Polar Protic vs Polar Aprotic Solvents
In this tutorial I want to talk about organic solvents.
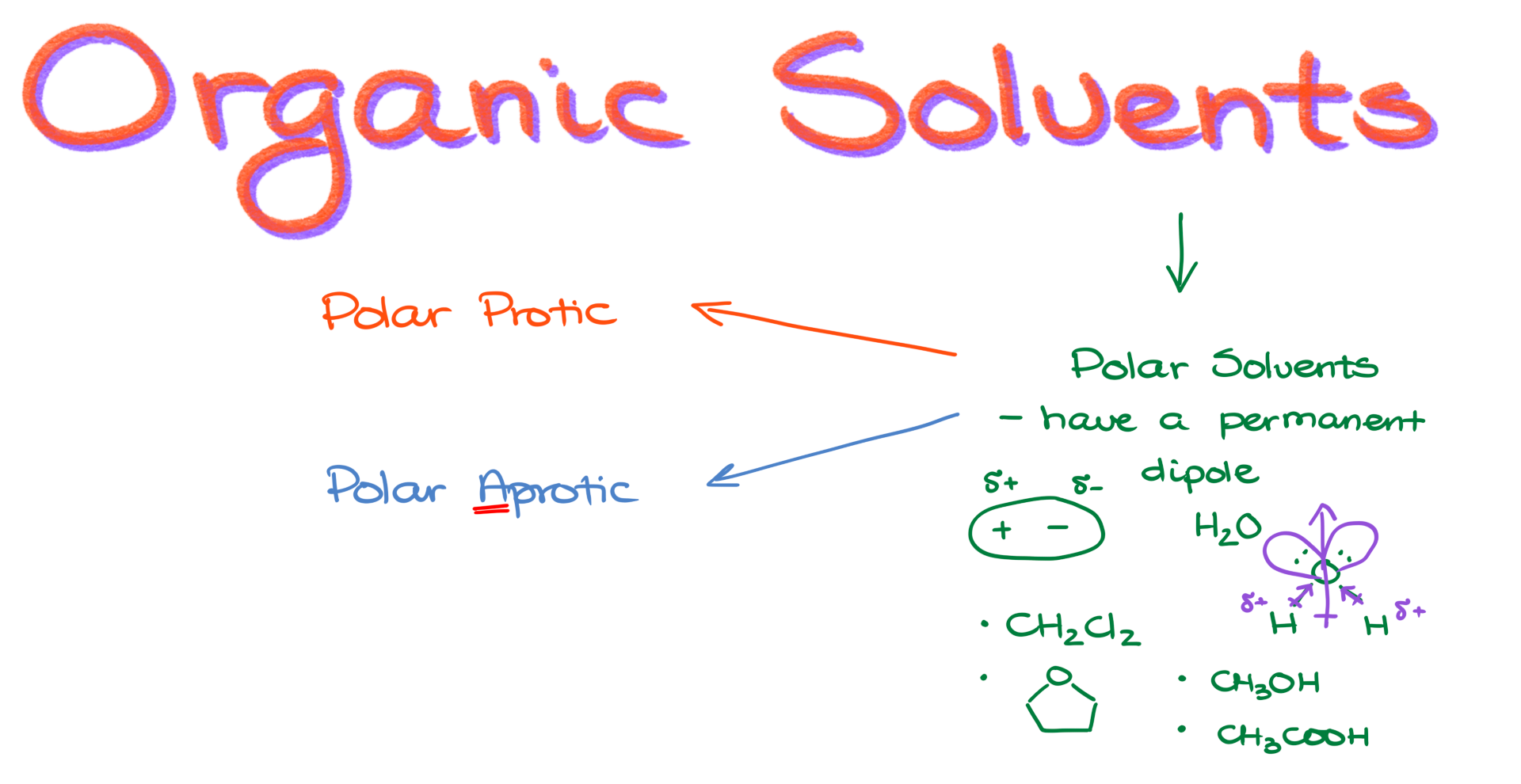
When it comes to organic solvents, we can generally divide them into two broad categories. Those are nonpolar solvents and polar solvents. Nonpolar solvents lack a permanent dipole. That means they are overall fairly uniform in their electron density, so they do not have a distinctly electron-rich part and an electron-poor part within the same molecule. In that sense, they are neutral.
Typical examples of nonpolar solvents include compounds like hexane and other alkanes. You might also see CCl₄, which is a very nonpolar compound. However, because of the environmental impact of this solvent, we generally try not to use it nowadays. Another somewhat common nonpolar solvent is benzene, which is an excellent solvent, but because of both its toxicity and environmental concerns, we also tend not to use it very often either.
Another important category of solvents is polar solvents. Unlike non-polar solvents, polar solvents do have a permanent dipole. That means one part of the molecule is going to be permanently more electron-rich than another part, so the molecule behaves like a little dipole. One end has a partial positive charge, and the other end has a partial negative charge. Sometimes we represent those with δ⁺ and δ⁻ signs.
A classic example of a polar solvent is water, H₂O. If we look at water and think about its structure, we have an oxygen atom connected to two hydrogen atoms. Oxygen also has two lone pairs. From the electron density perspective, oxygen pulls electron density toward itself because it is more electronegative than hydrogen. That makes the hydrogen atoms partially positive, or δ⁺, and the oxygen end partially negative, or δ⁻. As a result, the whole molecule has an overall dipole.
Other examples of polar solvents include dichloromethane, or DCM, tetrahydrofuran, or THF, methanol, CH₃OH, and even acetic acid. But here is the important thing, not all polar solvents are the same. We can further divide polar solvents into two categories of their own, polar protic solvents and polar aprotic solvents. It can be a little confusing when you hear one versus the other, so I will try to emphasize the “a” in aprotic whenever I talk about them.
So let’s talk a little more about what these solvents are all about.
Polar Protic Solvents
First, I want to discuss polar protic solvents. The most typical examples are water, alcohols such as methanol and ethanol, and various aqueous solutions of inorganic acids. We also see solutions of carboxylic acids, which can either be neat, meaning the pure carboxylic acid itself, like glacial acetic acid, which is essentially about 98% acetic acid, or they can be aqueous or even alcoholic solutions of carboxylic acids.
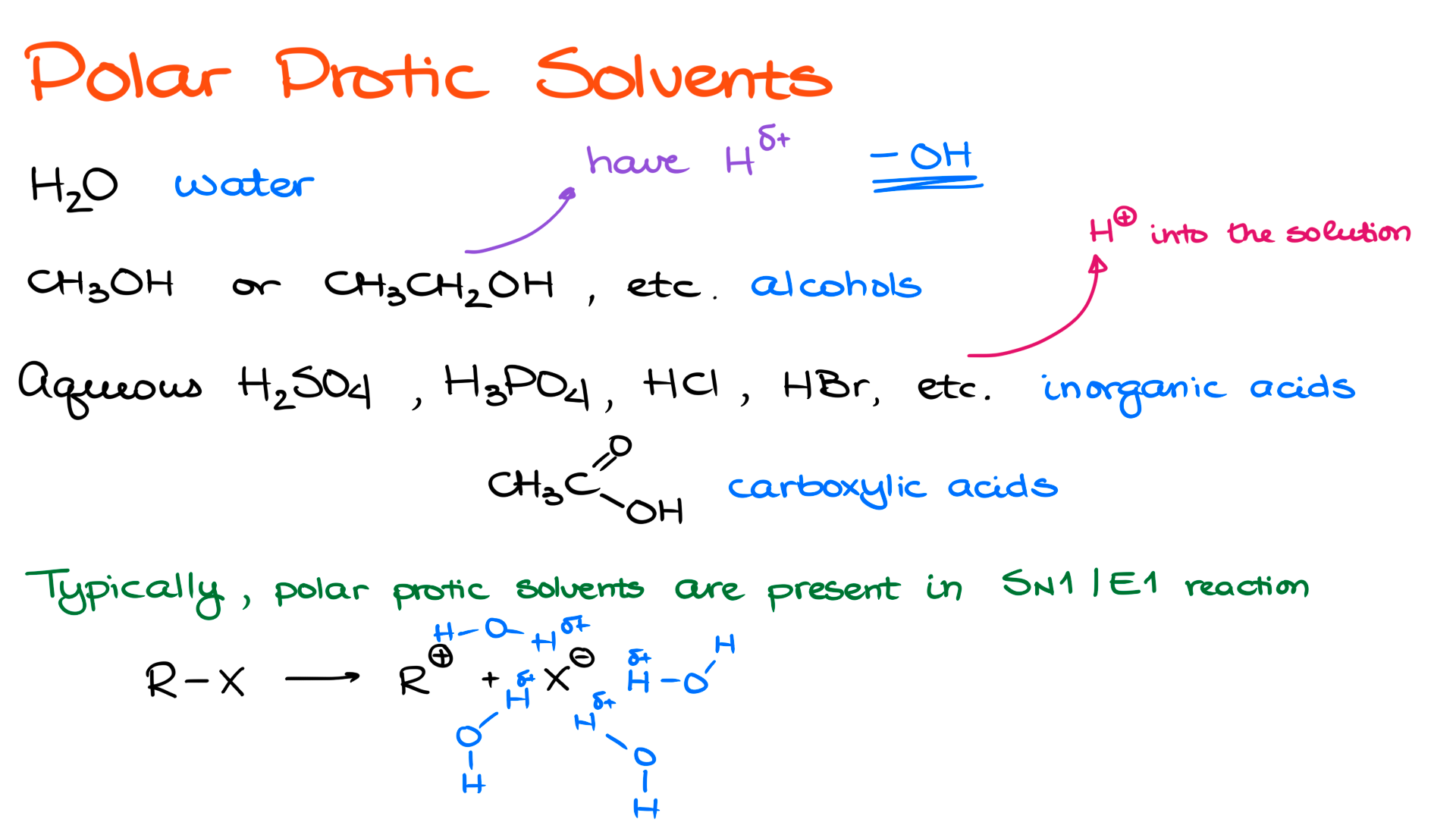
The common feature of all polar protic solvents is that they either contain H⁺ that can dissociate into solution, which is where strong inorganic acids and carboxylic acids come in, or they contain a hydrogen atom that is highly polarized because it is bonded to an electronegative atom like oxygen or nitrogen. So all of these solvents share the same key feature, either H⁺ or H(δ⁺).
Typically, we are looking either for strong inorganic acids or for molecules that contain an OH group as a source of protic conditions, and we classify those as protic solvents.
Also, because protic solvents either provide H(δ⁺) or a full positive charge in the case of dissociating strong acids, they are always polar solvents. It is impossible to have a protic solvent that is not polar at the same time. Protic solvents are always polar.
Now where do we normally see polar protic solvents? When it comes to substitution and elimination reactions, polar protic solvents are typically associated with unimolecular reactions, which means SN1 and E1 reactions.
Can SN1 and E1 reactions happen in other solvents? Sometimes. But why do we prefer polar protic solvents for SN1 and E1?
Well, if we think about either of those unimolecular reactions, we usually start with some sort of alkyl substrate bearing a leaving group, which I will abbreviate as X. When that leaving group dissociates, we typically get a carbocation, which I will show as R⁺, and also X⁻.
What the polar protic solvent does is surround X⁻ and prevent it from immediately attacking the carbocation again. So polar protic solvent helps facilitate carbocation formation and pulls the leaving group away from the molecule.
For example, if my polar protic solvent is water, it will surround X⁻ and create a kind of solvation shell around it. You can think of it as a little fur coat of solvent molecules. This prevents X⁻ from reattacking the carbocation and also stabilizes it at the same time.
So while unimolecular reactions could in principle occur without a protic solvent, that would be quite rare, because the polar protic solvent is essentially necessary to form the carbocation and stabilize the leaving group. Otherwise, there is really no good reason for the leaving group to depart in the first place. And even if it did leave, it would likely attack the carbocation again immediately and never separate far enough for another nucleophile to come in and react.
So in reactions like these, we almost always have polar protic conditions simply because that solvent is required for carbocation formation. Without it, there really is no carbocation.
Polar Aprotic Solvents
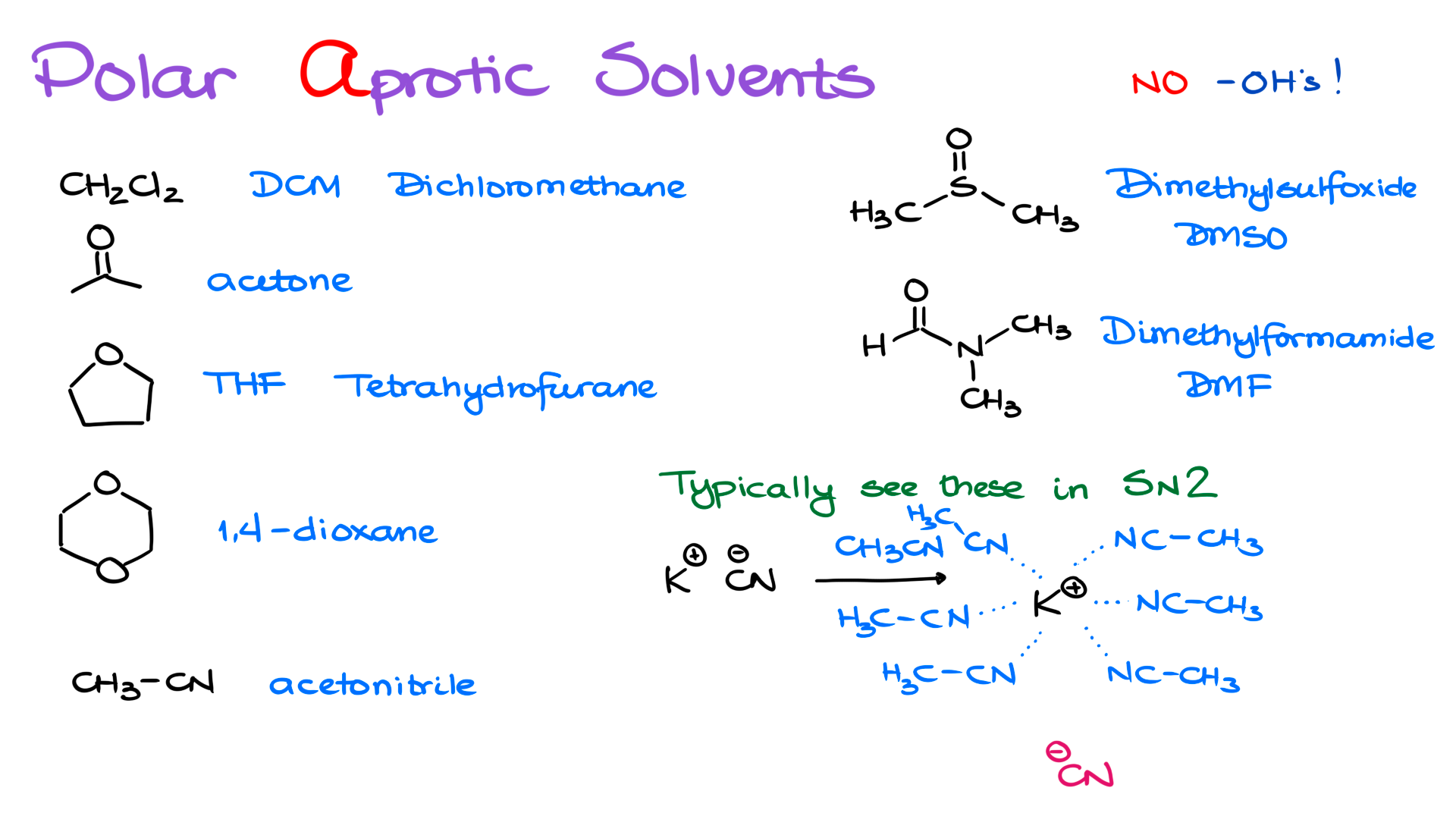
Now let’s move on to polar aprotic solvents. These compounds share one big feature, they do not have any OH groups. Other than that, they do not have many similarities beyond the fact that they are polar.
Because of that, the best way to recognize polar aprotic solvents is honestly just to memorize the common list. Fortunately, the list is not that extensive.
Among the most common polar aprotic solvents you are likely to see in your course are DCM, or dichloromethane, acetone, THF or tetrahydrofuran, 1,4-dioxane, which is often called just dioxane, acetonitrile, dimethyl sulfoxide, also known as DMSO, and finally dimethylformamide, also known as DMF.
This list is definitely not exhaustive, but these are some of the most common ones you are likely to encounter. Also pay attention to what your instructor prefers, because different instructors have different favorite solvents. Sometimes you will also see people use solvents like HMPA and others. So check your textbook, see what your instructor uses most often, and make your own list of the common solvents you are expected to know. Most likely, though, you will see all of the ones I just mentioned as well.
Now when do we normally use polar aprotic solvents?
These are commonly used in bimolecular reactions, most notably SN2 reactions. The big deal with polar aprotic solvents is that they are very good at solvating cations, which leaves the nucleophile more exposed.
Let me explain what I mean.
Suppose we have potassium cyanide, KCN. So we have K⁺ and CN⁻. If this compound is not dissolved in a suitable solvent, potassium and cyanide form a tight ion pair. There is no reason for them to separate. They are electrostatically attracted to each other, and they stabilize each other simply by sticking together.
What a polar aprotic solvent does is separate those ions. So if I take potassium cyanide and dissolve it in acetonitrile, the solvent comes in and starts pulling the ions apart. In a way, this is similar to what polar protic solvents do for anions, except now the aprotic solvent is especially good at surrounding the cation.
Here the solvent molecules surround K⁺, pulling it away from CN⁻. That means the cyanide ion is left relatively naked, without its counterion closely associated with it. And now that cyanide is much more reactive. It will aggressively pursue any reaction that allows it to donate that excess electron density and become more stable. In other words, it becomes a much stronger nucleophile.
So by using a polar aprotic solvent in an SN2 reaction, we increase the nucleophilicity of the anionic species and make the reaction proceed more readily. I like to joke that the solvent makes the nucleophile naked and aggressive.
Without the polar aprotic solvent, the SN2 reaction is still possible, but it is much less efficient because the cation remains closely associated with the anionic nucleophile. But once the solvent pulls those ions apart, with the cation wrapped in solvent molecules and the anionic nucleophile left exposed, the reaction becomes much easier.
So now you know the role of polar protic solvents in SN1 and E1 reactions, and why we use polar aprotic solvents in SN2 reactions.
