SN1 vs SN2 Reactions
In this tutorial I want to talk about SN1 vs SN2 reactions, the conditions under which they occur, and how to distinguish between them on an exam.
SN1 Reactions
Let’s start with SN1 reactions.
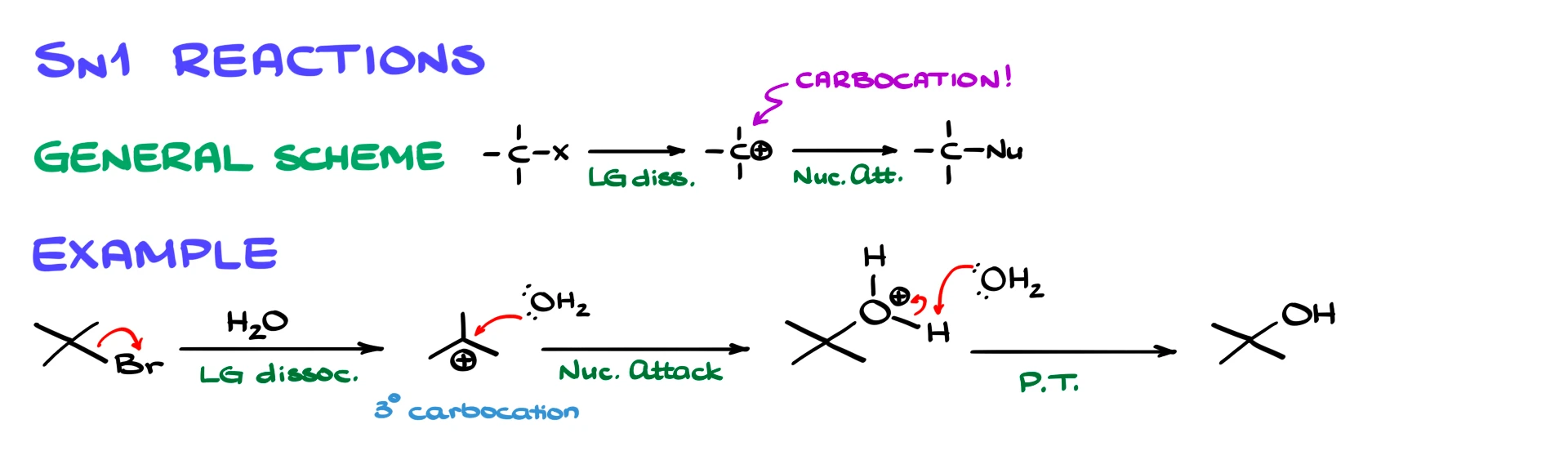
SN1 reactions occur in multiple steps. The first step is leaving group dissociation, which forms a carbocation. In the second step, a nucleophile attacks that carbocation, giving a protonated intermediate. Finally, a proton transfer gives the neutral product.
For example, if we take tert-butyl bromide and react it with water, the first step is the loss of bromide to form a tertiary carbocation. Then water attacks the carbocation, forming a protonated alcohol. A second molecule of water removes the extra proton, giving the final alcohol product.
SN2 Reactions
Now let’s compare that to SN2 reactions.
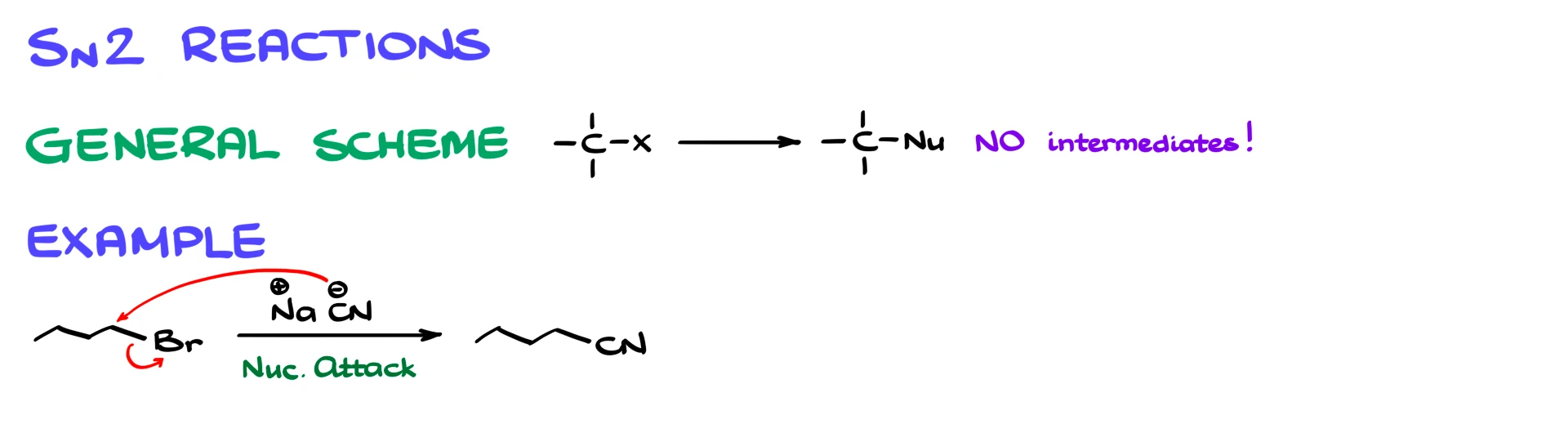
SN2 reactions are concerted, meaning everything happens in one step. There are no intermediates. The nucleophile attacks the carbon at the same time as the leaving group leaves.
For example, if we take 1-bromobutane and react it with sodium cyanide, the cyanide ion attacks the carbon and displaces bromide in a single step, giving the product directly.
So in a nutshell, SN1 involves a carbocation intermediate and proceeds stepwise, while SN2 is a one-step concerted mechanism with no intermediates.
Compare SN1 and SN2 Reactions
Let’s summarize the key differences.
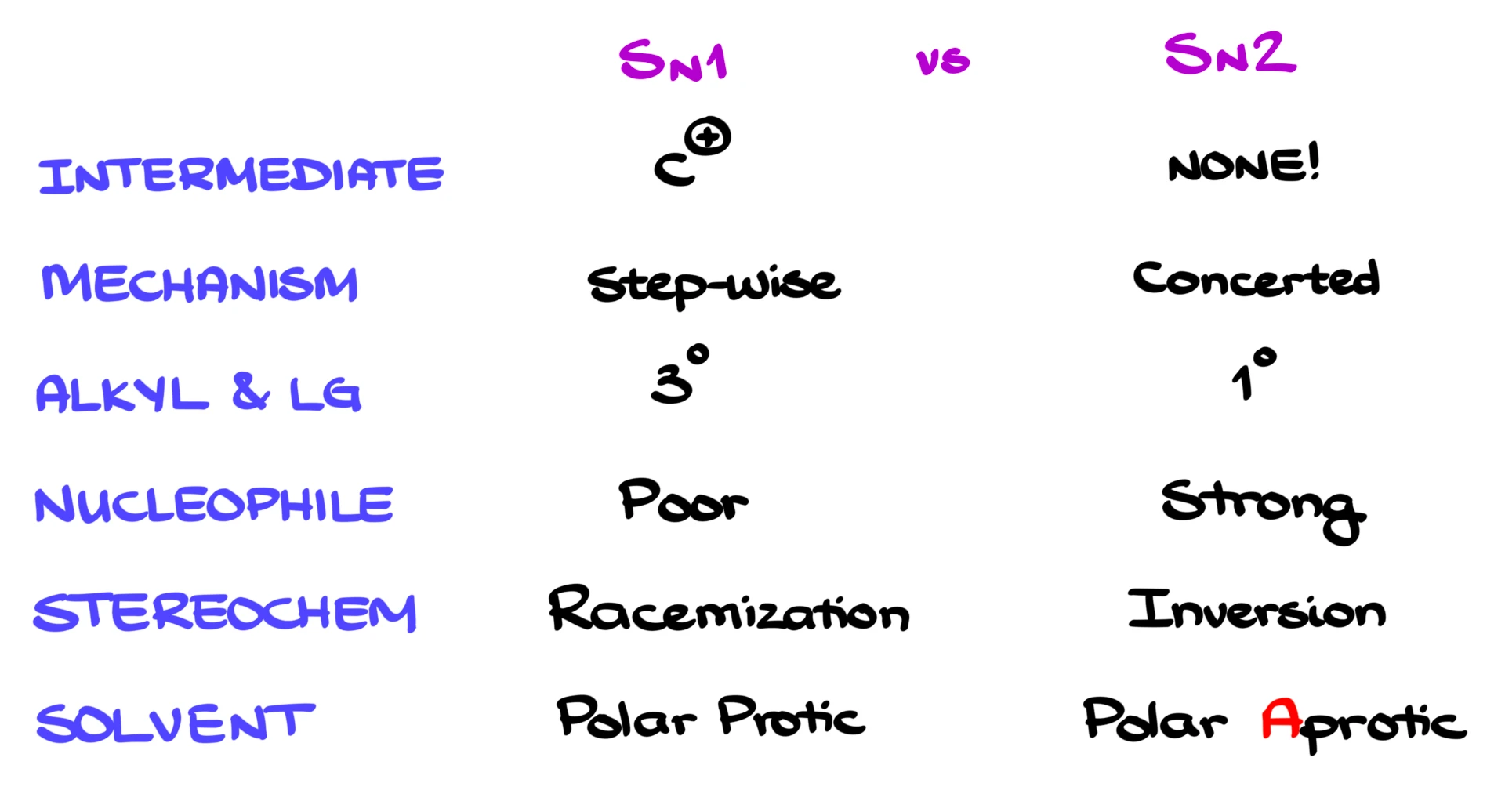
In SN1 reactions, we form a carbocation, so we typically need a tertiary substrate to stabilize that carbocation. SN2 reactions, on the other hand, are very sensitive to steric hindrance, so they work best with primary substrates.
In terms of nucleophiles, SN1 reactions usually involve weak nucleophiles, while SN2 reactions require strong nucleophiles.
From the stereochemistry standpoint, SN1 reactions give a mixture of products if the starting material is chiral, because the carbocation can be attacked from either side. SN2 reactions give inversion of configuration due to backside attack.
And finally, for solvents, SN1 reactions are favored in polar protic solvents, while SN2 reactions are favored in polar aprotic solvents.
Nucleophiles and Solvents in SN1 and SN2 Reactions
Now let’s talk about nucleophiles.
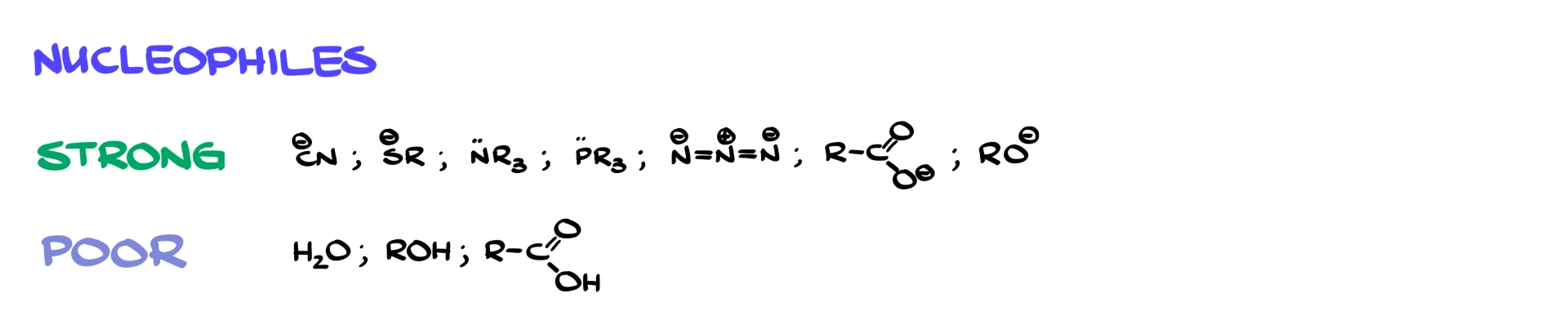
Strong nucleophiles are usually negatively charged or highly reactive species. Common examples include cyanide, sulfur-containing nucleophiles, nitrogen-containing nucleophiles, azide, alkoxides, carboxylates, and phosphines.
Weak nucleophiles are much simpler. Typically, these are neutral molecules like water, alcohols, or acids.
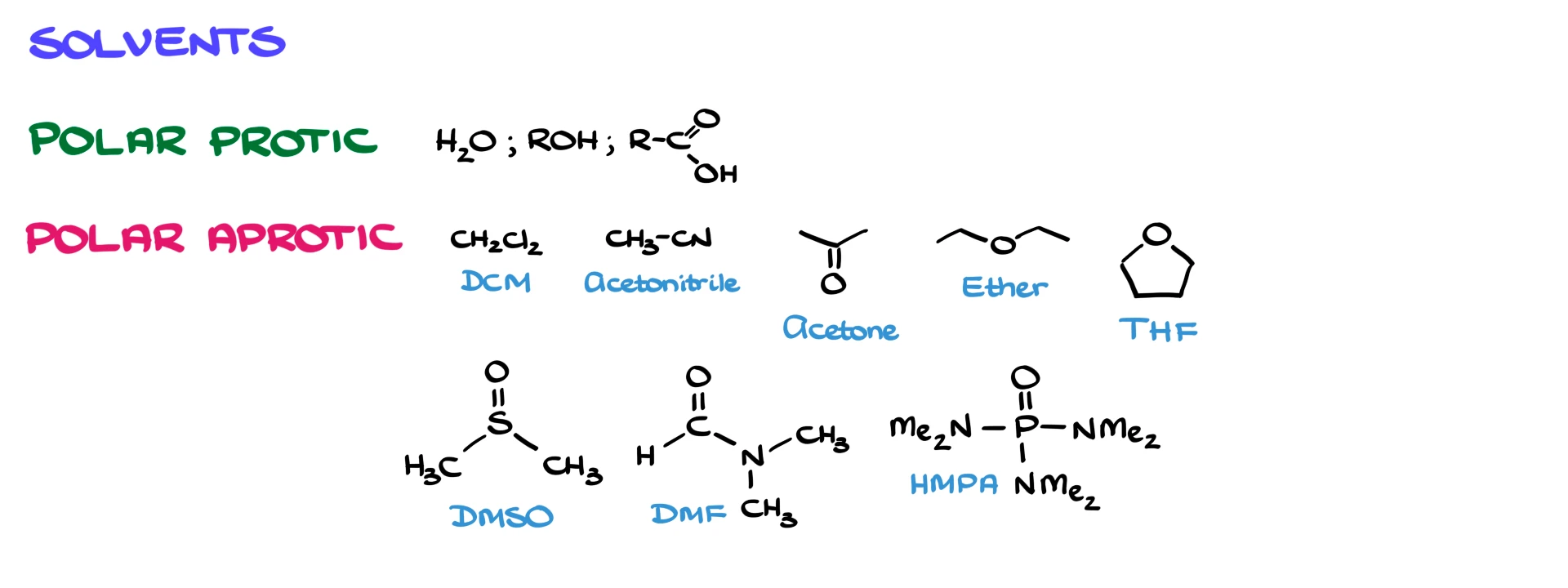
Interestingly, these weak nucleophiles are also commonly used as polar protic solvents, so you often see water or alcohols playing both roles.
For polar aprotic solvents, the list is a bit more varied. Common examples include dichloromethane, acetonitrile, acetone, diethyl ether, tetrahydrofuran (THF), dimethyl sulfoxide (DMSO), dimethylformamide (DMF), and sometimes hexamethylphosphoramide (HMPA). There is not much of a pattern here, so it is mostly a matter of recognizing the common ones used in your course.
Predictive Model
Now let’s build a simple predictive model.
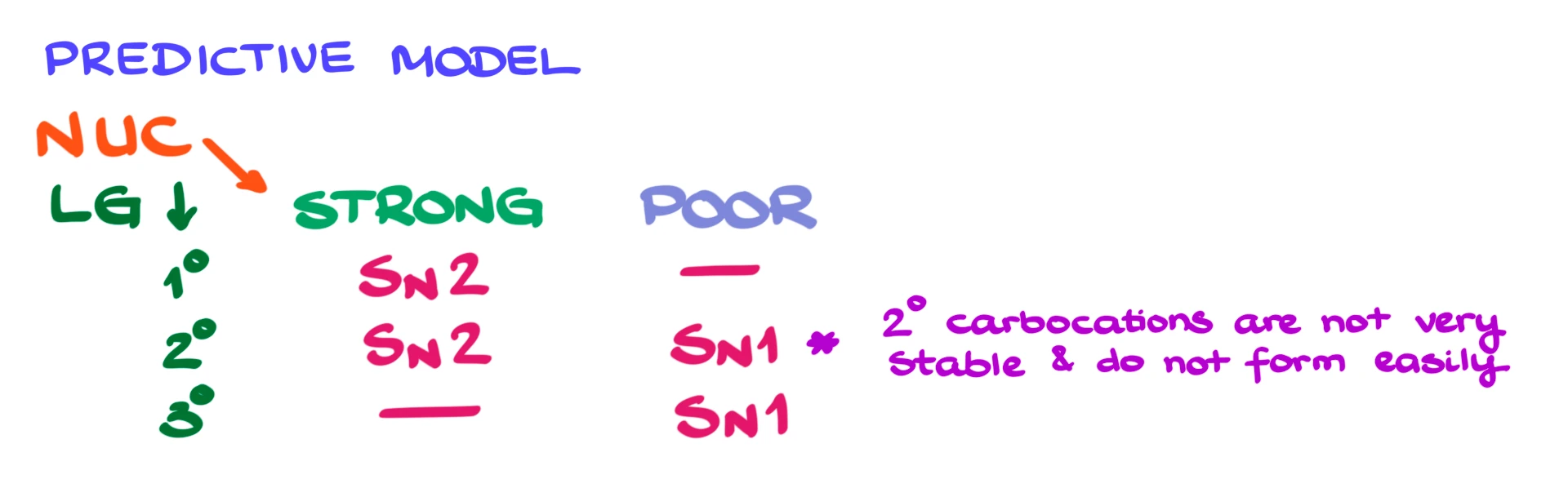
Whenever you see a substitution reaction, look at two things. First, identify the substrate, whether the leaving group is on a primary, secondary, or tertiary carbon. Second, determine whether the nucleophile is strong or weak.
From there, you can usually predict the mechanism.
If you have a primary substrate and a strong nucleophile, that points strongly toward SN2. If you have a tertiary substrate and a weak nucleophile, that points toward SN1.
Secondary substrates can go either way, and this is where things depend more on the specific conditions.

One important note is that some instructors treat secondary carbocations as too unstable and may not consider SN1 reactions for secondary substrates. So make sure you know how your instructor approaches this.
Examples
Now let’s look at a few examples.

If we react a primary alkyl bromide with cyanide, cyanide is a strong nucleophile and the substrate is primary. That clearly points to an SN2 reaction, where cyanide replaces bromine.
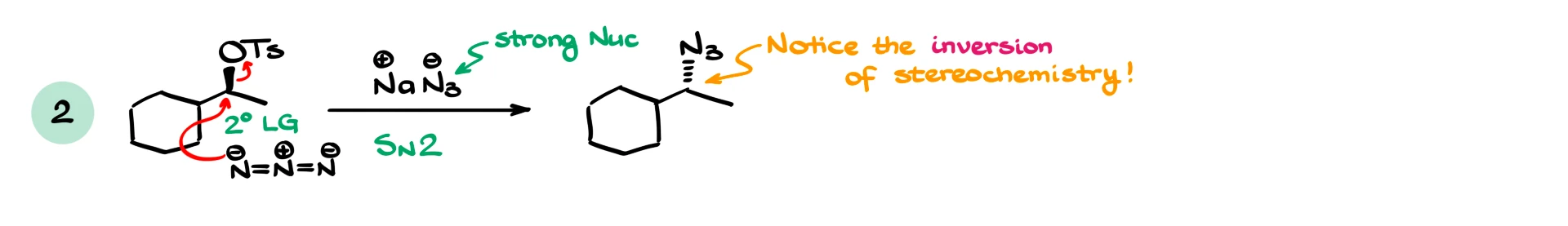
If we have a secondary substrate with a strong nucleophile like azide, we again expect SN2, and we must show inversion of stereochemistry. This is a very common test detail, so always pay attention to stereochemistry.
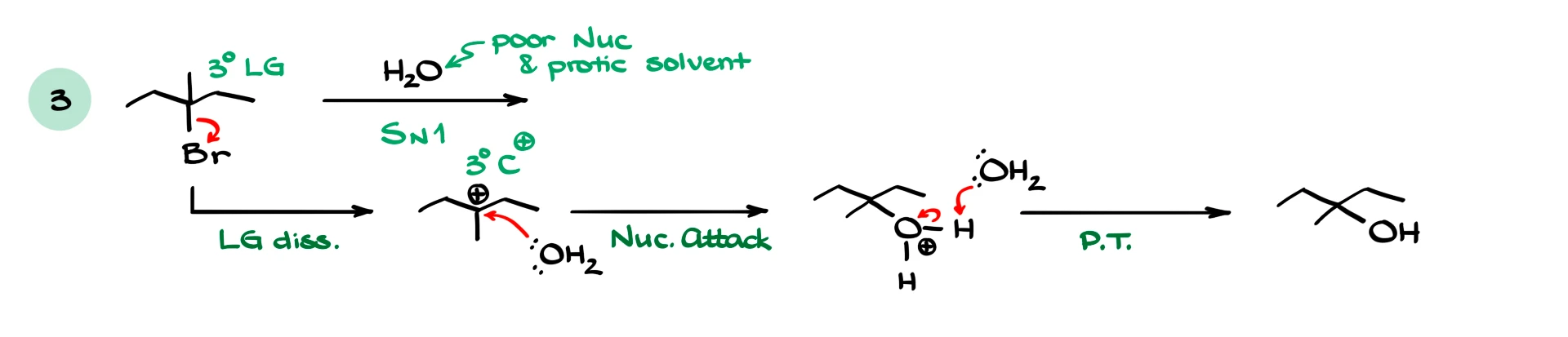
If we have a tertiary substrate and a weak nucleophile like water, we expect an SN1 reaction. The mechanism proceeds through carbocation formation, nucleophilic attack, and deprotonation to give the alcohol.
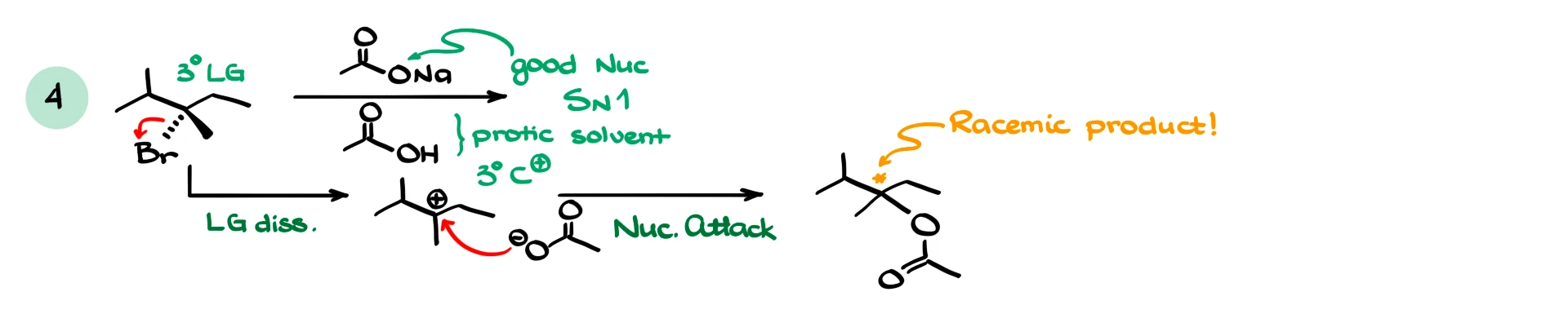
Now consider a slightly trickier case. Suppose we have a tertiary substrate and a good nucleophile, like acetate. Even though the nucleophile is strong, SN2 is essentially impossible because of steric hindrance at a tertiary carbon. So the reaction still proceeds via SN1, especially if we are in a polar protic environment.
In this case, we again form a carbocation, then the nucleophile attacks, giving the product. If the product is chiral, we typically get a racemic mixture, although you should always analyze the structure carefully rather than assuming that automatically.
Solvent Role
Up to this point, we have mostly ignored the solvent when making predictions. In many cases, that is fine because the substrate and nucleophile dominate. However, sometimes the solvent plays a critical role.

For example, consider a benzylic substrate like benzyl bromide. This is a primary carbon, so SN2 is favorable. But the carbocation that would form is also stabilized by resonance, so SN1 is also possible.
In this situation, the solvent determines the outcome. In a polar aprotic solvent, the reaction will most likely follow SN2. In a polar protic solvent, SN1 becomes more favorable.
These types of cases are less common, but they do show up as tricky exam questions. So whenever you see a substrate that is both unhindered and capable of forming a stabilized carbocation, be careful and consider the solvent.
So the key takeaway is this. Start with the nucleophile and substrate, then consider the solvent if needed. With practice, this approach will let you quickly and reliably distinguish between SN1 and SN2 reactions.
