Nomenclature of Aromatic Compounds
In this tutorial I want to talk about everything you need to know about the nomenclature of aromatic compounds.
We’re going to cover the must-know common names, the ortho, meta, and para nomenclature and its limitations, and of course the IUPAC, or systematic, nomenclature of aromatic compounds as well. So let’s jump right into the common names.
Common Names
When it comes to common names, there are must-know names and good-to-know names.
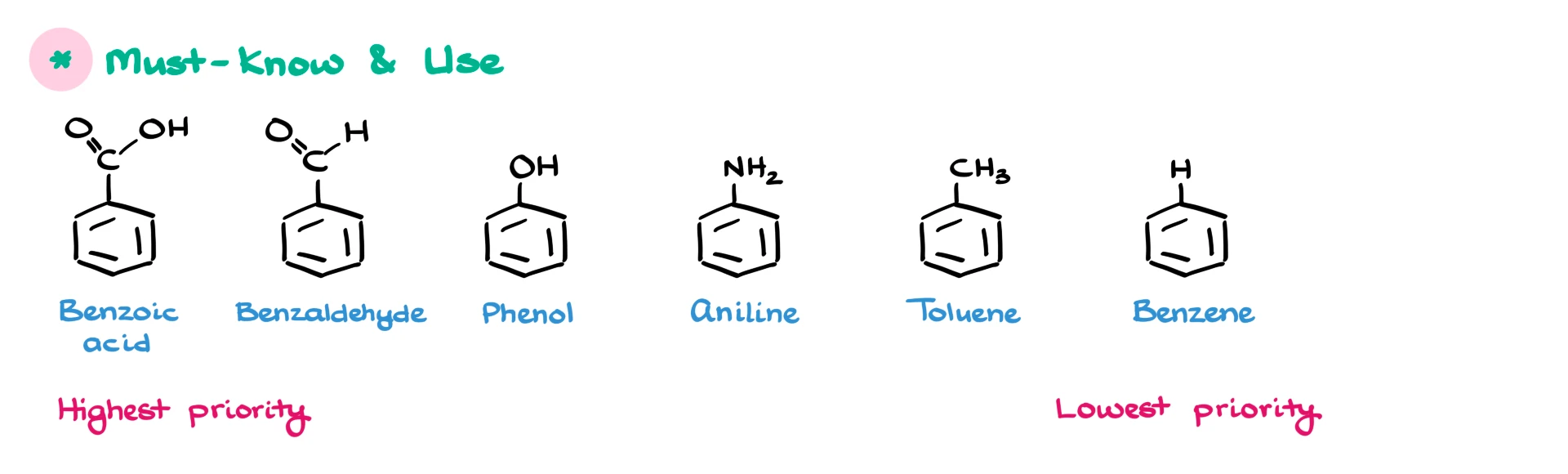
The must-know names are benzoic acid, benzaldehyde, phenol, aniline, toluene, and of course benzene. When I say must-know, I also mean must-use. These are retained names that IUPAC requires us to use for the nomenclature of aromatic compounds.
Although benzaldehyde has been somewhat demoted in recent updates, and the preferred IUPAC name would technically be benzenecarbaldehyde, the name benzaldehyde is still used in almost every textbook and is most likely what you are learning in class, so I’m going to stick with benzaldehyde.
The order in which I listed these compounds is intentional. The molecules on the left have higher priority than the ones on the right. That becomes very important when you have multiple substituents on your aromatic ring.
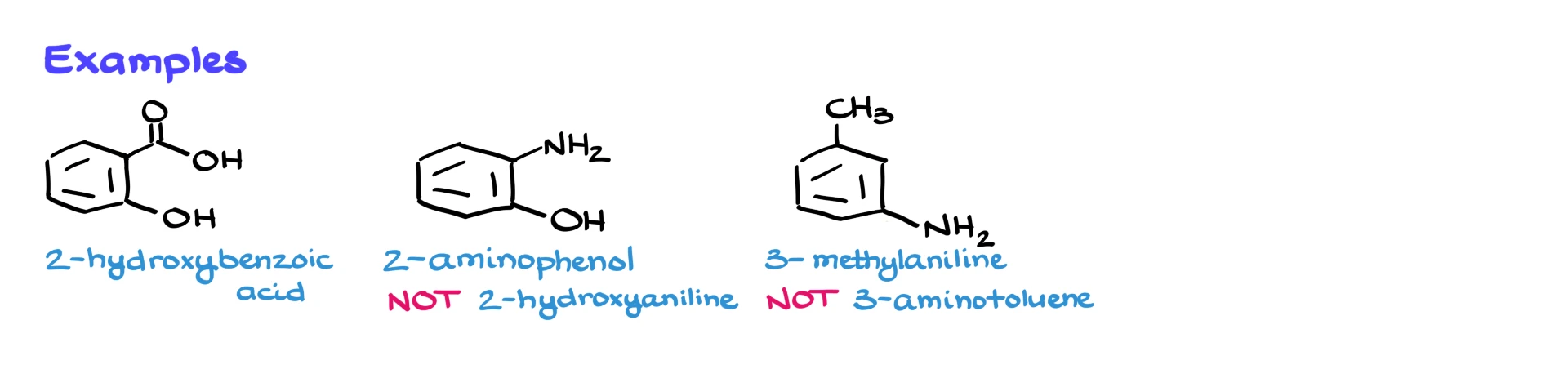
For example, consider salicylic acid. We have a carboxylic acid group and an –OH group. Since the carboxylic acid has higher priority, we must name this molecule as a derivative of benzoic acid rather than phenol. So the systematic name is 2-hydroxybenzoic acid, and the common name is salicylic acid.
Here’s another example. Suppose we have an –NH₂ group and an –OH group on the aromatic ring. Since –OH has higher priority, this compound is a derivative of phenol rather than aniline. The proper name would be 2-aminophenol, not hydroxyaniline.
Or consider a molecule with an amino group and a methyl group. The amino group has higher priority than the methyl group, so this compound would be named 3-methylaniline, not aminotoluene.
Make sure you memorize this list and its order. This is very commonly tested, and you can almost guarantee it will show up on your exam.
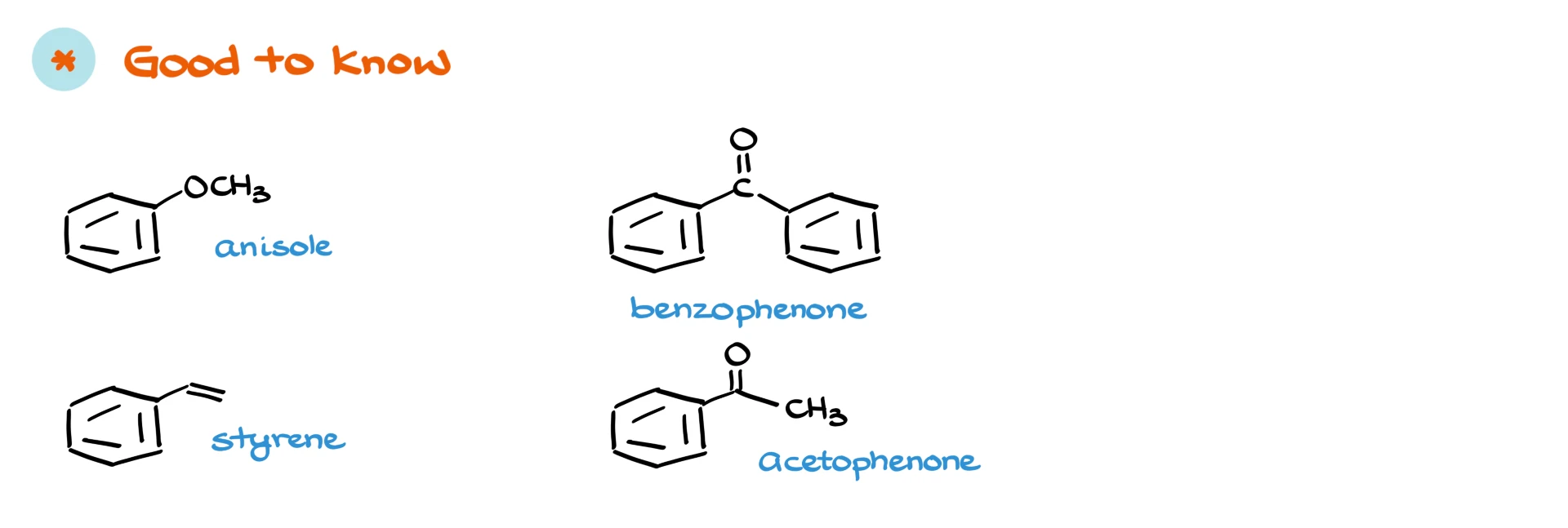
Now let’s move on to some good-to-know common names. A few that show up all the time are anisole, styrene, benzophenone, and acetophenone. There are others you might encounter depending on your course, but these are among the most common. These are not required names in the same way as the must-know list, but you should recognize them when you see them in a textbook or hear your instructor mention them.
Ortho, Meta, and Para Nomenclature
Next, let’s talk about ortho, meta, and para nomenclature, often abbreviated as o-, m-, and p-.

Unlike IUPAC numbering, ortho, meta, and para describe relative positions to a reference group on the aromatic ring. Let’s say we have a generic aromatic compound with an R group. That R group serves as our reference group. Typically, the reference group is the substituent with the highest priority.
The carbon bearing the reference group is carbon number 1. The positions adjacent to it, carbons 2 and 6, are the ortho positions. Carbons 3 and 5 are the meta positions. The carbon directly across the ring, carbon 4, is the para position.
If you’re curious, the position of the reference group itself is sometimes called the ipso position, but that term does not appear in naming, so we won’t spend time on it.
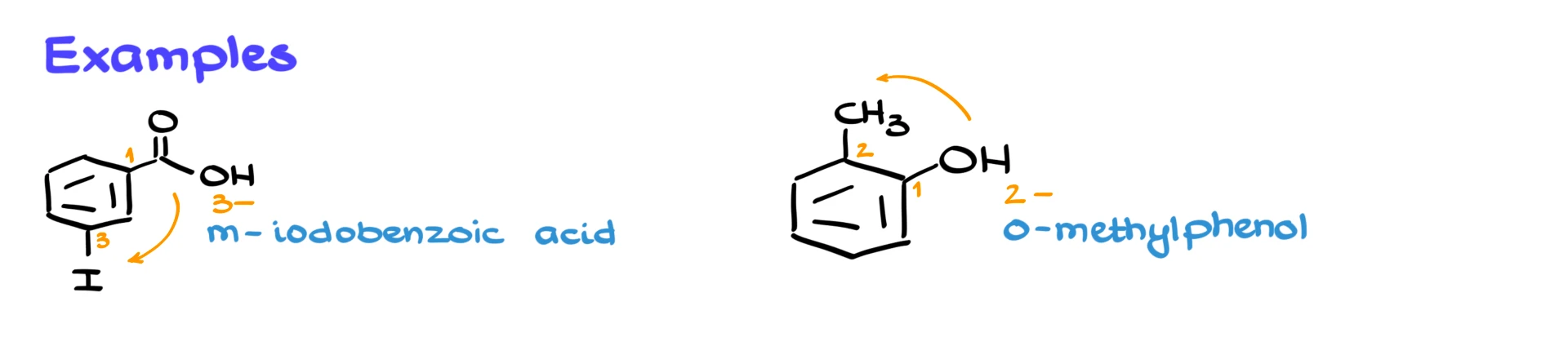
Let’s look at an example. Suppose we have a benzene ring with a carboxylic acid and an iodine substituent. The carboxylic acid has the highest priority, so it is carbon number 1. It doesn’t matter how the molecule is oriented in space. The highest priority group is always carbon 1.
To assign the lowest possible numbers, we number the ring clockwise, placing iodine at carbon 3. Since carbon 3 is a meta position, the compound is called meta-iodobenzoic acid. Alternatively, using numbers, it would be 3-iodobenzoic acid.
Here’s another example. Suppose we have an –OH group and a methyl group. The –OH has higher priority, so it is carbon number 1. To give the lowest numbers, we number counterclockwise, placing the methyl group at carbon 2. Carbon 2 is an ortho position, so the compound is called ortho-methylphenol, or 2-methylphenol if we use numbers.
Now here’s the biggest limitation of ortho, meta, and para nomenclature: you can only use it when there are exactly two substituents on the ring. If there are three or more substituents, you must use the full IUPAC numbering system.
IUPAC Nomenclature of Aromatic Compounds
When it comes to systematic naming, the rules are the same ones you’ve seen before.

We already know the parent is the aromatic ring. First, identify the highest priority functional group. That carbon becomes number 1. Then number the rest of the ring to give the substituents the lowest possible set of numbers. After assigning the numbers, priorities no longer matter. The only other consideration is alphabetical order in case of a tie.
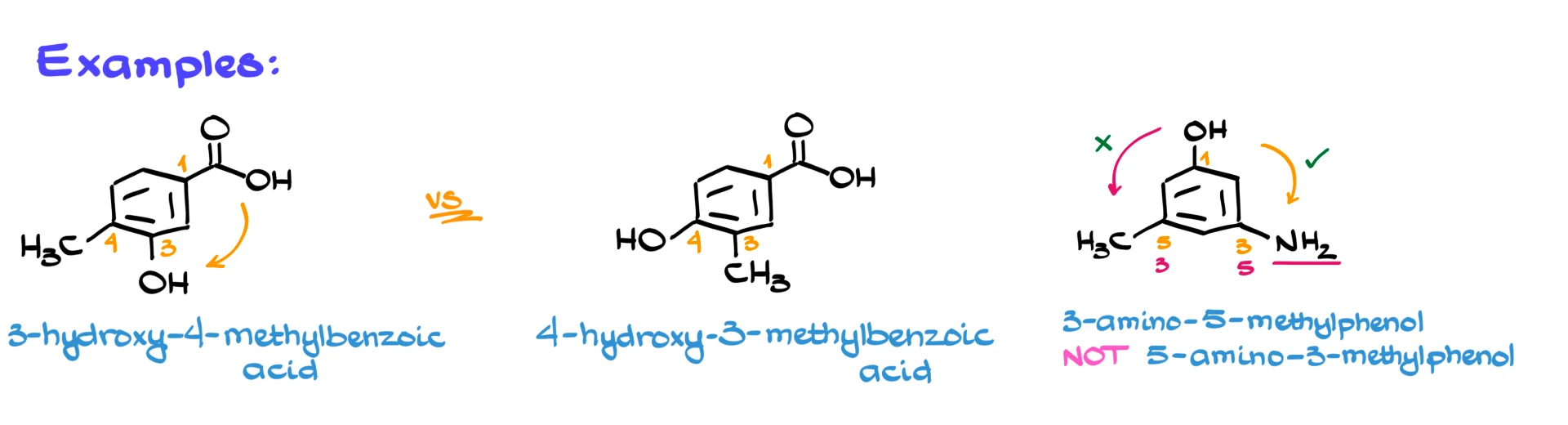
For example, suppose we have two compounds, each with a carboxylic acid group. In both cases, that group is carbon number 1. In the first compound, numbering clockwise gives –OH at carbon 3 and –CH₃ at carbon 4. In the second compound, –CH₃ is at carbon 3 and –OH is at carbon 4.
So the first compound is 3-hydroxy-4-methylbenzoic acid, and the second is 4-hydroxy-3-methylbenzoic acid. When writing the name, we alphabetize substituents. Hydroxy comes before methyl because H comes before M, even if hydroxy has the higher number. Alphabetical order is based on the names, not the numbers.
Here’s another example with –OH, –NH₂, and –CH₃. The –OH has the highest priority, so it is carbon number 1. If we number clockwise, we get substituents at carbons 3 and 5. If we number counterclockwise, we also get 3 and 5. That’s a tie.
In that case, we use alphabetical order as a tiebreaker. Amino comes before methyl, so we choose the numbering that gives the lower number to amino. The final name is 3-amino-5-methylphenol.
Let’s do a few more examples.

If we have a phenol with a propyl group and a chlorine, the –OH is carbon 1. Numbering counterclockwise gives chlorine at carbon 4 and propyl at carbon 3. The final name is 4-chloro-3-propylphenol. Even though chloro is at carbon 4 and propyl is at carbon 3, we still list chloro first because C comes before P alphabetically.

In another example, suppose we have an aldehyde, an –OH, and an ethyl group. The aldehyde has the highest priority, so it is carbon number 1. We number counterclockwise to give ethyl carbon 3 and hydroxy carbon 5. The final name is 3-ethyl-5-hydroxybenzaldehyde.

In the final example, suppose we have an aniline derivative with a methyl and an isopropyl group. The amine has the highest priority, so it is carbon 1. Numbering counterclockwise gives methyl carbon 2 and isopropyl carbon 5, which is the lowest possible set of numbers. After alphabetizing, remembering that “iso” counts in alphabetical order, the name becomes 5-isopropyl-2-methylaniline.
Aromatic Rings as Substituents
Before we wrap up, one more important point: aromatic rings can also act as substituents.
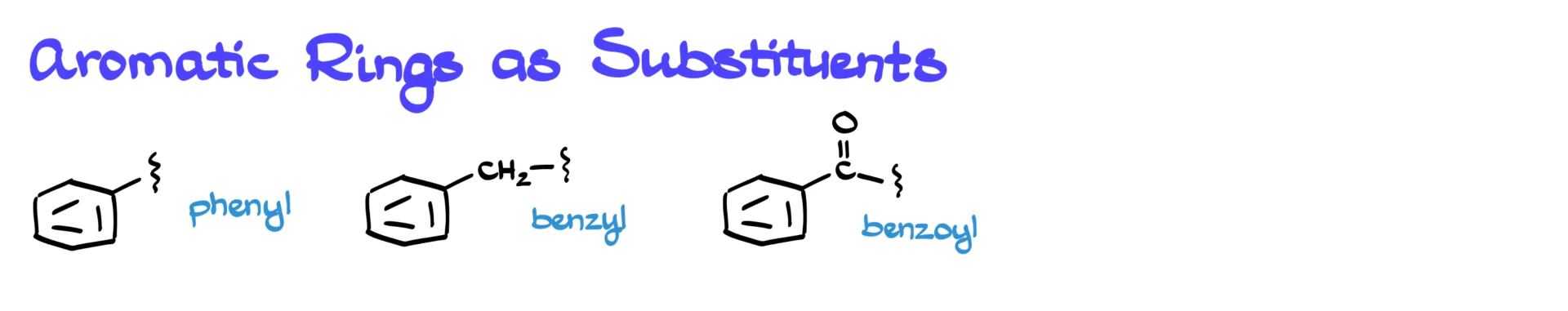
If the aromatic ring itself is a substituent, it is called a phenyl group. Notice the pronunciation difference. PhEnyl is the substituent, phenOl is the aromatic compound with an –OH group.
If there is a –CH₂– group connecting the ring to the rest of the molecule, that substituent is called benzyl. If there is a carbonyl group connecting the ring, it is called benzoyl.
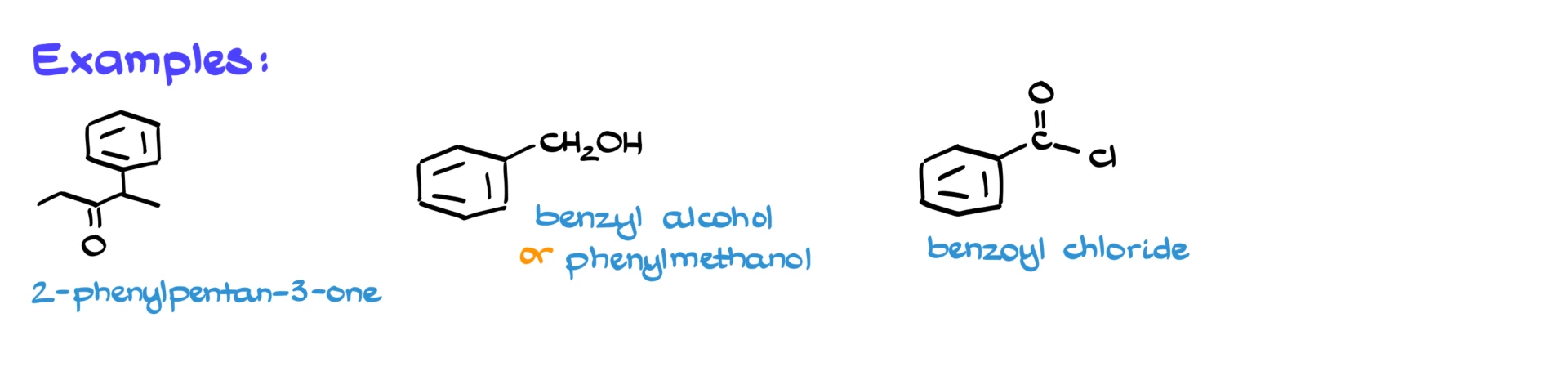
For example, consider a five-carbon chain with a ketone at carbon 3 and a phenyl substituent at carbon 2. The parent chain is pentane-3-one, since the ketone has higher priority than the aromatic ring. The full name is 2-phenylpentane-3-one.
Another example is benzyl alcohol. We could call it phenylmethanol using strict IUPAC naming, but benzyl alcohol is the commonly used name.
And finally, benzoyl chloride is the acid chloride derived from benzoic acid. That’s a very common compound, so you definitely want to recognize that name.
