Cannizzaro Reaction
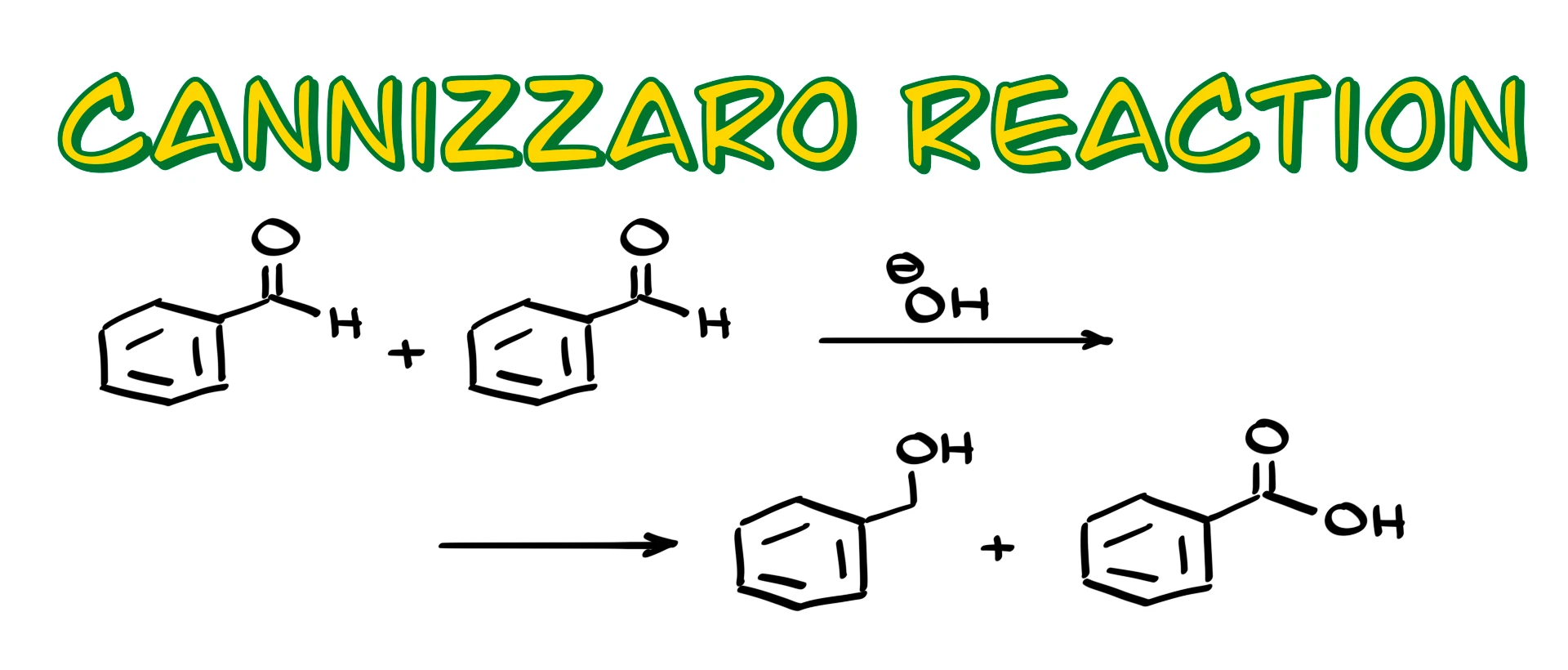
In this tutorial I want to talk about the Cannizzaro reaction, a classic disproportionation reaction of aldehydes in strongly basic media. The outcome of this reaction is an alcohol and a carboxylic acid, or more accurately a carboxylate, formed in a one-to-one ratio.
Mechanism of Cannizzaro Reaction
Let’s start with the basic mechanism.
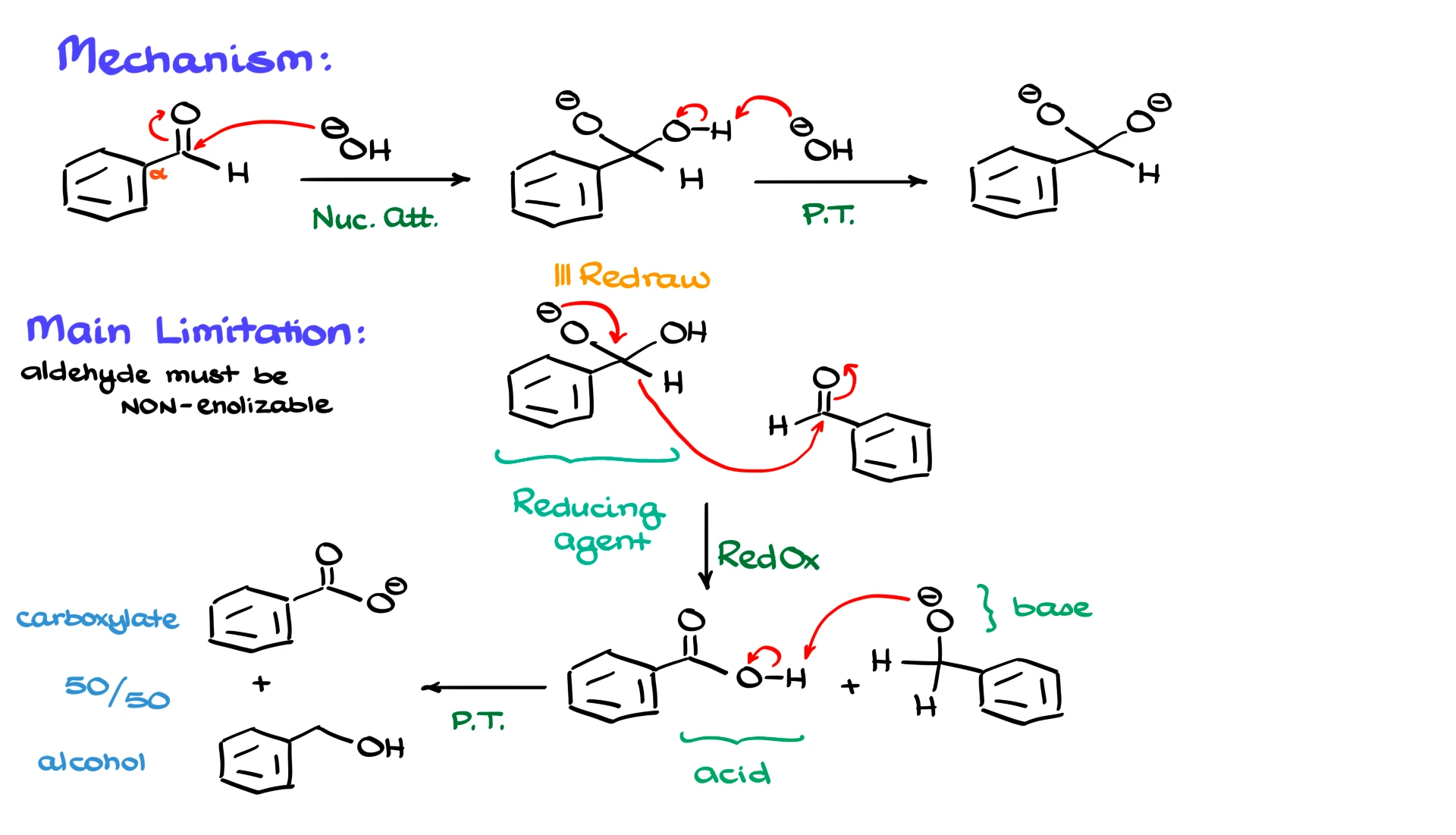
We need an aldehyde, and I will use benzaldehyde as my example, and we also need a strong base, so I am going to bring in OH⁻. The first step is a straightforward nucleophilic addition. Hydroxide attacks the carbonyl carbon of the aldehyde, giving a tetrahedral intermediate.
Because Cannizzaro reactions are typically carried out in very concentrated base, the OH group on that tetrahedral intermediate can be deprotonated further. If another equivalent of OH⁻ is present, a simple proton transfer can occur, giving a dianionic species. Whether this extra deprotonation happens depends on the base concentration and reaction conditions. To keep things simple, I will continue with the monoanionic intermediate, since both pathways ultimately lead to the same products.
This anionic intermediate is very different from the original aldehyde. Instead of being electrophilic, it is now electron rich. If we bring in another equivalent of aldehyde, we get the key redox step of the Cannizzaro reaction. The negatively charged species acts as a reducing agent. The electrons on oxygen collapse back down to reform the carbonyl, and at the same time a hydride, meaning hydrogen with both electrons, transfers to the second aldehyde molecule. One aldehyde is oxidized to a carboxylic acid, and the other is reduced to an alkoxide.
At this point we have an acidic species, the carboxylic acid, and a basic species, the alkoxide. These two cannot coexist, so an immediate proton transfer occurs. The alkoxide grabs the proton from the carboxylic acid, giving the final products: an alcohol and a carboxylate. If we want the free carboxylic acid, we simply perform an acidic workup at the end.
If the reaction proceeds through the dianionic intermediate instead, the carboxylate is formed directly and the final proton transfer step is skipped. Either way, the net result is the same. One aldehyde molecule is reduced to an alcohol, and another is oxidized to a carboxylate. Under ideal conditions the two products form in a 50:50 ratio.
Limitations of the Cannizzaro Reaction
Now for the limitations. The most important restriction of the Cannizzaro reaction is that the aldehyde must be non-enolizable. In other words, the α-position next to the carbonyl must not contain any acidic hydrogens. Under the strongly basic conditions required for this reaction, aldehydes that have α-hydrogens can form enolates. Once enolates are present, side reactions such as aldol reactions quickly take over and you end up with a messy mixture instead of clean Cannizzaro chemistry.
Another practical issue is efficiency. In the classic Cannizzaro reaction you lose half of your aldehyde to oxidation, which makes the reaction unattractive for synthesis. Because of that, the simple Cannizzaro reaction is more of a conceptual reaction than a workhorse transformation.
Crossed Cannizzaro Reaction
To get around this problem, chemists often use the crossed Cannizzaro reaction.
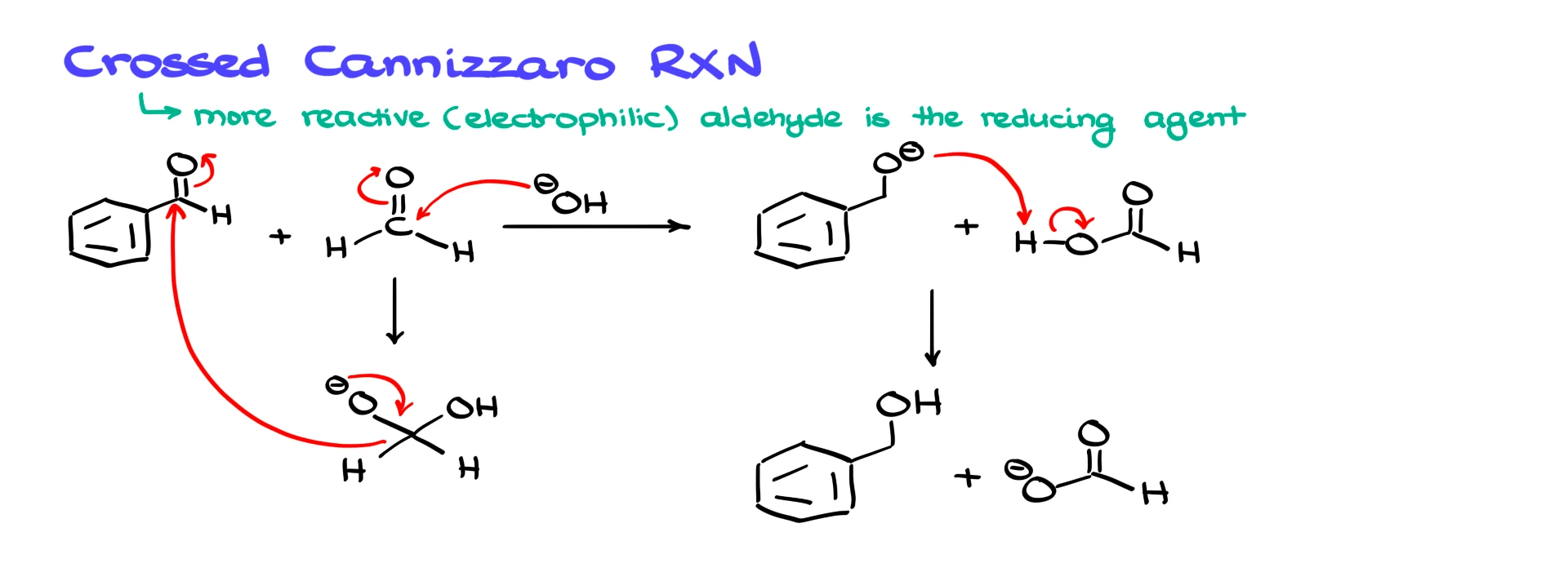
In this version, two different aldehydes are used. One aldehyde acts as a sacrificial reducing agent, and the other is the aldehyde you actually care about. The classic sacrificial aldehyde is formaldehyde. Formaldehyde is extremely electrophilic and very cheap, which makes it ideal for this role.
For example, if we mix benzaldehyde with formaldehyde under strongly basic conditions, hydroxide attacks formaldehyde preferentially. That gives an anionic intermediate derived from formaldehyde. This species then donates a hydride to benzaldehyde. Benzaldehyde is reduced to benzyl alcohol, while formaldehyde is oxidized to formate. A proton transfer then gives benzyl alcohol and the formate anion. In this way, we avoid losing half of our valuable aldehyde, and we can obtain the alcohol in a much better yield.
Even so, it is still fair to ask why one would use a Cannizzaro reaction instead of a simple reducing agent like sodium borohydride. In practice, Cannizzaro chemistry is more important conceptually than synthetically, but it does show up in problem solving and mechanism questions.
Tishchenko Reaction
Before wrapping up, let’s briefly talk about a related variation called the Tishchenko reaction.
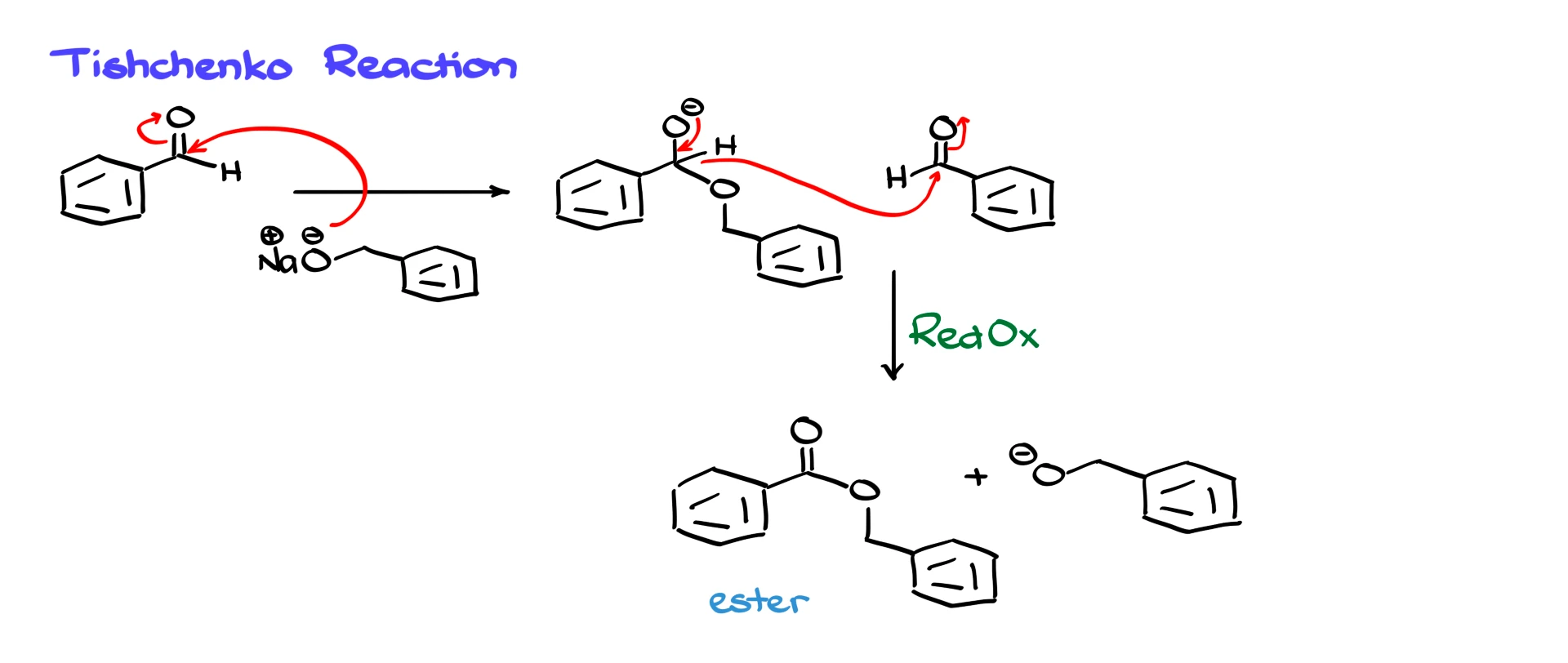
In the Tishchenko reaction, instead of using hydroxide as the base and nucleophile, we use an alkoxide. The aldehyde is still the electrophile, and the alkoxide attacks the carbonyl to form a tetrahedral intermediate.
This intermediate then acts as a reducing agent, just like in the Cannizzaro reaction. A hydride transfers to another aldehyde molecule, but the key difference is the product. Instead of forming a carboxylic acid or carboxylate, the reaction produces an ester. At the same time, an alkoxide is regenerated. Because the alkoxide formed is the same species we started with, the alkoxide can be used catalytically rather than in stoichiometric amounts.
The Tishchenko reaction is a neat way to convert aldehydes directly into esters, where both the acid part and the alcohol part of the ester come from aldehyde chemistry. It is somewhat niche, but it is a nice illustration of how small changes in conditions can lead to very different outcomes.
That is the Cannizzaro reaction and its Tishchenko variation in a nutshell. Non-enolizable aldehydes, strong base, hydride transfer, and disproportionation. If you want to see how this chemistry connects to other rearrangements and redox reactions, stay tuned for the next tutorial.
