Sulfonate Esters of Alcohols
Alcohols are very versatile when it comes to their chemistry and transformations. However, the -OH group itself is a horrible leaving group.
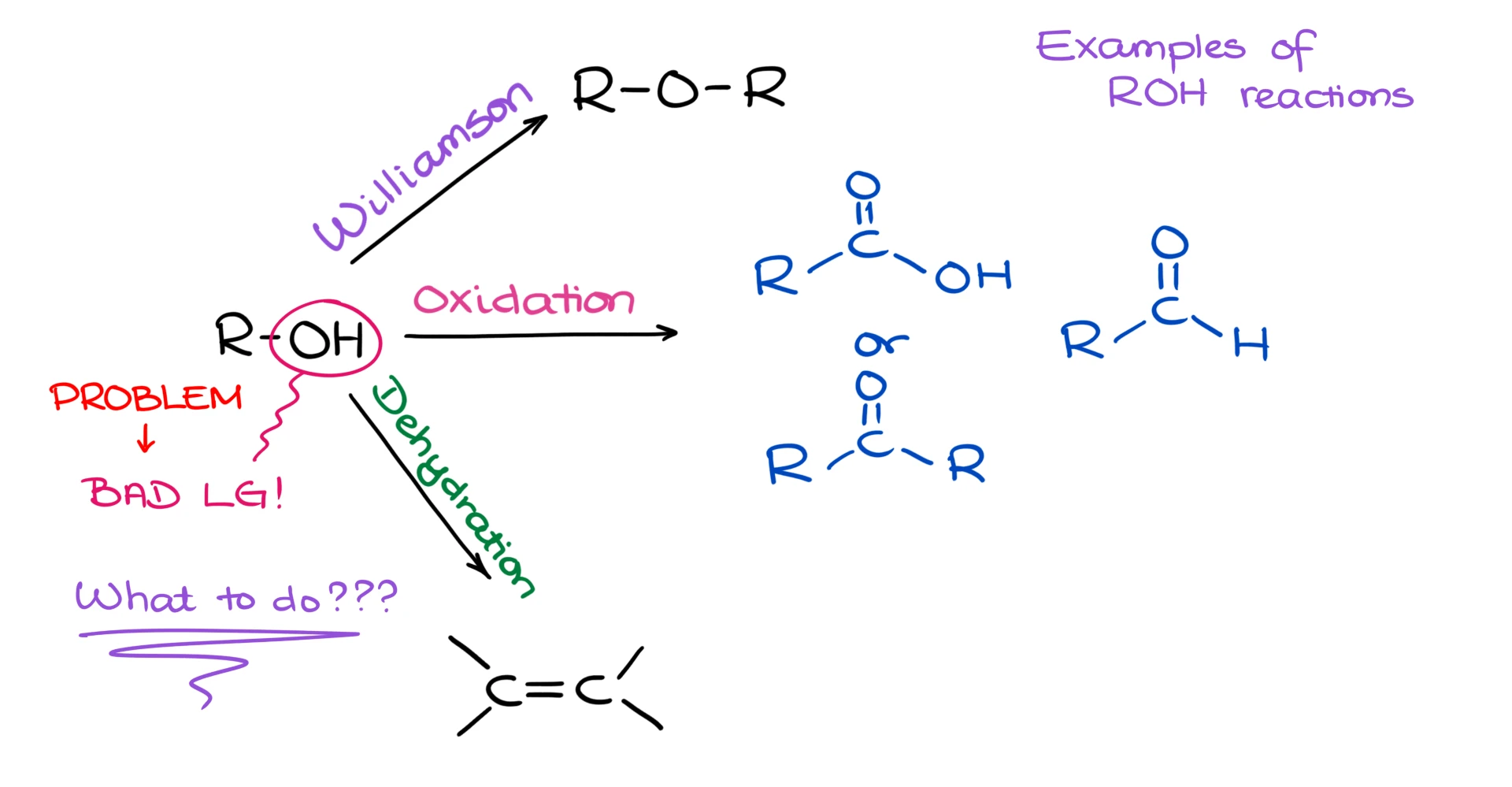
And if you need to substitute it with something or use it in an elimination reaction, you’re going to run into a problem. Is there anything we can do about it? Absolutely! The most common way of turning an -OH group into a good leaving group is by converting it into a sulfonate ester. This process is sometimes called an “alcohol activation” via sulfonate esters or sulfonyl chlorides. So, let’s look closer at what exactly it is and how it’s useful for us.
General Scheme of the Alcohol Activation via Sulfonate Esters
As I’ve mentioned earlier, the -OH of an alcohol is a pretty bad leaving group. So, if we wanna do something with that alcohol, we better change it into a good leaving group. And while we can easily replace the -OH with a halide, it’s not always the best way to go about it. Sometimes, we just want to convert the -OH into a good leaving group without completely replacing it.
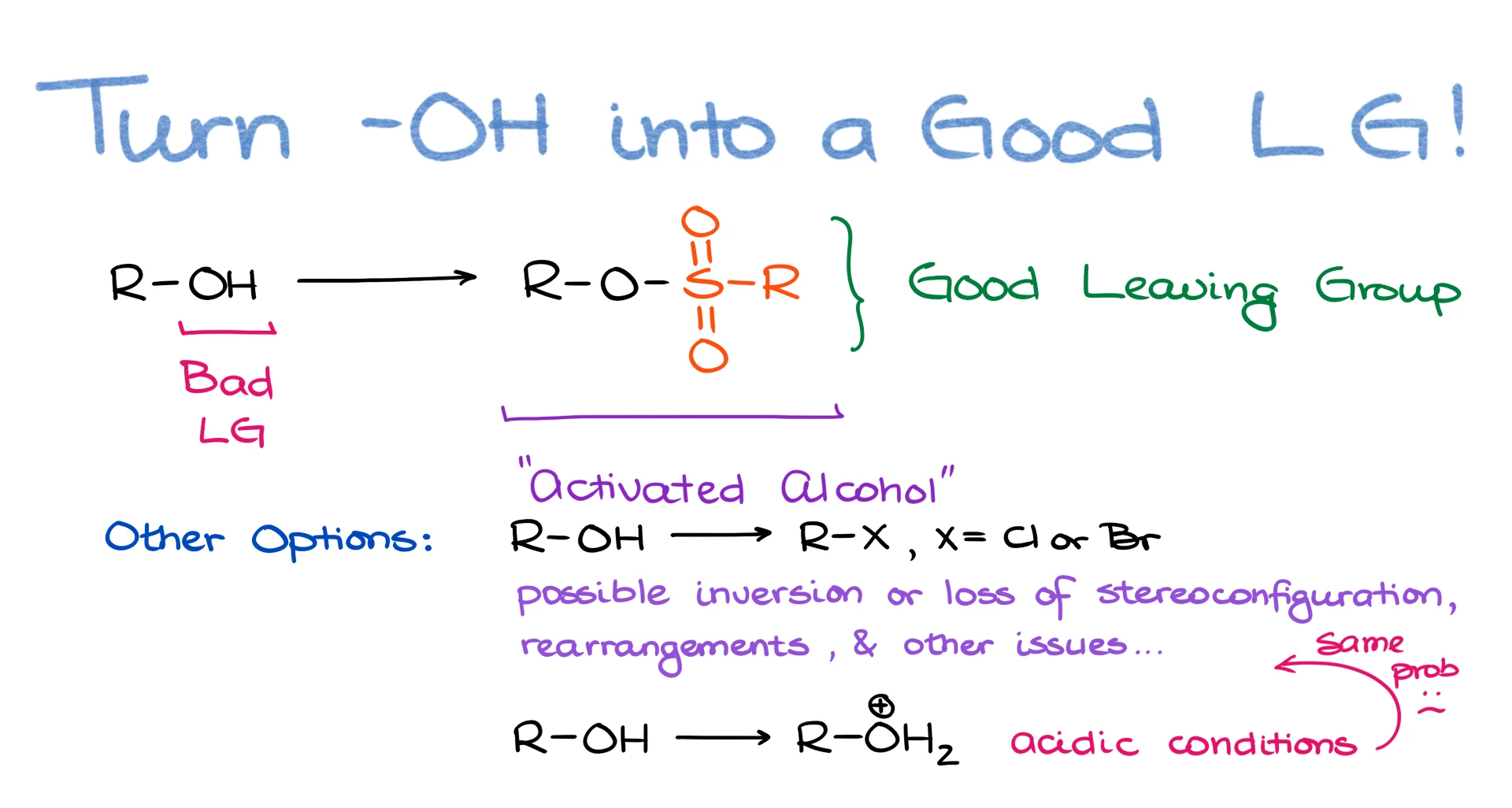
Probably, the first thing that comes to mind is protonation. We take an acid, protonate our alcohol, and it makes a good leaving group. Well, yes, it does. But what if the acidic conditions are not a viable option? Maybe your molecule is acid sensitive. Or what if the carbocation forms and you end up with a carbocation rearrangement? Or how about the stereochemical outcomes? Acidic conditions often result in SN1 and E1 reactions which would completely erase the relevant stereochemistry of your molecule. So, in many cases, doing reactions in acidic conditions for alcohols is going to be a no-go.
This is where the sulfonate esters come to play. We use a sulfonyl chloride in pyridine to turn an -OH group in an alcohol into a good leaving group.
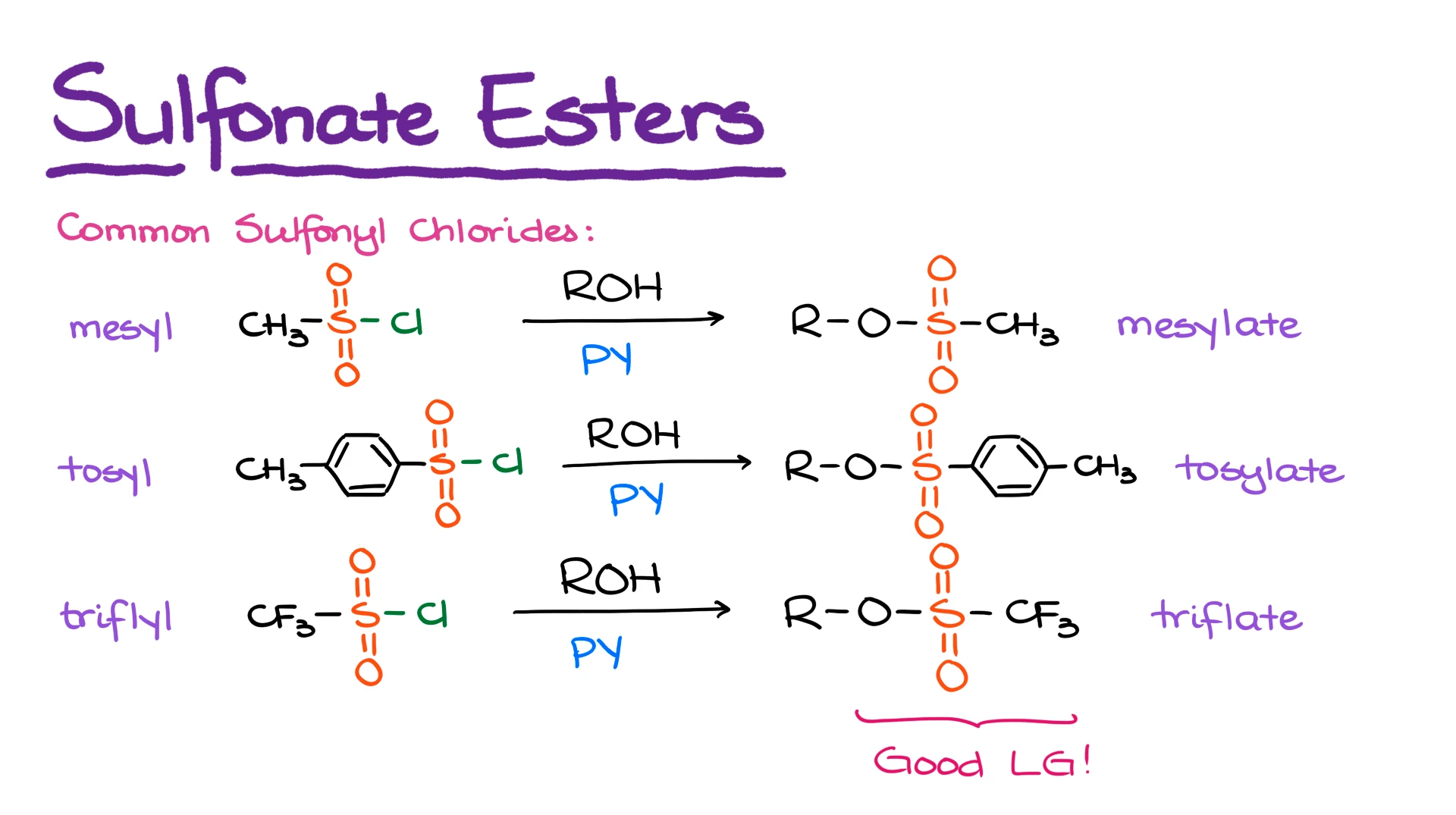
There are three most common sulfonyl chlorides you’re going to see in your class. Those are the mesyl, tosyl, and triflyl chlorides. And once they react with alcohols, they make corresponding mesylate, tosylate, and triflate sulfonyl esters. And all of those make awesome leaving groups. For the purposes of a typical sophomore organic chemistry class, they are interchangeable.
Mechanism of Alcohol Sulfonate Ester Formation
The reaction between an alcohol and a sulfonyl chloride is pretty straightforward.
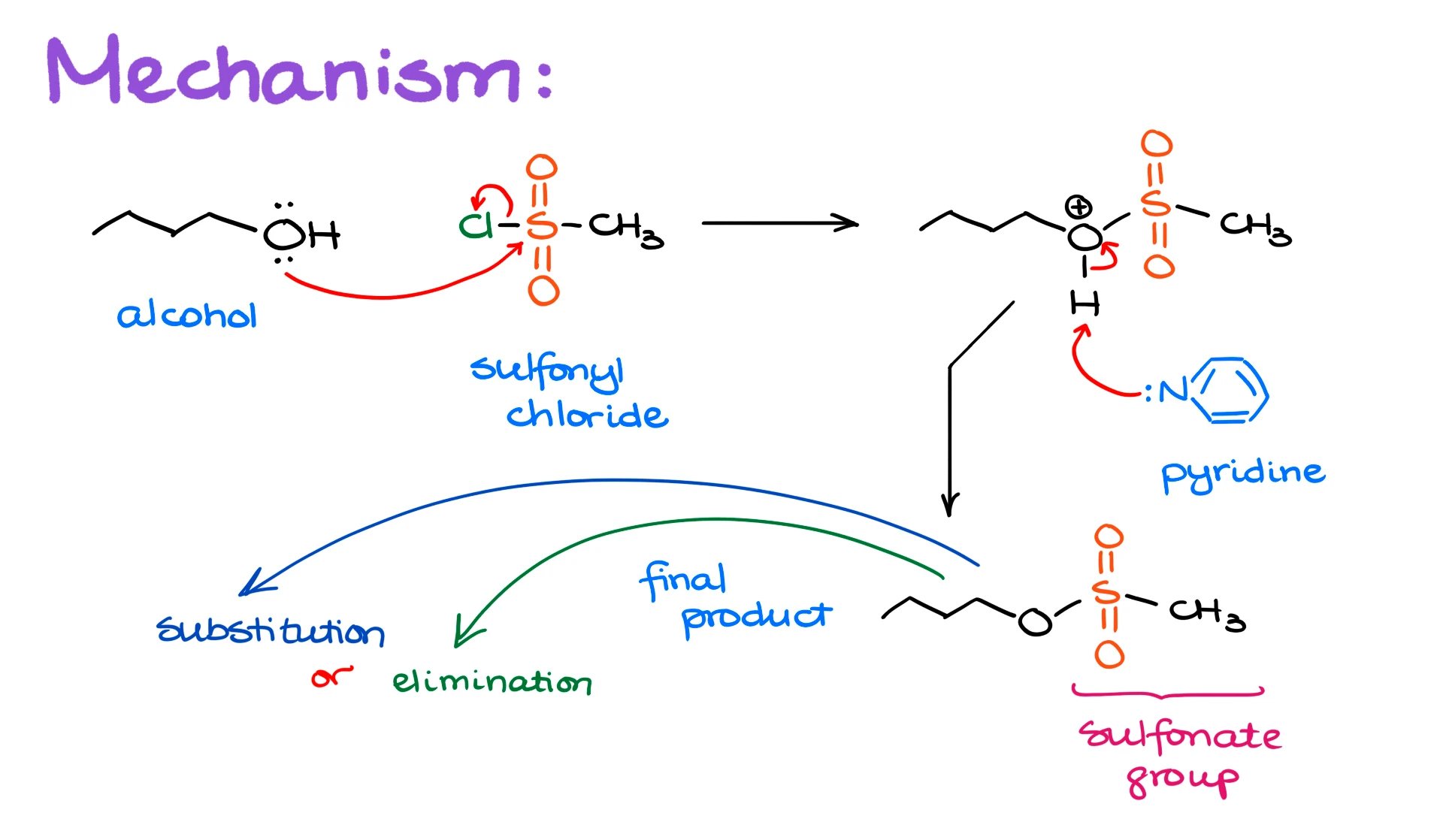
We take the alcohol, we take the sulfonyl chloride, and the alcohol attacks the sulfur atom displacing the chlorine. This forms a protonated intermediate, which quickly loses the proton to pyridine and gives us the final product where instead of the -OH group you have the corresponding sulfonate group.
Once you have your sulfonate ester, you can proceed with the elimination or a substitution reaction of your choice. Pretty simple.
Examples of the Sulfonate Ester Applications
There are a couple if important aspects of this reaction that you may wanna keep in mind when designing your synthesis. First, the sulfonate ester formation always proceeds with the retention of stereoconfiguration of your alcohol.
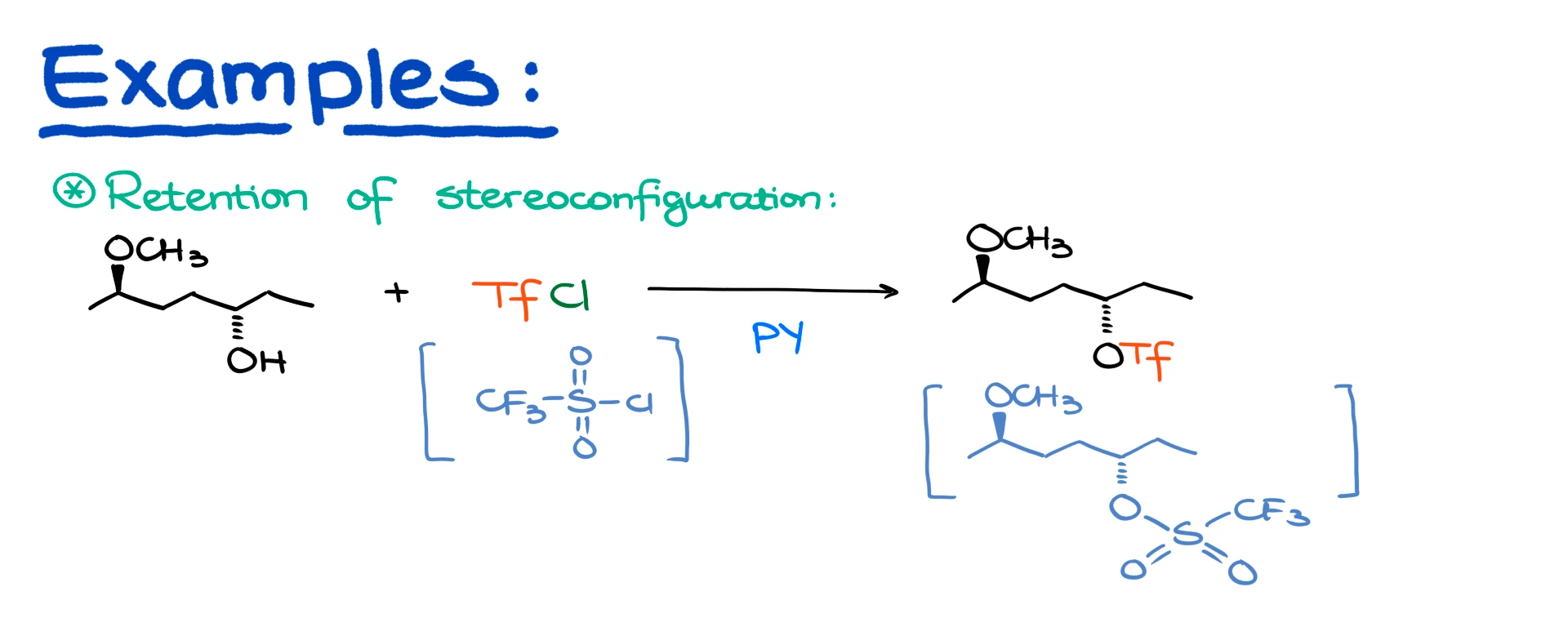
So, for instance, if I took this alcohol and reacted it with triflyl chloride, I’d get the product where the triflate group is looking in the same direction where the original alcohol used to look like. This way you can control the stereochemistry of your reaction. So, if now you choose to do the SN2 reaction, the nucleophile will replace the triflate and will give you the corresponding product where you would have predictable stereoconfiguration of the product. If you tired to do something of the sort with the original alcohol, you’d most likely need to do that in acidic conditions, and it would result in a horrible mess.

Another curious aspect of this reaction you might find useful, is the fact that you can selectively convert one alcohol into a leaving group while leaving the other alcohol(s) you might have in your molecule untouched. Let me elaborate. One of the sulfonates I’ve introduced at the beginning of this tutorial is quite bulky. I’m talking about the tosyl chloride, of course. Due to the size of this reagent, it is more selective towards the less sterically hindered alcohols. So, if your alcohol has a sterically hindered alcohol and a non-septically hindered alcohol, and you react it with only one equivalent of your tosyl chloride, the tosyl will predominantly attack the less sterically hindered alcohol. This is a nifty way how you can control which alcohol will be converted into a leaving group and selectively replace it with whatever you might need. Unfortunately, there’s no way to make the most substituted alcohol into a leaving group selectively while leaving the less substituted one intact. Not directly at least. You could, however, use protecting groups to accomplish that but that’s a topic for a whole other conversation.
