Conversion of Amides to Nitriles
Formation of nitriles from amides is one of those mechanisms that you either know well or it can come back to surprise you on an exam. So let’s go through it carefully.
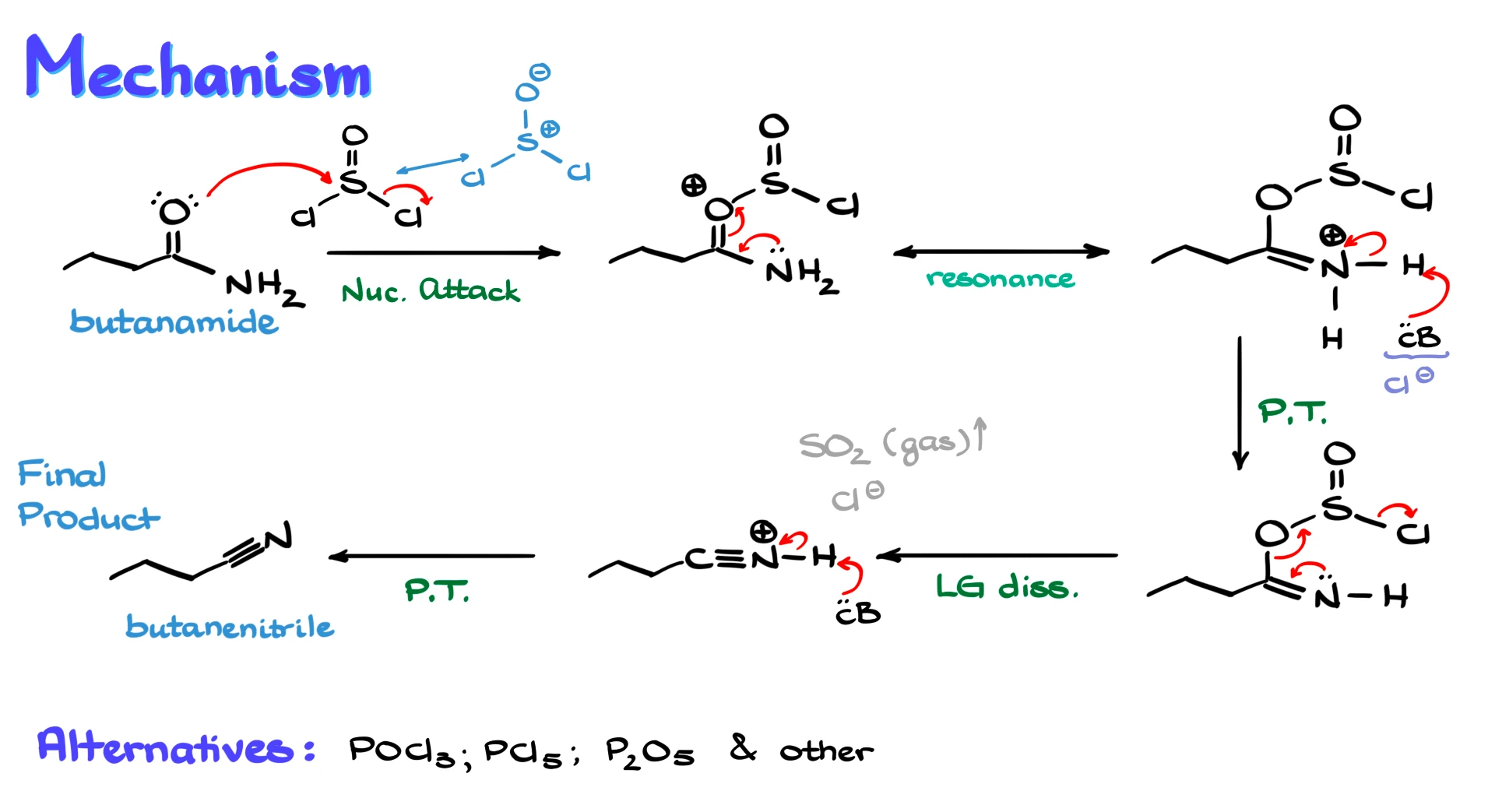
In this example, I will use butanamide and treat it with thionyl chloride, SOCl₂, as the dehydrating agent.
To begin the mechanism, it helps to redraw thionyl chloride in its structural form. We typically show a double bond between sulfur and oxygen, although an alternative resonance form with a single bond and formal charges on sulfur and oxygen is also common and, in many ways, more accurate.
The first step is a nucleophilic attack from the amide onto the sulfur atom of thionyl chloride. Specifically, the oxygen of the amide attacks sulfur, and one of the chlorides is expelled. This gives us a new intermediate where the amide is now attached to sulfur.
It might be tempting to use the lone pair on nitrogen for this step, but remember that for carboxylic acids and their derivatives, the oxygen is the nucleophilic site, not the nitrogen. This is largely because the resulting structure is stabilized by resonance.
We can show this stabilization by drawing a resonance structure where the lone pair on nitrogen forms a π bond with carbon, giving nitrogen a positive charge and converting the C–O bond into a single bond.
Next, we have a proton transfer step. A generic base, often Cl⁻ in this reaction, removes the proton from nitrogen. This gives us a neutral intermediate.
At this point, the group attached to the carbonyl oxygen has become an excellent leaving group. The next step is driven by the lone pair on nitrogen. That lone pair forms a triple bond with carbon, pushing out the leaving group.
As this happens, we form a protonated nitrile, and at the same time we release SO₂, typically as a gas, along with Cl⁻. The formation of gaseous SO₂ makes this step thermodynamically very favorable, which helps drive the reaction forward.
Finally, we need to remove the extra proton from the nitrile. This can be done by chloride or any other available base in solution. Once that proton is removed, we obtain the final product, the nitrile, in this case butanenitrile.
And that completes the mechanism.
One important note is that thionyl chloride is not the only reagent that can carry out this transformation. Other common dehydrating agents include POCl₃, PCl₅, and P₂O₅, also known as phosphoric anhydride. Mechanistically, they all do essentially the same thing, they convert the oxygen into a good leaving group, which then departs, allowing formation of the nitrile.
From a test perspective, however, if your instructor expects you to know this mechanism in detail, it will most likely be the thionyl chloride mechanism. While other reagents can perform the same transformation, their mechanisms are very similar and are less commonly tested explicitly.
And that is everything you need to know about converting amides into nitriles.
