HOMO-LUMO Interactions
In this tutorial I want to tackle one of the most confusing topics for many students, molecular orbitals, and specifically HOMO–LUMO interactions. Molecular orbitals can seem intimidating, but I promise that by the end of this tutorial you will be able to identify and properly label the molecular orbitals participating in a chemical reaction much more easily.
But first things first, we need to talk about the terminology and make sure we agree on what all of these terms mean.
So first of all, what exactly is a molecular orbital? When we use the term molecular orbital, we are referring to orbitals that belong to a molecule. But where do those orbitals come from?
From general chemistry, we already have a good idea about atomic orbitals. Whenever we have an atom, that atom contains a certain set of orbitals. So if you look at the periodic table, you probably would not have much trouble writing out the electron configuration for a random atom.
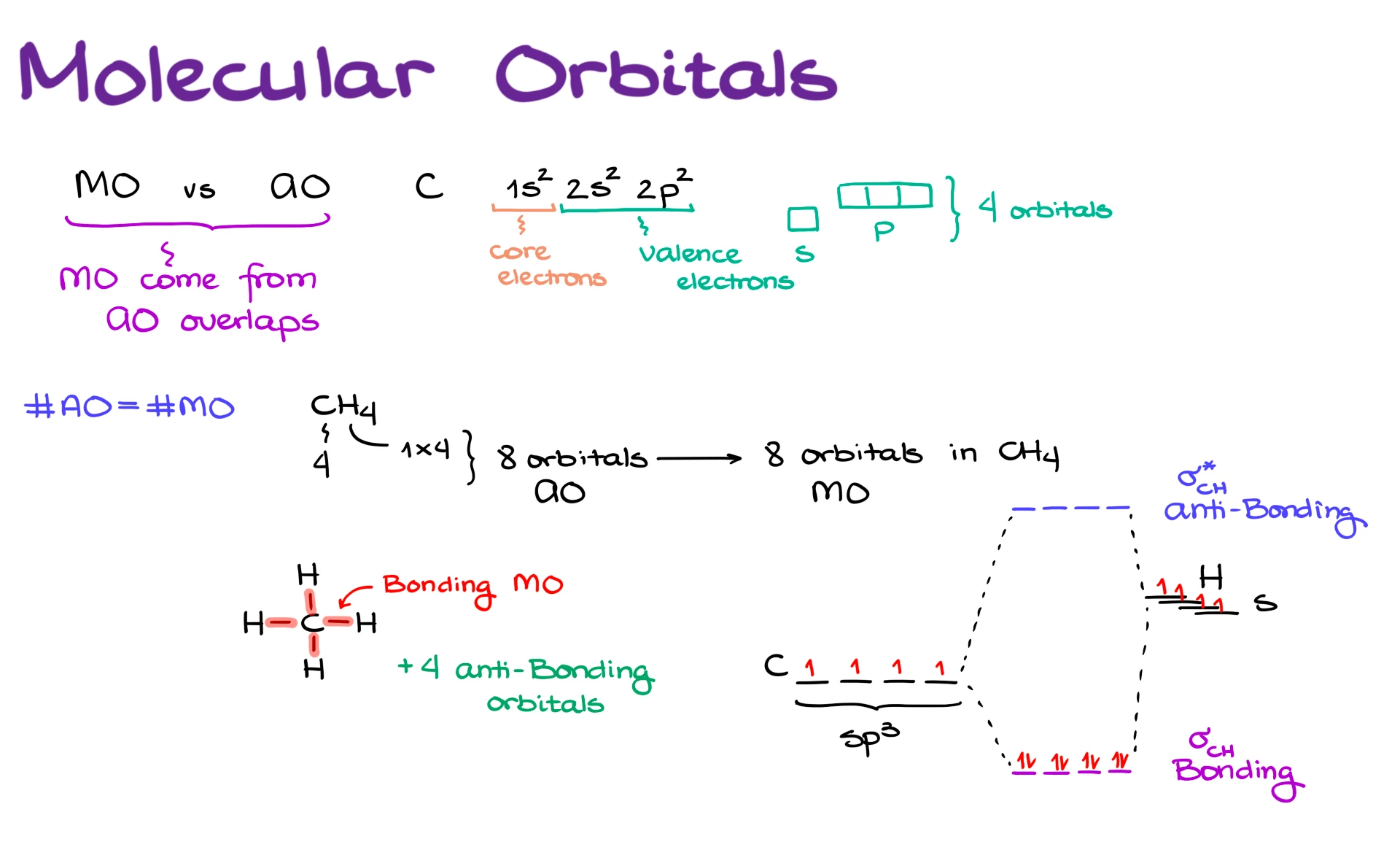
For example, let’s take carbon, which is at the core of organic chemistry. Carbon has the following electron configuration. We have the 1s orbital, which is filled with two electrons. Then we have the 2s orbital, which is also filled with two electrons. And then we have the 2p orbitals, where we place the remaining two electrons.
The 1s electrons are what we normally refer to as the core electrons. In general, we make the same broad assumption here that we borrow from valence bond theory, core electrons do not participate in bonding.
The next shell, where we have the 2s and 2p electrons, contains the valence electrons, and those are the electrons we use to make bonds.
We also know from general chemistry that the s orbital is just a single orbital. However, the p orbitals come as a set of three, px, py, and pz. So overall, when it comes to the valence level of carbon, we have four orbitals total.
Now, when we take an atom with those orbitals, such as carbon, and overlap those orbitals with orbitals from other atoms, we form a molecule. And that molecule is going to have molecular orbitals.
So in other words, molecular orbitals come from atomic orbital overlap.
Another important thing to keep in mind is that the number of atomic orbitals is always equal to the number of molecular orbitals we end up with. So if I look at something like methane, CH₄, carbon brings in four orbitals, and each hydrogen brings in one orbital. Since there are four hydrogens, that gives us a total of eight atomic orbitals.
Because the number of molecular orbitals must equal the number of atomic orbitals, once all of those atomic orbitals combine, methane will also have eight molecular orbitals.
If we draw the Lewis structure for methane, we can see that it contains four bonds. Bonds correspond to bonding molecular orbitals. Without exception, whenever you see a bond in an organic molecule, or really in any molecule, that bond corresponds to some kind of bonding molecular orbital.
But that only accounts for four orbitals. What about the other four, since we know we need a total of eight?
Those other four are what we call antibonding orbitals. Normally we do not show antibonding orbitals explicitly in standard structural drawings, and normally those orbitals are empty, meaning they do not contain electrons.
If I were to draw a very simplified molecular orbital energy diagram, I would show carbon with four degenerate sp³ hybrid orbitals, each containing one electron. Then I would show four hydrogen 1s orbitals, all at the same energy level, each containing one electron as well.
When all of these orbitals combine, we form four bonding molecular orbitals, which are lower in energy than the original atomic orbitals, and four antibonding molecular orbitals, which are higher in energy. I am simplifying a little here by treating them as degenerate, but that is perfectly fine for our purposes.
Overall, methane has eight electrons, so when I fill the molecular orbitals, I place those eight electrons into the four lower-energy bonding orbitals. Since all of the bonds in methane are single bonds, these are σ bonding orbitals between carbon and hydrogen. The corresponding antibonding orbitals are written as σ* between carbon and hydrogen.
So the first big takeaway is that the number of atomic orbitals is always equal to the number of molecular orbitals.
Another thing to remember is that bonding orbitals are always lower in energy than the atomic orbitals that formed them, while antibonding orbitals are always higher in energy than the atomic orbitals that formed them.
When it comes to electrons, we want electrons in bonding orbitals, because that creates bonds. We do not want electrons in antibonding orbitals, because populating an antibonding orbital weakens or even breaks a bond.
And finally, the number of bonding orbitals is always equal to the number of antibonding orbitals. Every bonding orbital has an antibonding counterpart, its evil twin at higher energy.
Pi BOnds
Now those were σ bonds, but what about π bonds?
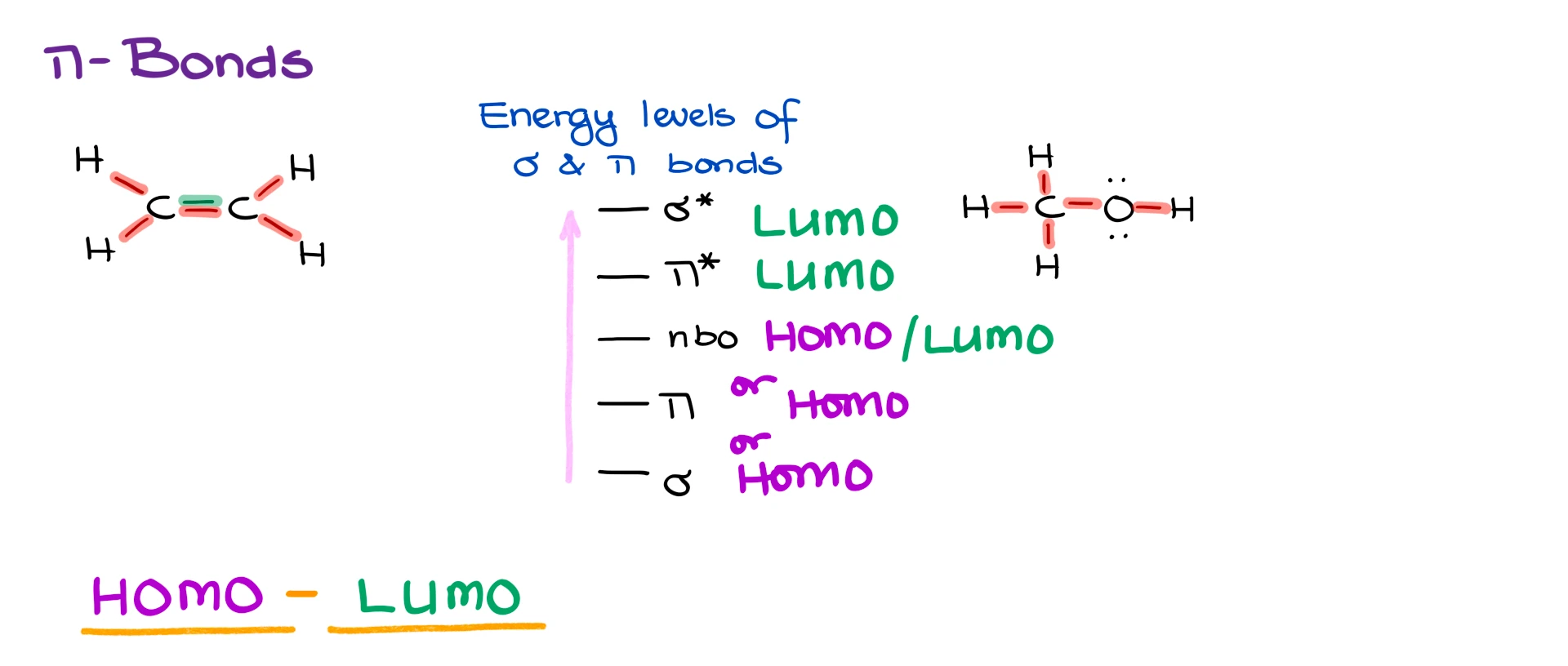
The idea is very similar. Let’s say we have ethylene, a simple alkene with a C=C double bond. In this case, we have σ bonds between carbon and hydrogen, and we also have a σ bond between the two carbons. On top of that, we have a π bond between the carbons as well.
So whenever we have a double bond, we have one σ bond and one π bond.
When it comes to energy, σ bonding orbitals are typically the lowest-energy occupied orbitals in the molecule. Then above those we have the π bonding orbitals. So π electrons are usually higher in energy than σ electrons.
Above the π bonding orbitals we have the π antibonding orbitals, π, and finally at the very top we have the σ antibonding orbitals, σ.
So in a simple picture, all of the σ electrons sit at relatively low energy, the π electrons sit somewhat higher, then above them come the unoccupied π* orbitals, and above those the σ* orbitals.
That is, of course, a simplified picture, but it is more than enough for what we need here. We do not need every exact orbital energy. What matters is the general order. σ bonding orbitals are lower than π bonding orbitals, and π* orbitals are usually lower than σ* orbitals.
Before we start looking at reactions, there is one other type of orbital we need to talk about, nonbonding orbitals.
In general chemistry, we often talk about lone pairs as nonbonding electrons. So if I look at something like water, the O–H bonds correspond to σ bonding orbitals, but oxygen also has two lone pairs. Those lone pairs are what we refer to as nonbonding orbitals.
Typically, nonbonding orbitals sit somewhere in the middle of the energy scale. They are usually higher in energy than π bonding orbitals, but lower in energy than π* orbitals.
So for our purposes, the general trend in molecular orbital energy looks like this. At the bottom we have σ bonding orbitals, then π bonding orbitals, then nonbonding orbitals, then π antibonding orbitals, and finally σ antibonding orbitals.
This general picture works for most of the molecules you are going to encounter in a standard organic chemistry course. Of course, some molecules will not have π orbitals, some will not have nonbonding electrons, and so on, but the overall pattern still holds.
Now we need two more important terms.
The first is the highest occupied molecular orbital, or HOMO. This is literally the highest-energy molecular orbital that contains electrons. Very often the HOMO is a nonbonding orbital, because lone pairs tend to sit fairly high in energy. In other cases, the HOMO can be a π bond. And if the molecule has neither π bonds nor lone pairs, then the HOMO may simply be a σ bond.
The second term is the lowest unoccupied molecular orbital, or LUMO. This is the lowest-energy molecular orbital that does not contain electrons. Very often the LUMO is a π* orbital or a σ* orbital, depending on the structure. In some special cases, an empty nonbonding orbital can serve as the LUMO, as in carbocations or boron-containing compounds.
Now here is the really important and really cool idea. Every interaction in chemistry is fundamentally an interaction between the HOMO of one species and the LUMO of another species.
The closer those orbitals are in energy, the better the interaction tends to be. The farther apart they are, the worse the interaction tends to be. That is one reason why some reactions happen very easily while others refuse to go, no matter how much you beg them to.
Once those orbitals interact, the electrons redistribute into newly formed molecular orbitals. That redistribution is what leads to bond formation and bond breaking.
This is a somewhat different way of thinking from the usual textbook approach. In intro chemistry, we often think of electrons simply moving around and making or breaking bonds. But from the molecular orbital perspective, it is really the orbitals that interact first, and the electrons then populate the new orbitals that result from that interaction.
So whenever we draw a mechanism, what we are really representing is a HOMO–LUMO interaction.
Examples
Let’s go through a few examples.
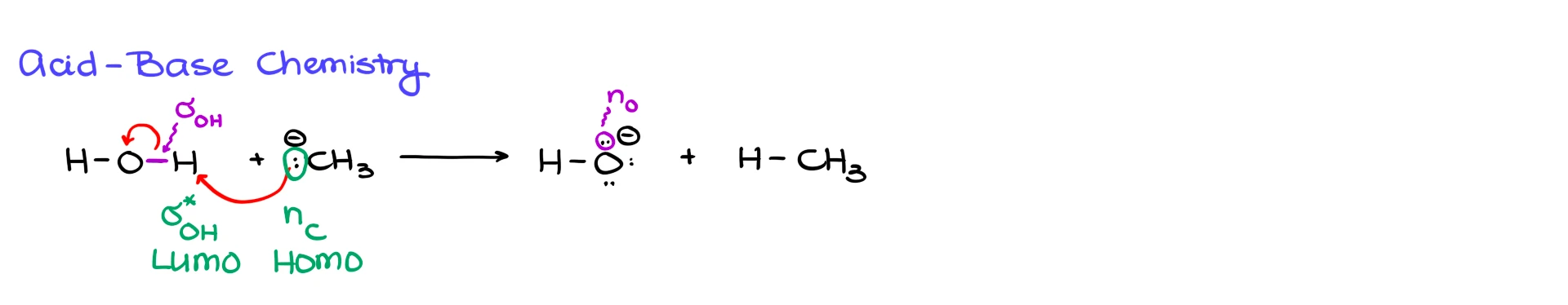
First, let’s look at a simple acid–base reaction. Suppose we have a carbanion reacting with water. In the mechanism, the electron pair on carbon attacks the hydrogen of water, and the O–H bond breaks, giving us methane and hydroxide.
From the orbital perspective, the electrons on the carbanion are a nonbonding orbital on carbon. Since that is where the electrons originate, that orbital is the HOMO.
Those electrons are being donated into the orbital associated with breaking the O–H bond. Since we are breaking a single bond, the acceptor orbital is the σ* orbital of the O–H bond. That is the LUMO.
The second curved arrow, the one showing the O–H bond electrons moving onto oxygen, is not a separate HOMO–LUMO interaction between two species. It is just the transformation of a σ bond into a nonbonding orbital on oxygen as the bond breaks.
Now let’s look at a substitution reaction. Suppose we take a thiolate anion and react it with bromomethane.
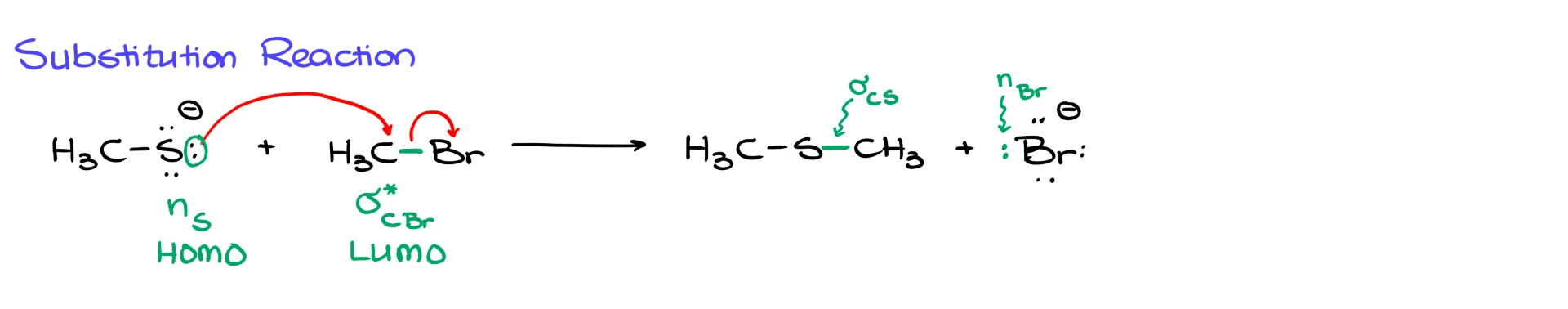
In the mechanism, the lone pair on sulfur attacks the methyl carbon, and the C–Br bond breaks to give bromide and the thioether product.
In this case, the electrons start on sulfur as a nonbonding orbital on sulfur, so that is the HOMO.
Those electrons are going into the orbital that allows the C–Br bond to break. Since we are breaking a single bond, that acceptor orbital is the σ* orbital of the C–Br bond. That is the LUMO.
As a result of that interaction, the former nonbonding orbital on sulfur becomes a new σ bond between sulfur and carbon, while the former C–Br σ bond becomes a nonbonding orbital on bromine.
Now let’s move to something a little more involved, electrophilic addition to an alkene.
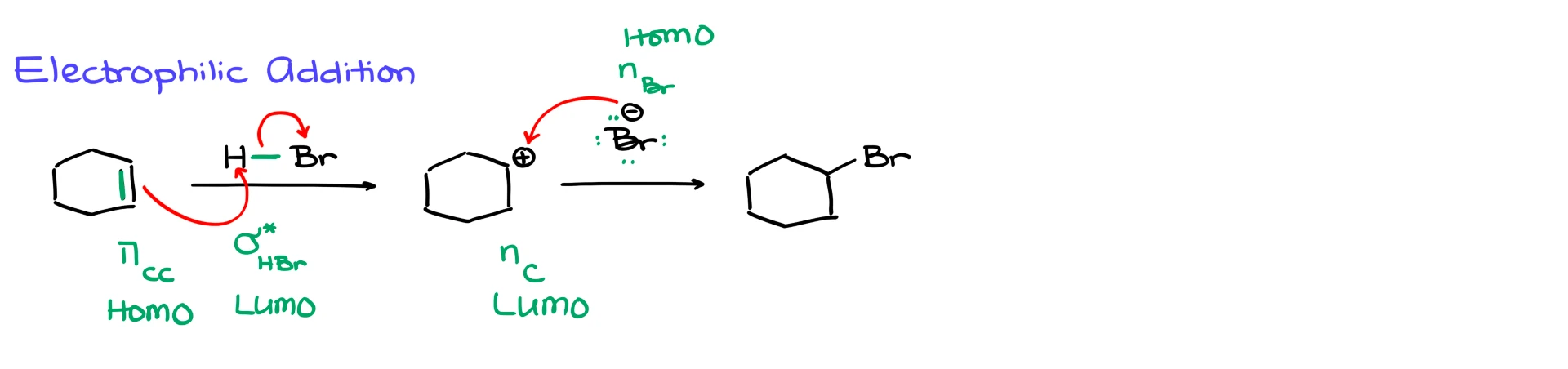
Suppose we take cyclohexene and react it with HBr. In the first step, the electrons of the π bond attack the hydrogen, and the H–Br bond breaks, giving us a carbocation and bromide.
From the orbital perspective, the electrons start in the π bonding orbital of the alkene, so that is the HOMO.
Those electrons are donated into the orbital that allows the H–Br bond to break. Since that is a single bond, the acceptor orbital is the σ* orbital of H–Br. That is the LUMO.
Then in the second step, bromide attacks the carbocation to give bromocyclohexane.
In this case, the electrons start from a lone pair on bromide, so the HOMO is a nonbonding orbital on bromine.
Those electrons are going into the empty orbital on the carbocation. Since that is simply an empty orbital on carbon, the LUMO is an empty nonbonding orbital on carbon.
So notice that the LUMO is not always a σ* or π* orbital. Sometimes it is just an empty orbital.
The practical version of all of this is actually pretty simple. As long as you know the mechanism, you can usually identify the interacting orbitals directly from the electron flow.
Wherever the electrons start, that is your HOMO. Wherever the electrons go, that is your LUMO.
If the electrons start from a lone pair, the HOMO is a nonbonding orbital. If they start from a bond, the HOMO is a σ or π bonding orbital. If the electrons are going into an orbital that breaks a single bond, the LUMO is a σ* orbital. If they are going into an orbital that breaks a π bond, the LUMO is a π* orbital. And if the electrons are going into an empty orbital, then the LUMO is simply that empty orbital, often treated as a nonbonding orbital.
So yes, this is only scratching the surface of molecular orbital theory and how it applies to organic chemistry. But this is a very good starting point. It gives you a practical way to identify the orbitals involved in the reactions you are already learning and to connect mechanisms to orbital interactions.
Later on, once you get a little deeper into the course, you can use molecular orbitals to explain more subtle patterns of reactivity in organic chemistry. But that is a topic for a more advanced conversation.
