Reformatsky Reaction
In this tutorial I want to talk about the Reformatsky reaction. While this is technically an organometallic reaction, in reality it has much more to do with enols and enolates than with classical organometallic chemistry.
Let’s take a closer look.
Zinc Enolates
The first step in the Reformatsky reaction is the formation of a zinc enolate. This species is generated when an α-halo ester reacts with zinc metal. When these two components come into contact, zinc inserts into the carbon–halogen bond through oxidative insertion, giving an organozinc compound where zinc is attached directly to carbon.
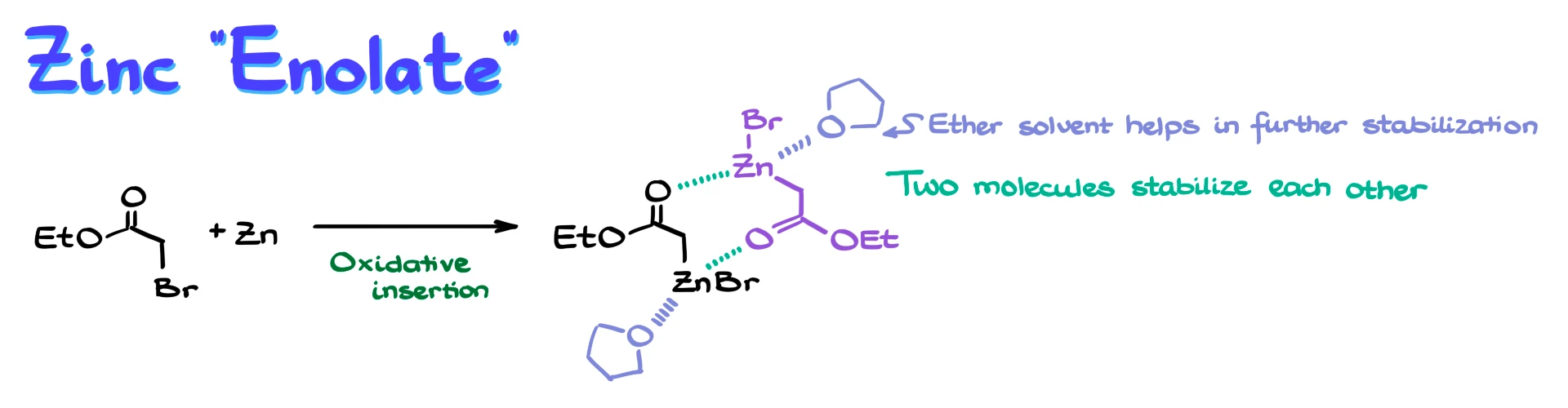
At this stage, we do not yet have a true enolate. However, in solution, another molecule of this organozinc compound can interact with it, forming a dimeric complex that helps stabilize the system. In addition, the reaction is typically carried out in a polar aprotic solvent, usually an ether such as THF, which also coordinates to zinc and stabilizes the overall structure.
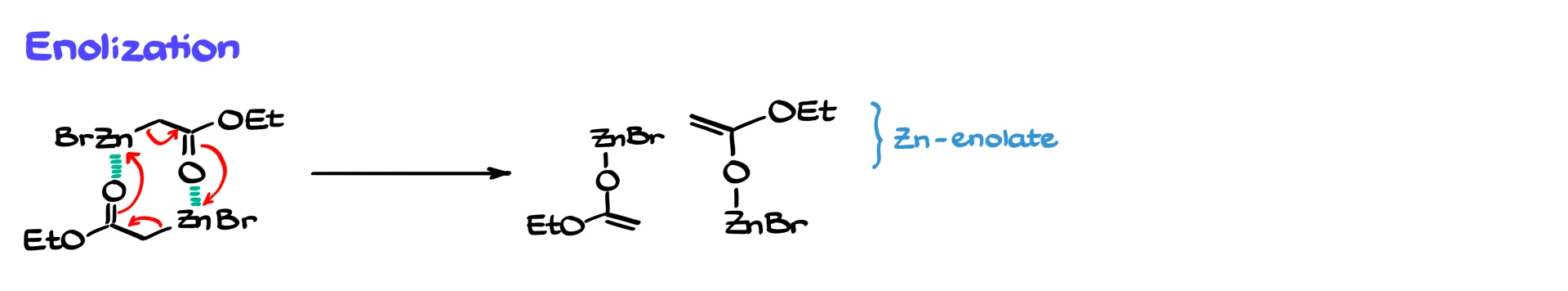
Now comes the key step. These two interacting molecules do not just sit next to each other, they react. Through a rearrangement involving electron flow, zinc shifts to the oxygen, and we form what is effectively an enolate, but with zinc as the counterion. These are therefore called zinc enolates.
Because zinc is now bound to oxygen rather than carbon, these species behave differently from typical organometallic reagents. They are less reactive, milder as nucleophiles, and significantly less basic than organolithium or Grignard reagents.
Reactions With Carbonyls
So what can these zinc enolates do?
Like any enolate, they are nucleophilic and can react with electrophiles. The most common electrophiles in this reaction are aldehydes and ketones.
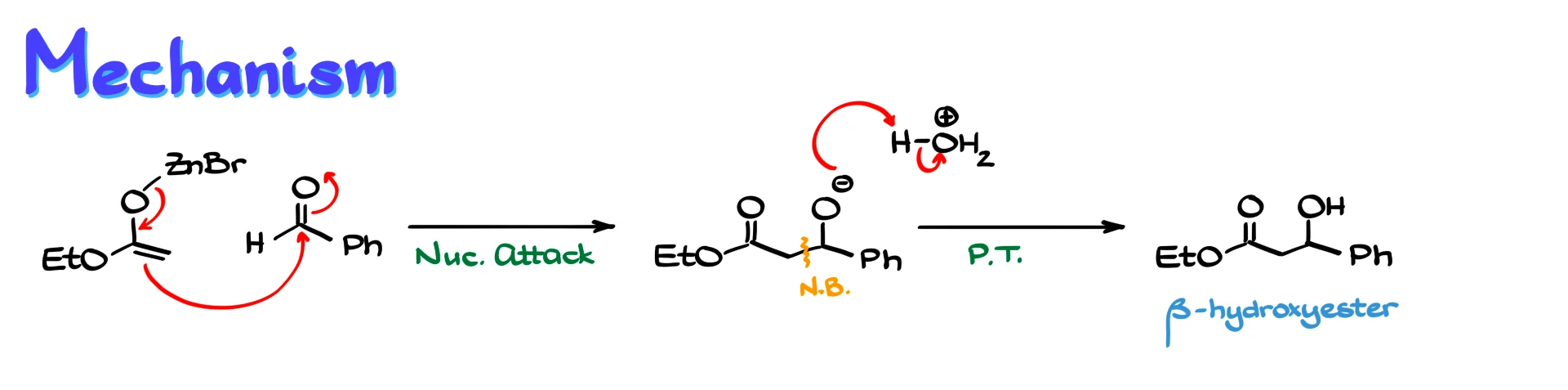
For example, if we react a zinc enolate with benzaldehyde, the enolate attacks the carbonyl carbon. The electrons from the carbonyl move onto oxygen, forming an alkoxide intermediate. This creates a new carbon–carbon bond. After an acidic workup, the alkoxide is protonated, giving a β-hydroxy ester as the final product.
One of the biggest advantages of the Reformatsky reaction is that we do not need to prepare the organometallic reagent in advance. We can mix the α-halo ester, the carbonyl compound, and zinc together, and the reaction proceeds in one pot. This works because zinc is relatively mild and does not react aggressively with aldehydes or ketones, unlike lithium or magnesium reagents.
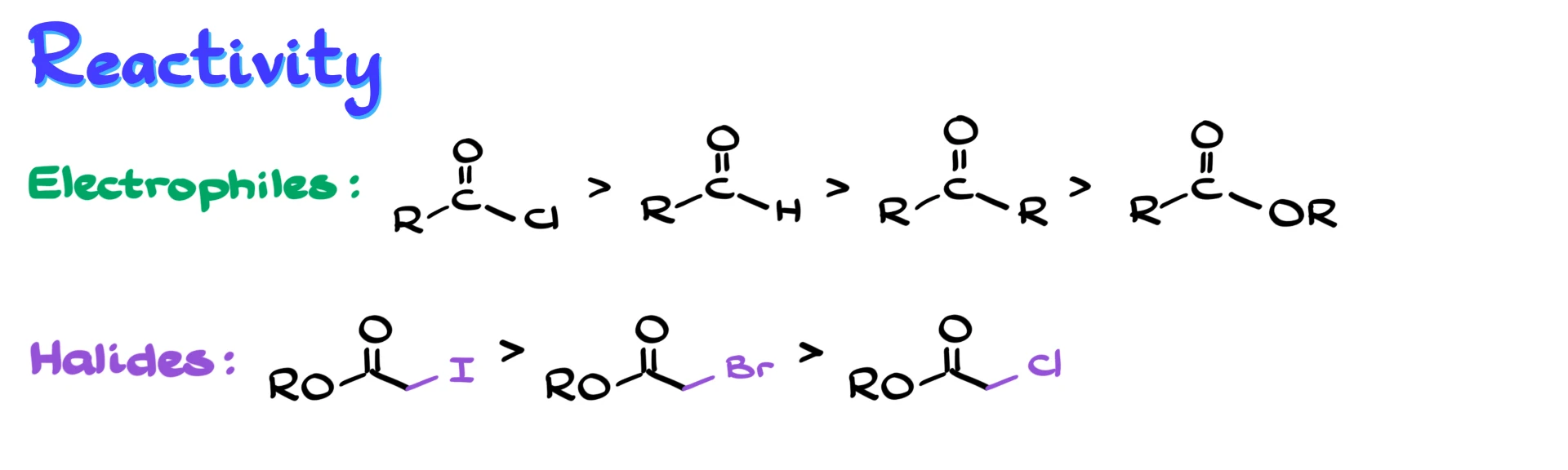
In terms of reactivity trends, nothing too surprising happens. Among electrophiles, acid chlorides are more reactive than aldehydes and ketones, which are more reactive than esters. For the halides, iodides are the most reactive toward zinc, while chlorides are the least reactive. In fact, many α-chloro esters are so unreactive that the reaction often does not work well. That is why α-bromo esters are the most commonly used substrates, they strike a good balance between reactivity and availability.
Examples of the Reformatsky Reaction
Let’s look at a few general scenarios.
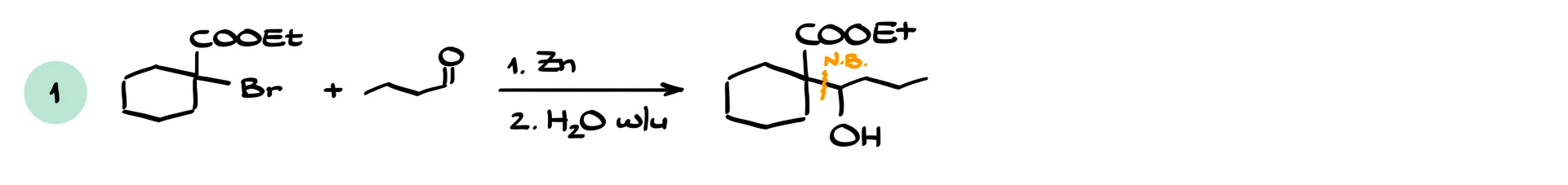
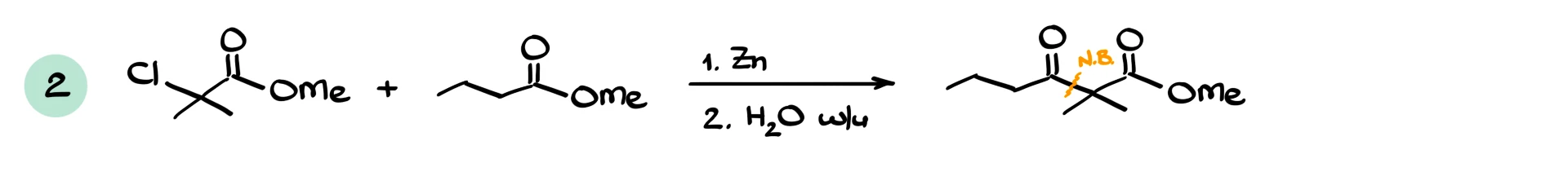
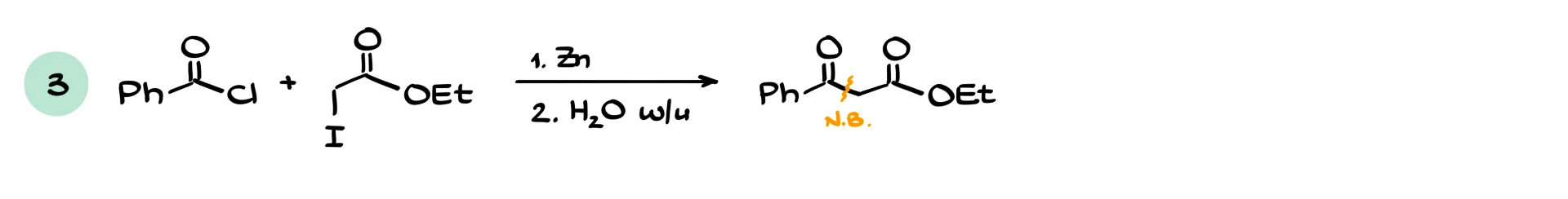
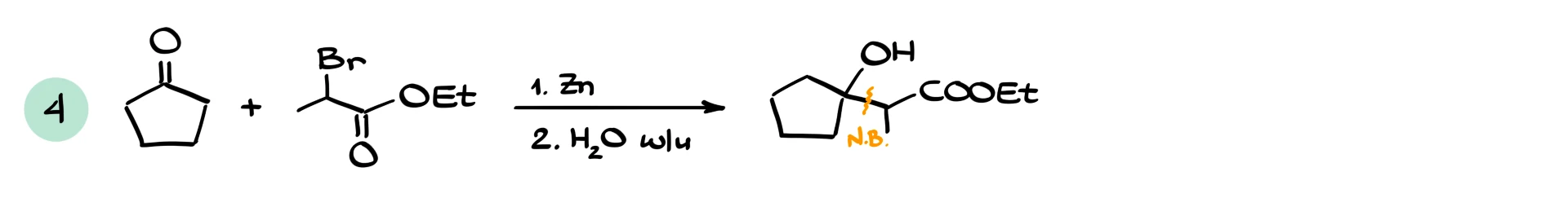
If we react an α-bromo ester with an aldehyde, we typically obtain a β-hydroxy ester with a new carbon–carbon bond. If we react with another ester, the reaction becomes more similar to a Claisen-type process, and side products are more likely. If we react with an acid chloride, the higher reactivity of the acid chloride usually ensures a smooth reaction. Reactions with ketones also work and give β-hydroxy esters.
Side Reactions in Reformatsky Reaction
However, there are several side reactions to be aware of.
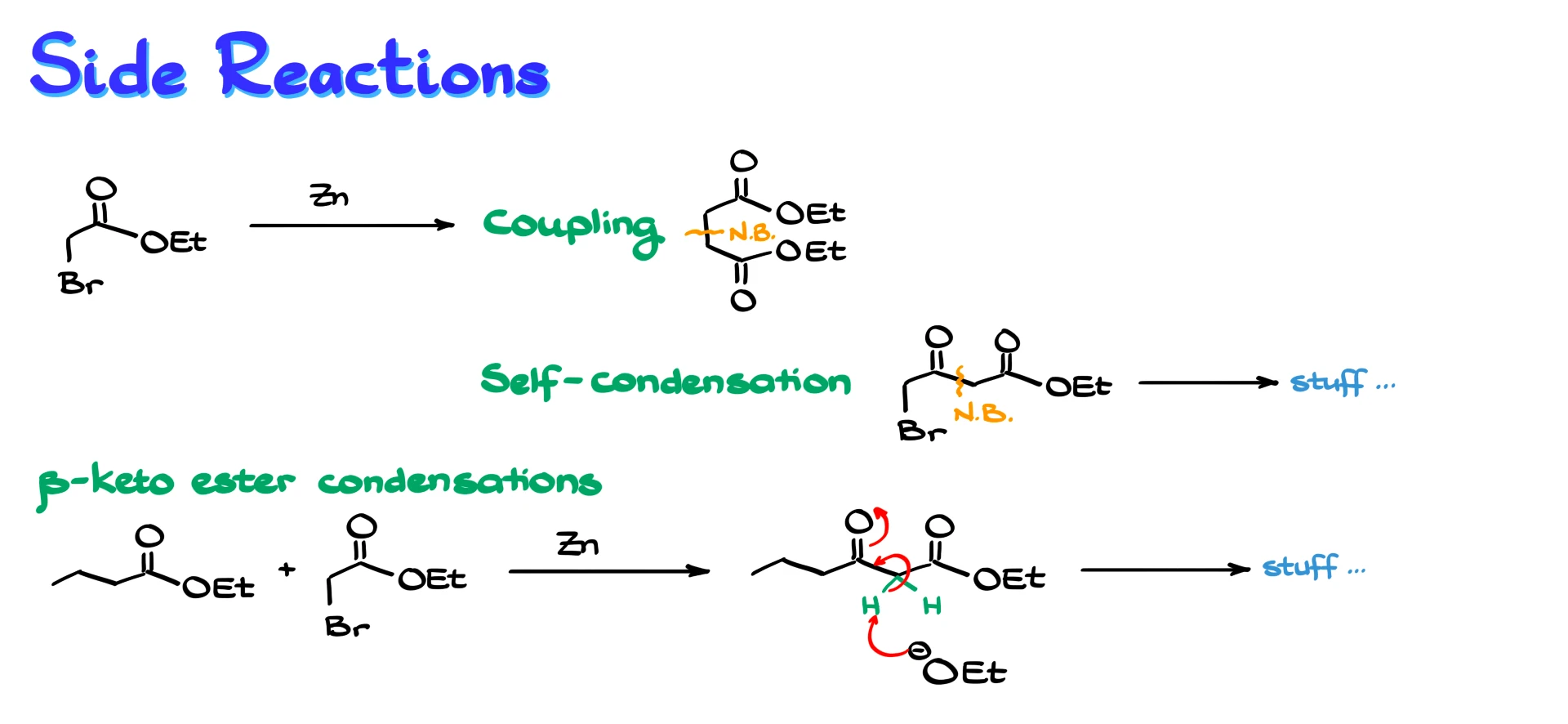
One common issue occurs early in the reaction. Before the organozinc species rearranges into the zinc enolate, it can react with another molecule of the starting α-halo ester, leading to coupling products instead of the desired reaction.
Another issue arises when less reactive electrophiles, such as esters, are involved. In this case, the zinc enolate can react with unreacted starting material, leading to self-condensation side products.
A third problem appears when forming β-keto esters. The hydrogen between the two carbonyl groups is quite acidic, and bases present in the reaction mixture, such as alkoxides, can deprotonate it. This generates additional enolates and leads to further side reactions.
There are ways to address these issues. For example, the use of palladium(0) catalysts has been shown to reduce side reactions in some cases.
Blaise Reaction
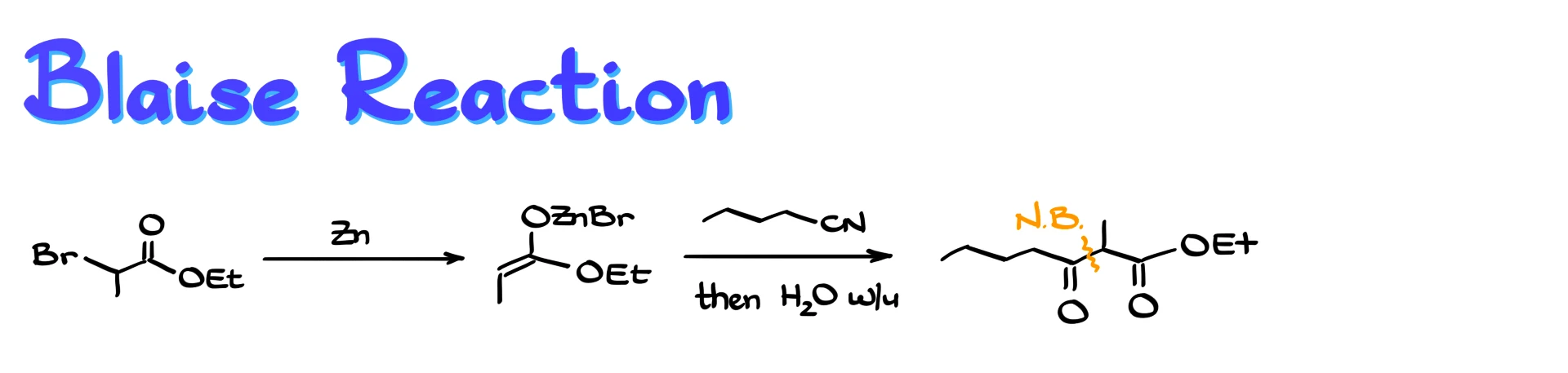
Another interesting workaround is the Blaise reaction, a variation of the Reformatsky reaction. In this approach, the zinc enolate is first formed, and then reacted with a nitrile instead of a carbonyl compound. Because nitriles are less reactive, this is typically done as a two-step process.
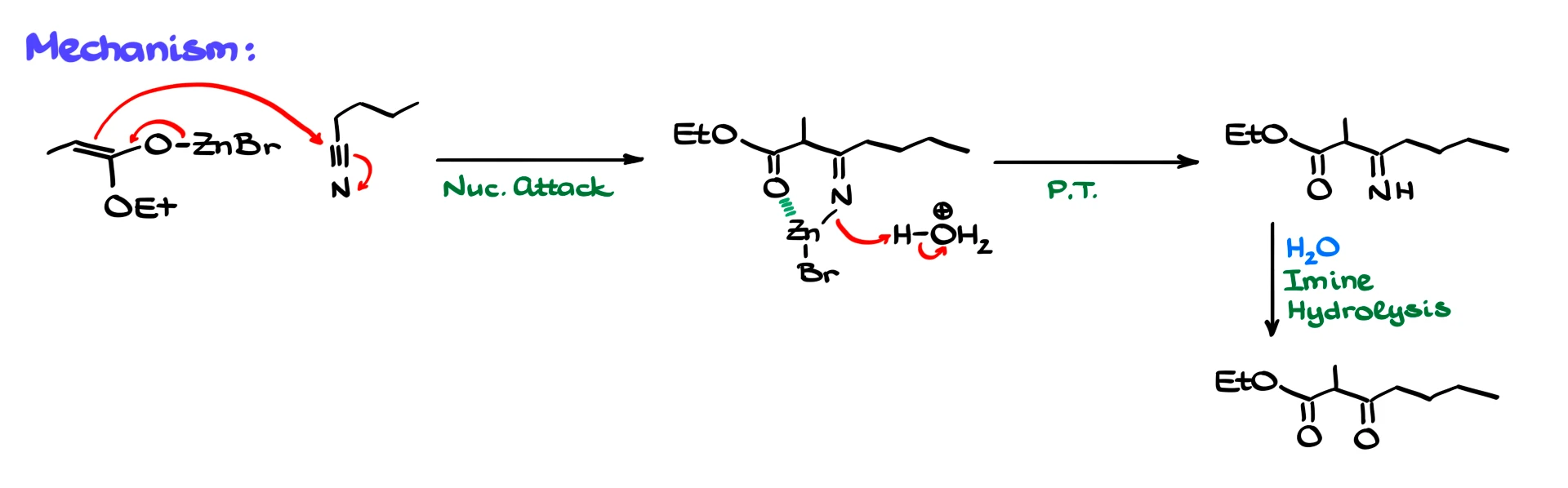
Mechanistically, the zinc enolate attacks the nitrile carbon, forming an intermediate in which zinc coordinates to both nitrogen and oxygen. After acidic workup, this intermediate is converted into an imine, which can then be hydrolyzed to give a dicarbonyl compound.
So while the Reformatsky reaction and its Blaise variation are not as universally useful as Grignard or organolithium chemistry, they are very valuable when you need a milder nucleophile and better functional group tolerance. The fact that the reaction can often be done in one pot without preforming the reagent is also a major practical advantage.
Because of this balance of reactivity and convenience, the Reformatsky reaction still appears in the literature from time to time, even though it is an older transformation with some limitations.
