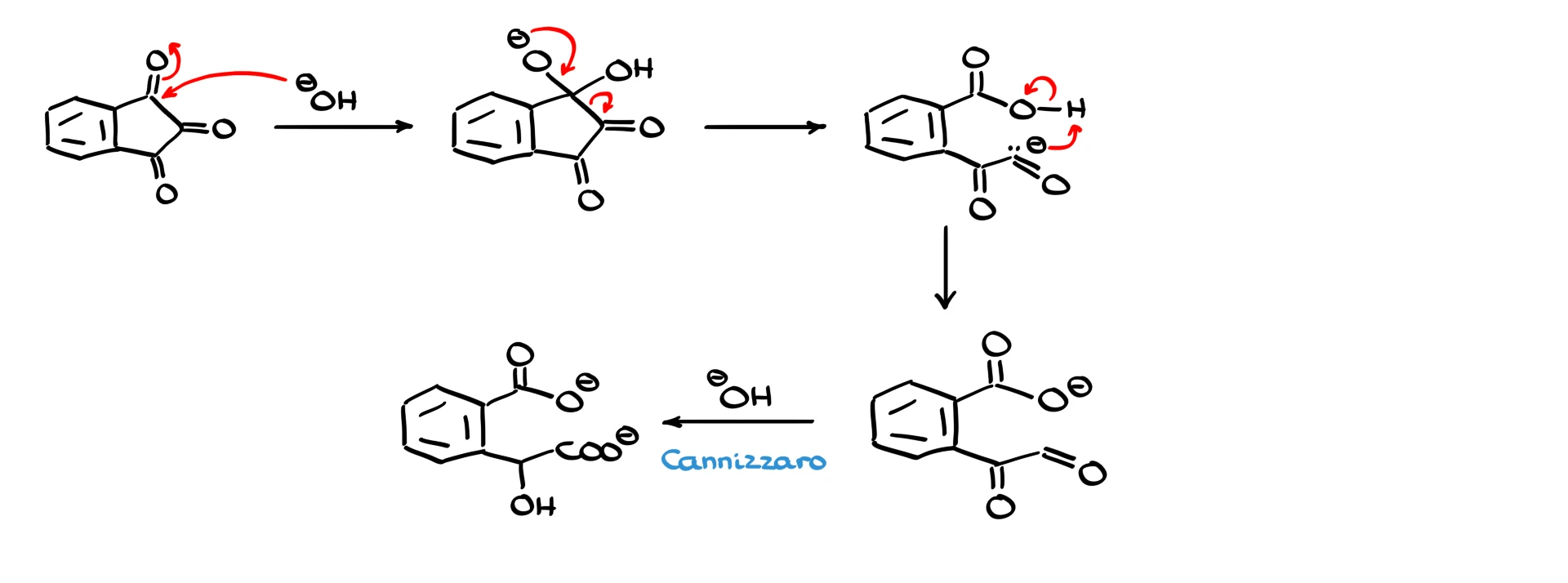
A similar situation arises with compounds like ninhydrin. If ninhydrin is treated with base, hydroxide attacks one of the carbonyls to form a tetrahedral intermediate. When this intermediate collapses, phenyl migration does not occur. Instead, the ring opens, giving a carbanion that is somewhat stabilized by the neighboring carbonyl groups. A proton transfer then gives an aldehyde-containing intermediate.
This is still not the final product. Under the strongly basic conditions of the reaction, this intermediate undergoes an intramolecular Cannizzaro reaction, ultimately producing a dicarboxylate. The Cannizzaro reaction itself is a topic for another discussion, and I will cover it in a separate tutorial.
So to summarize, the benzilic acid rearrangement involves nucleophilic attack on an α-dicarbonyl, followed by aryl migration and a proton transfer that drives the reaction forward. It is a powerful method for ring contraction, but when ring strain becomes too severe, the reaction pathway shifts toward ring opening instead.
