Sharpless Epoxidation
In this tutorial I want to talk about enantioselective epoxidation.
So, when it comes to regular epoxidation reactions, we typically see two main scenarios. Either we react an alkene with a peroxy acid like mCPBA or another peroxy acid, giving us the corresponding epoxide, or, if we have an electron-deficient alkene, we can use hydrogen peroxide in basic media, that’ll also give us the epoxide.
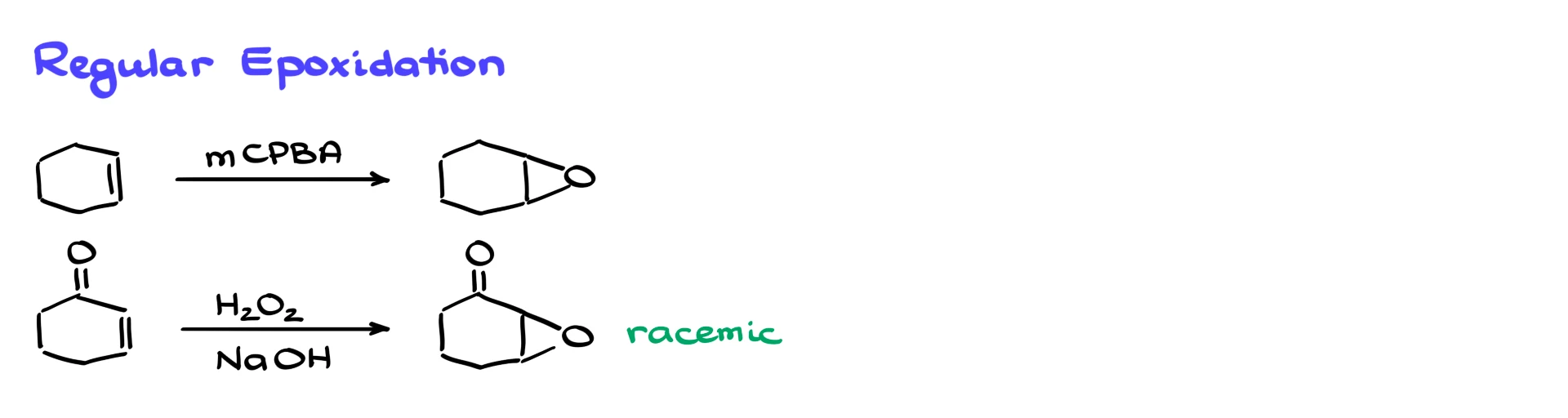
But the obvious problem with both of these methods is that they’re not stereoselective. So, if multiple stereoisomers are possible, you’re going to get a mix.
Now, there are some ways to persuade the reaction to give one stereoisomer over the other. For instance, if there’s a very bulky group, the epoxide will usually form away from it. But then we have the problem of what to do with that bulky group after the reaction, especially if we no longer need it.
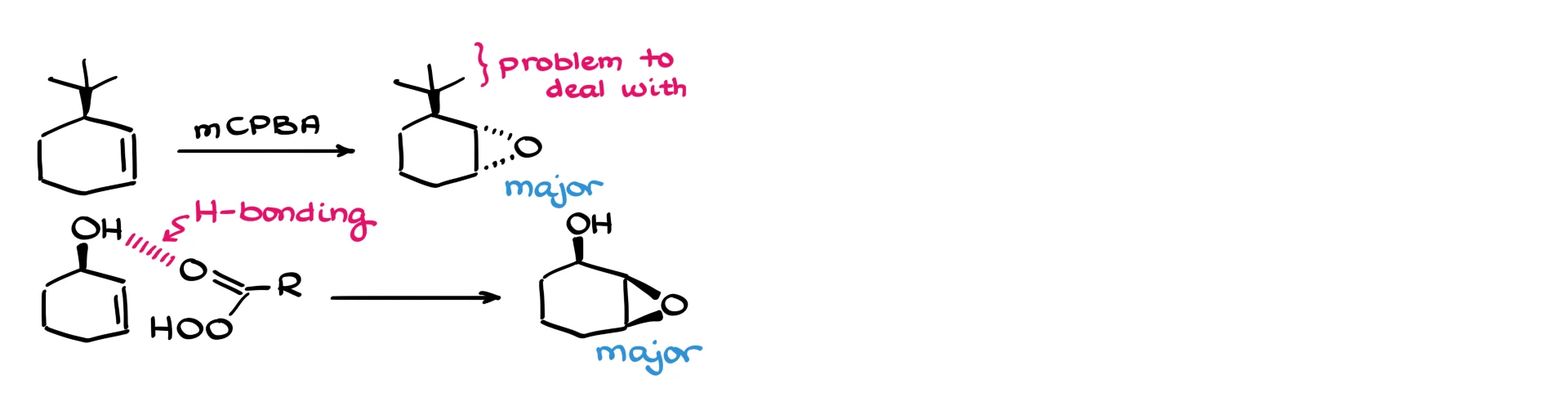
Another example is when the molecule has an –OH in the allylic position. In that case, hydrogen bonding can coordinate the peroxy acid around that –OH group, and the epoxide will end up facing us. But if the molecule is more complex, if it has more –OH groups or other hydrogen bonding sites, this approach quickly falls apart, and it’s not very selective to begin with.
This is where the Sharpless epoxidation really shines.
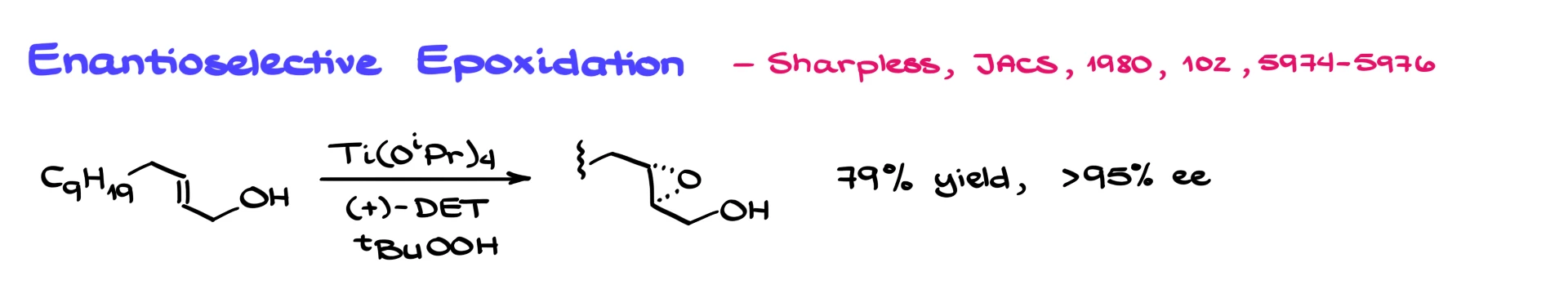
In their original 1980 paper published in JACS, Tsutomu Katsuki and Barry Sharpless described about a dozen examples showing that, by using titanium complexes with corresponding tartrates, we can not only get decent yields (typically 70–90%) but also achieve excellent enantiomeric excess in the epoxidation of various allylic alcohols.
So, in this tutorial, I want to talk about the details of the Sharpless epoxidation, how it works and why it works the way it does.
Sharpless Epoxidation Components
Probably the best place to start is by looking at the components of the reaction, because this is a multi-component system.
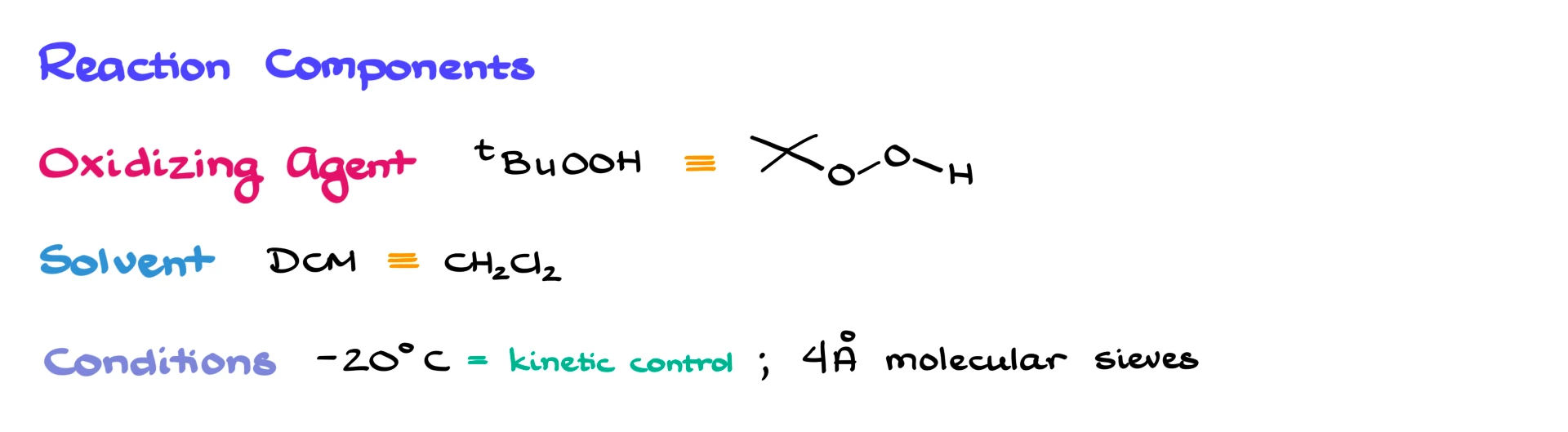
The oxidizing agent is tert-butyl hydroperoxide, which is a fairly common oxidizer in more advanced organic chemistry, though we don’t usually discuss it much in introductory courses.
The reaction is typically carried out in dichloromethane, although other solvents may also be used. It’s run under kinetic control, usually at comparatively low temperatures (around -20 °C) but there are many examples at both lower and higher temperatures.
The components are moisture-sensitive, so you’ll usually see the use of 4 Å molecular sieves to capture any water that may have snuck into the reaction mixture.
Now, the fun part: the catalyst system.
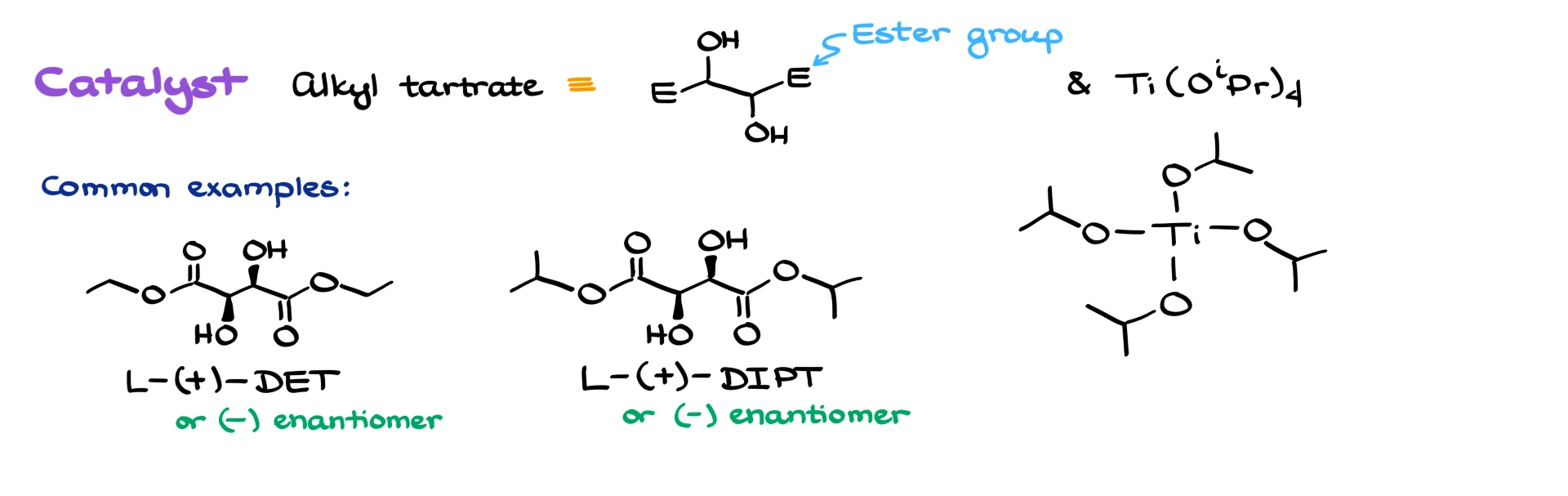
We use a dialkyl tartrate. Here I’ve shown a generic structure with ester groups abbreviated as “E” and titanium tetraisopropoxide. The two most common tartrates are:
- Diethyl tartrate, shown on the left.
- Diisopropyl tartrate, shown on the right.
Each tartrate has two stereocenters, so we can use either the dextrorotating or levorotating isomer depending on the stereochemistry we want. The meso compound, which these molecules can also form, is completely useless for our purposes, so we don’t care about it here.
General Mechanism of the Sharpless Epoxidation
Alright, now that we know what goes into our “soup,” let’s look at the general mechanism.
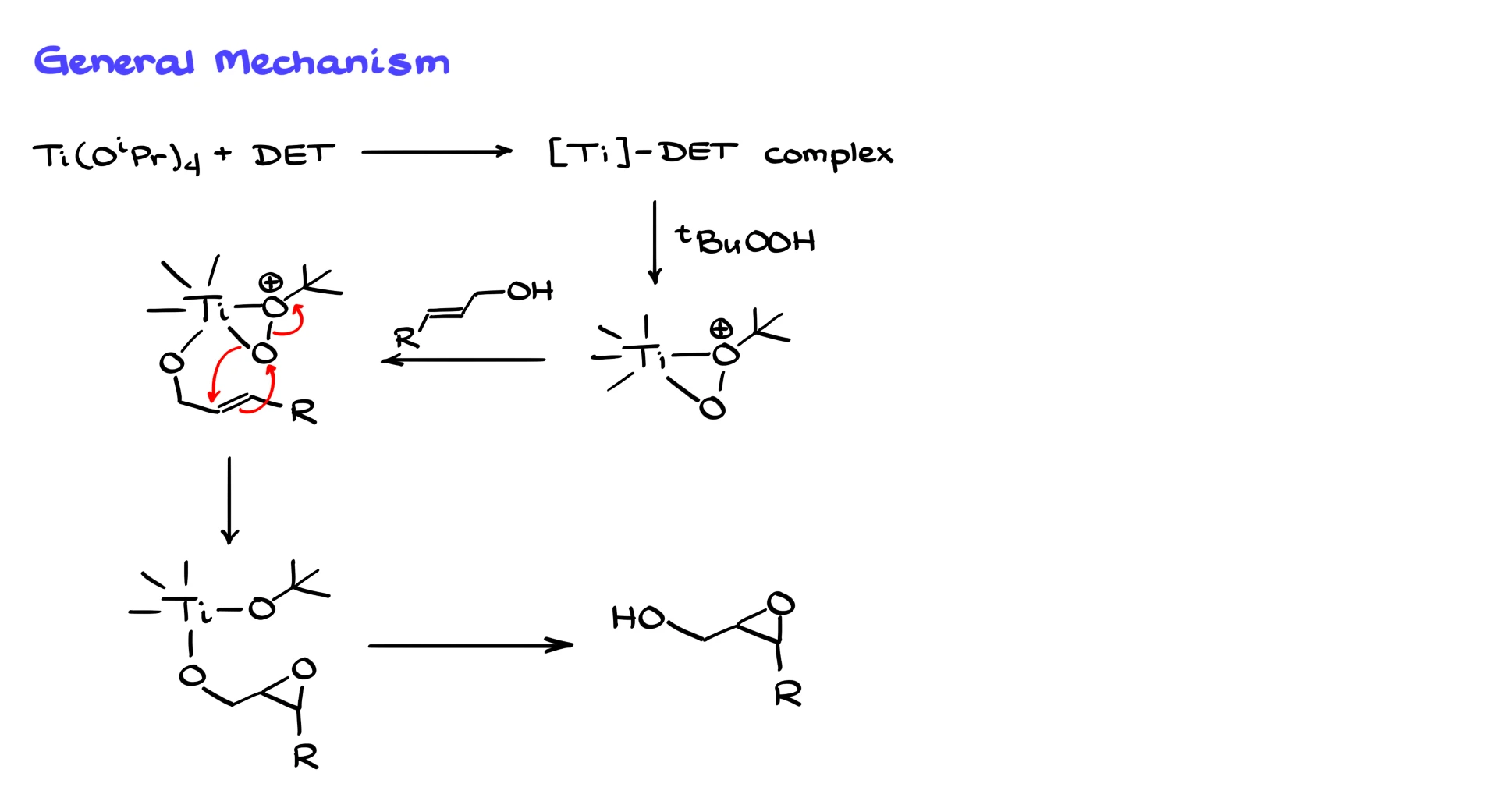
The reaction starts with titanium reacting with the tartrate to form a complex. I’ll talk about this complex in detail later, but for now, let’s just call it “some sort of magic complex” and not worry too much about what it looks like.
Next, we bring in tert-butyl peroxide, which coordinates with titanium and forms this funky-looking structure. I’ve specifically marked the tert-butyl peroxide part; the rest of the ligands connected to titanium aren’t important for this moment.
Then we bring in our allylic alcohol, which also coordinates around titanium. And now that all the components are in place, we move on to the epoxidation step.
The mechanism here is fairly typical. The electrons from the alkene reach for the oxygen, the O–O bond breaks, and the oxygen inserts onto the other carbon of the double bond. Once the product is released from titanium, we get our final epoxide.
So from a mechanistic standpoint, outside of coordinating everything around titanium, it’s a pretty straightforward reaction.
Sharpless Catalyst Complex Structure
But here’s where the fun begins: the structure of the complex.
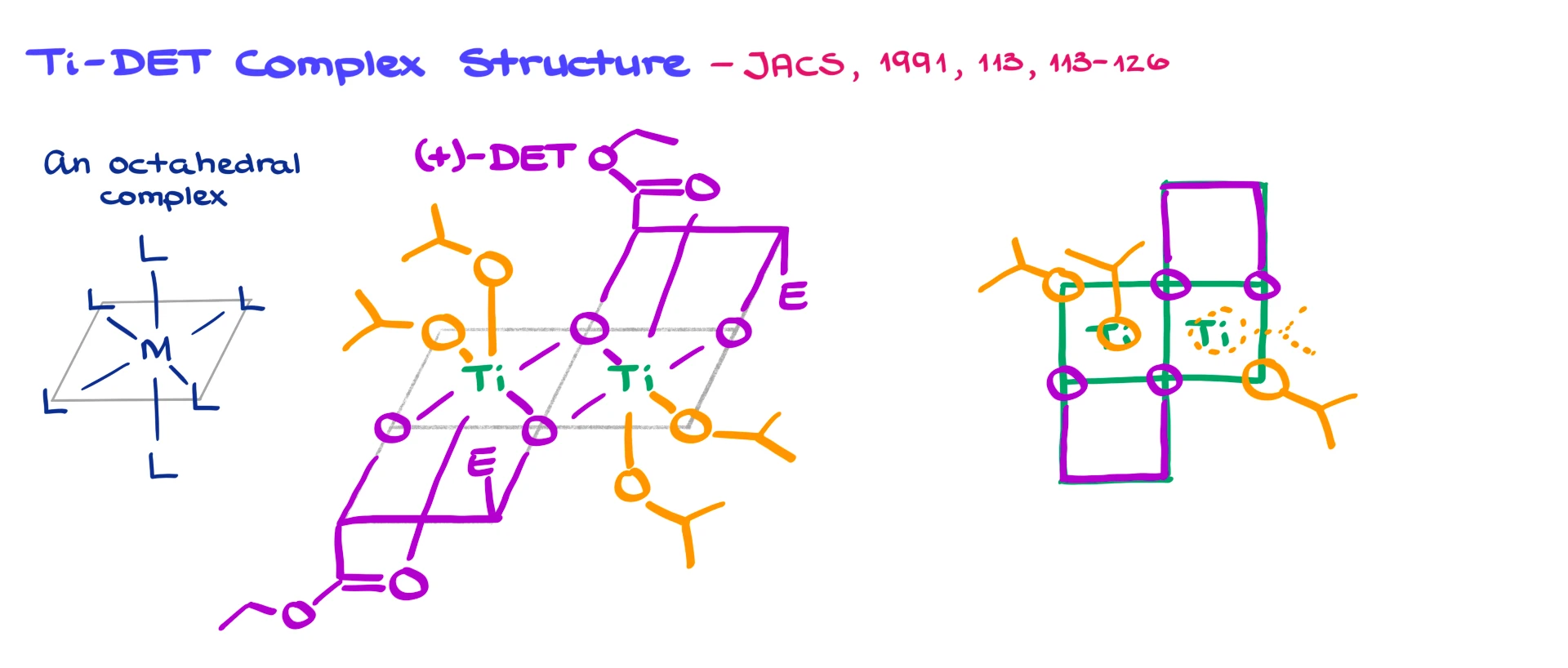
A lot of research has gone into figuring out what this complex looks like. There have been many proposals, but it’s now established that we’re dealing with octahedral complexes of titanium.
As a quick reminder, in an octahedral complex, we have a central metal with four equatorial ligands and two axial ligands. Now, in the Sharpless epoxidation, it’s not just one titanium center—it’s actually two!
So let me draw a rough guide for this.
On the right, we have one octahedral titanium complex, and on the left, another. I’ll put Ti in the center of each. I’m going to use the (+)-diethyl tartrate (DET) for this example.
The first tartrate coordinates to the titanium on the right, with one ester group drawn and the second abbreviated as “E.” The second tartrate does the same on the left side.
Then we add isopropoxide ligands. On the right, one goes in the corner and another at the bottom. Same thing on the left.
Now we finish building the octahedral geometry by coordinating the ester oxygen atoms to the titanium.
So, on the right side, I have titanium with a base, one vertical bond, and another vertical bond. On the left, same setup.
If I were to draw a top view, it would look like four squares arranged in a grid. Titanium sits in the center, with one tartrate in the top right, another in the bottom left. We’ve got isopropoxy groups in the two remaining corners, and axial groups above and below.
Next, we coordinate tert-butyl peroxide to the right titanium. To do that, I erase one of the isopropoxy groups and replace it with t-BuOOH.
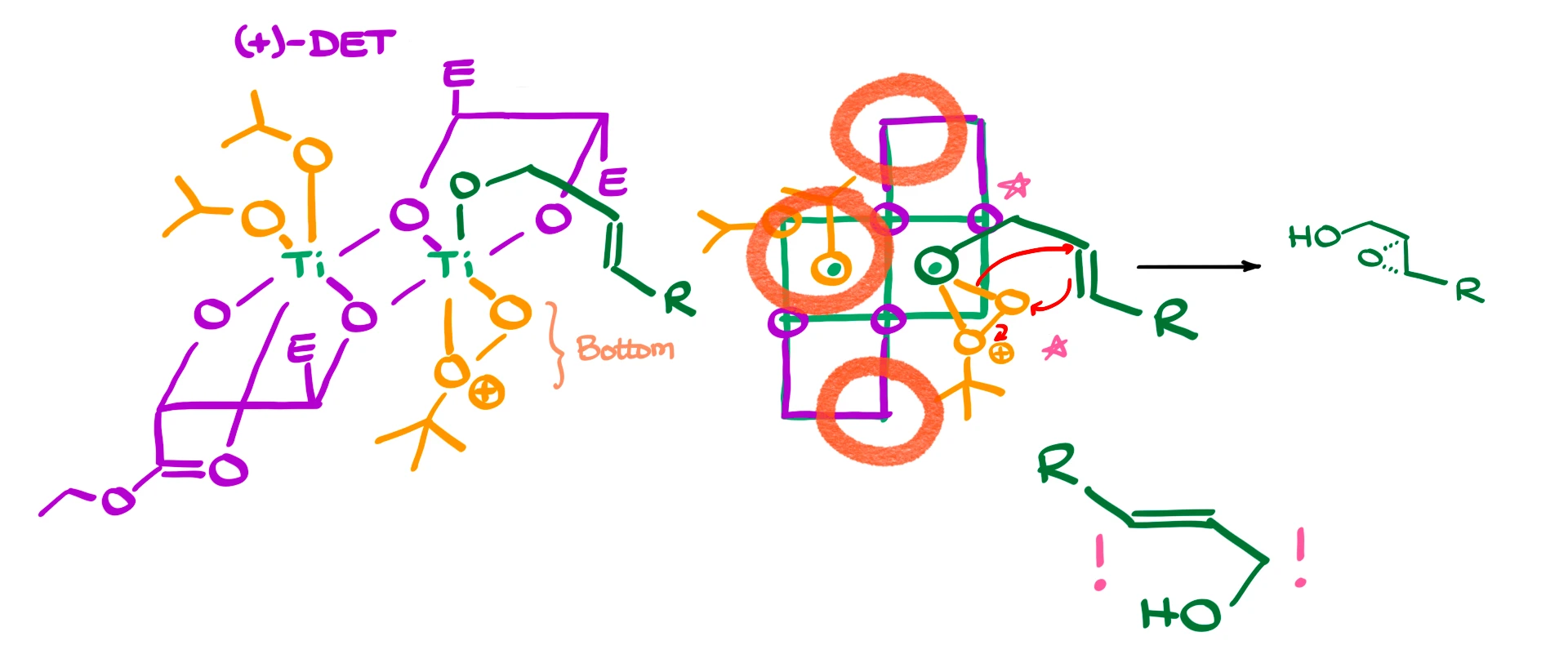
Then we bring in our allylic alcohol, which comes in and coordinates in place of one of the ester groups—so I erase that ester and draw the alcohol instead.
At this point, it gets messy, so let’s clean up and focus on the final active complex.
If we return to the top view, with all components placed, there’s something important to highlight: steric hindrance.
- This esters create a steric wall.
- The isopropoxy group next to it does the same.
- This other ester adds more hindrance.
So, the allylic alcohol is forced to coordinate in a very specific way.
That means the alkene and the peroxide will react in a fixed geometry, giving us the epoxide on a specific face of the molecule. In the way I’ve drawn this, the peroxide is on the bottom, so the epoxide ends up pointing down—away from us.
And just like in most stereoselective reactions, the transition state geometry controls everything. The molecule physically can’t react any other way, which is why we get enantiomeric excess values as high as 95–98%, sometimes nearly 100%.
And now you can see why the position of the –OH group and what’s cis to it is so important.
- If there’s something bulky cis to the –OH, that part will overlap with the peroxide, slowing the reaction down.
- If there’s something bulky on the carbon with the –OH, that’ll also interfere with coordination and hurt the reaction.
So two things to remember:
- The molecule should adopt a conformation where the –OH is tucked in toward the double bond.
- Avoid bulky groups cis to –OH and on the carbon holding the –OH.
Mnemonic Device to Predict the Stereochemistry of the Sharpless Epoxidation
But of course, going through this huge complex every time you want to predict stereochemistry isn’t practical.

So here’s the mnemonic I use:
Draw the double bond horizontally, and imagine the –OH group as a handle sticking up. As a right-handed person, I imagine grabbing the molecule by that –OH.
- If you use (+)-DET, the epoxidation happens from the top face.
- If you use (–)-DET, it happens from the bottom face.
So, for example, take the simplest allylic alcohol. Using (+)-DET, you get the epoxide looking up. With (–)-DET, the epoxide points away from you.
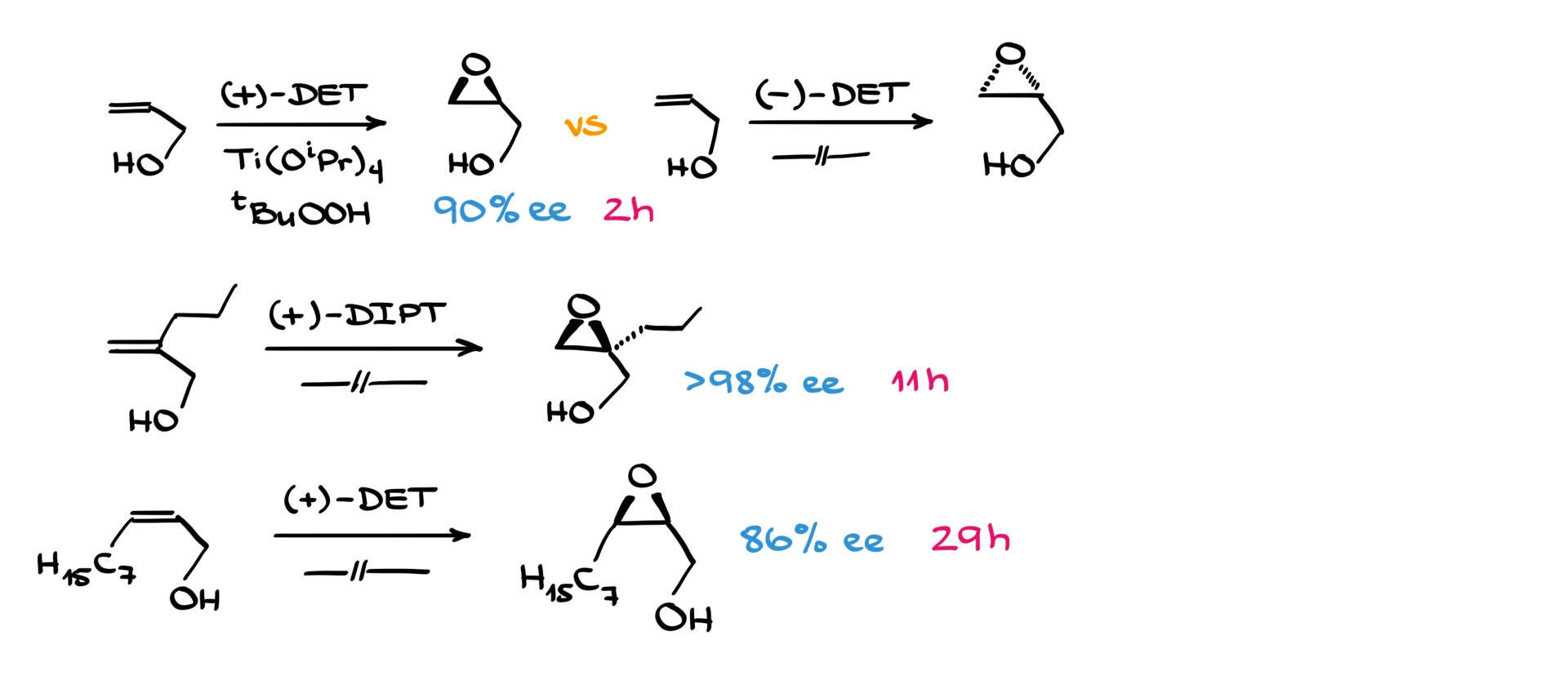
The enantiomeric excess is excellent. With 90% ee, that’s 95% of one stereoisomer and 5% of the other. Using diisopropyl tartrate can give you 98% ee, meaning 99:1 stereoselectivity.
Here’s another example that illustrates the problem with bulky groups cis to –OH. In terms of reaction rates, we go from a couple of hours to 11 hours, and then with a bulky group present, it jumps to almost 30 hours.
Kinetic Resolution Using the Sharpless Epoxidation
The rate also depends on the carbon bearing the –OH.
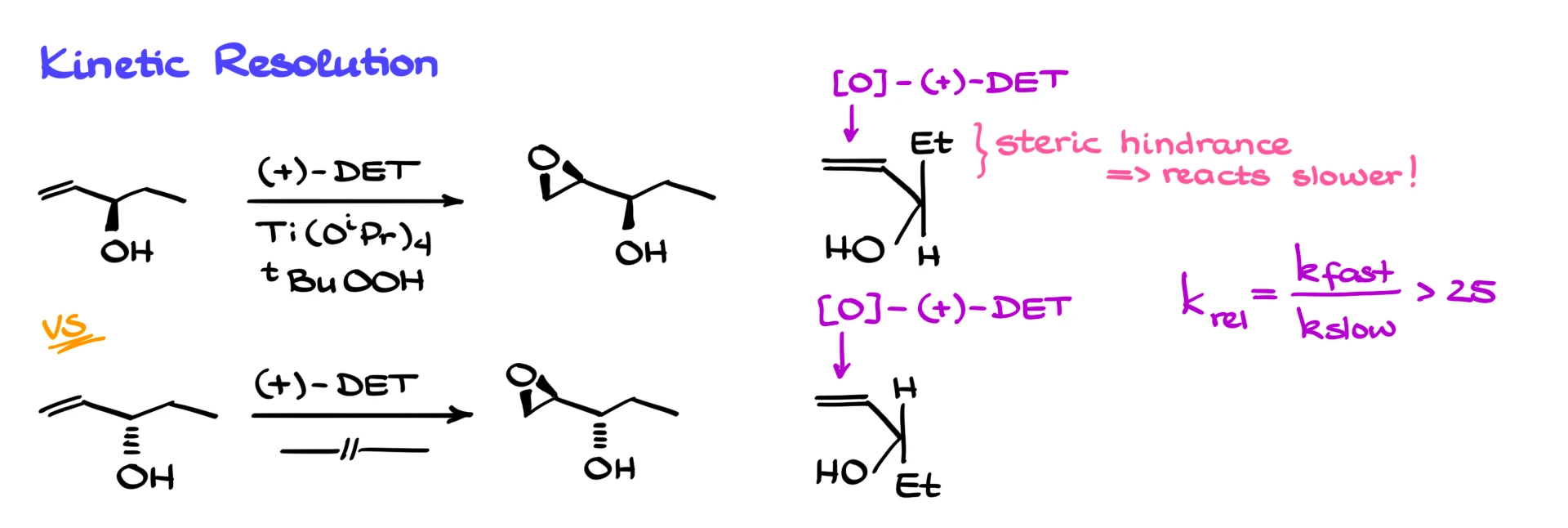
Here’s an example with two enantiomeric allylic alcohols. Under identical conditions: same titanium, same t-BuOOH, same tartrate, the only difference is the chirality of the starting material.
Using our mnemonic, we can predict the product stereochemistry. But one reaction will be fast, and the other will be glacially slow. The bulky group on one enantiomer clashes with the titanium complex, and the difference in rates can be 25, 30, even 50 times.
So if we start with a racemic mixture, one enantiomer reacts fast, the other slow. If we stop the reaction early, we’ll mostly have:
- The epoxide from the fast-reacting enantiomer.
- The unreacted alcohol from the slow-reacting one.
That means we can kinetically resolve enantiomers!
And this trick even works for achiral molecules. Let’s say we have an alcohol with two alkenes. Subject it to Sharpless conditions, and one alkene will react fast, the other slow.
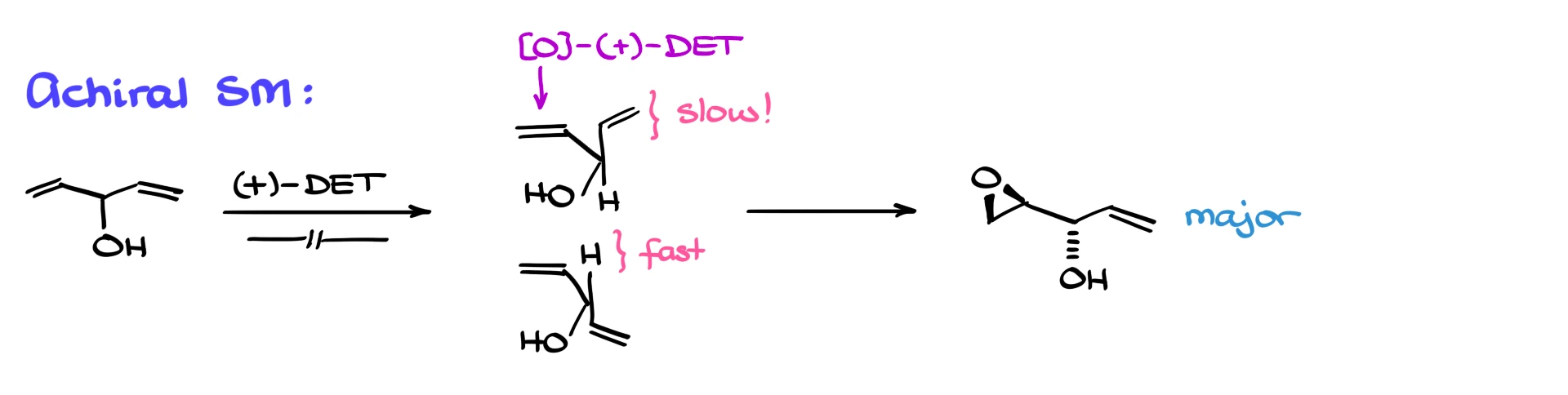
Use the mnemonic to figure out which is which. The alkene that’s not clashing with the titanium complex will react first, giving us a selective epoxide.
And if you’re struggling to visualize this in 3D, grab your model kit and build the molecule. Stereochemistry is never easy.
Before I wrap up, I have a little challenge for you.
If you take the product from this reaction and continue reacting it with (+)-DET, where will the second epoxide go? Will it look at us or away from us? And will that step be fast or slow?
