E1 vs E2 Reactions
In this tutorial, I want to walk you through the differences between E1 and E2 reactions. Even though both are elimination reactions, they behave quite differently, so it is important to clearly understand how they compare.
E1 Reactions
Let’s start with the E1 reaction. In general, an E1 reaction happens in two steps. First, the leaving group departs, forming a carbocation intermediate. Then, a base, which does not need to be strong, removes a proton from a β-carbon, leading to the formation of an alkene. Essentially, anything with a lone pair can act as the base in this step.
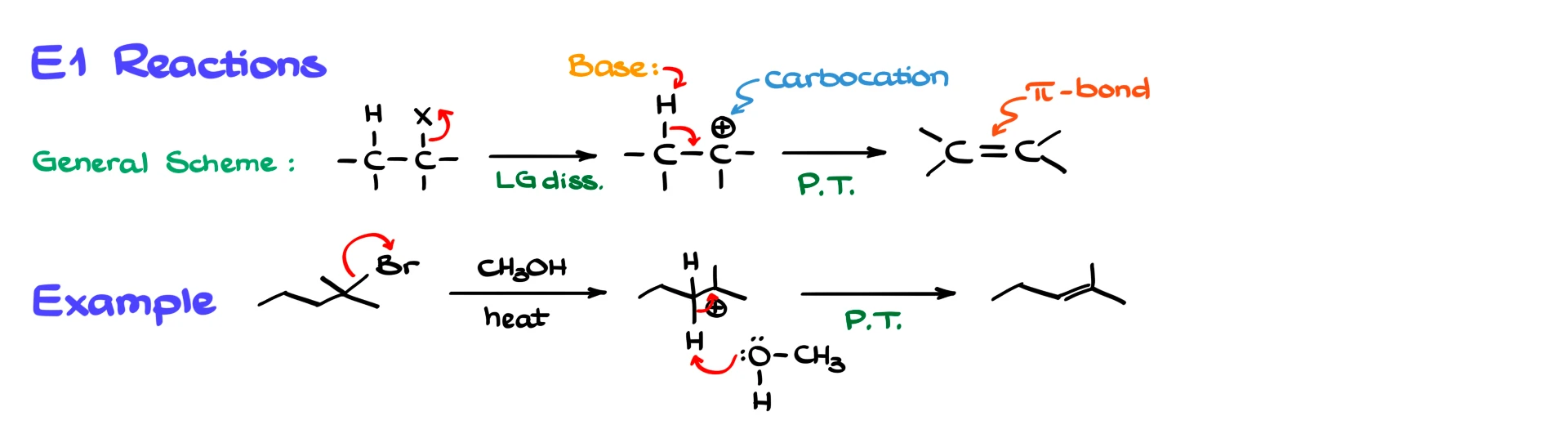
For example, if we begin with a tertiary bromide, the first step is the loss of the leaving group, which gives us a tertiary carbocation. From there, a molecule like methanol can act as the base. It comes in, removes a β-hydrogen, and we form the double bond, giving the alkene product.
E2 Reactions
Now compare that with the E2 reaction. E2 reactions occur in a single, concerted step, meaning everything happens at once and there are no intermediates. When the base removes a β-hydrogen, the electrons shift immediately to form the π bond while the leaving group leaves at the same time.
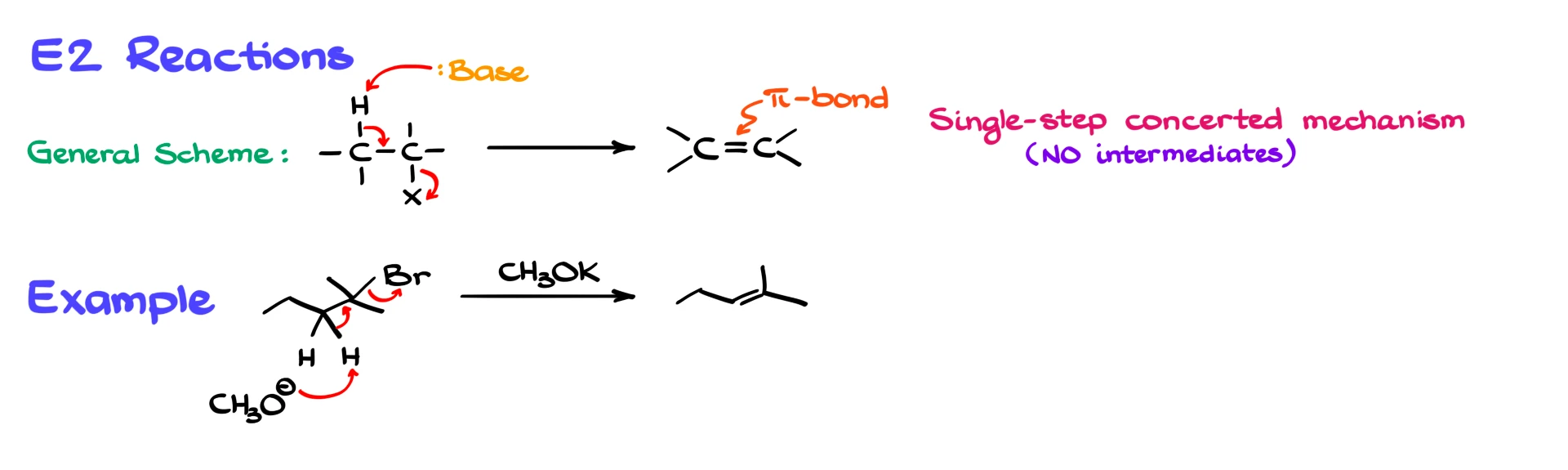
If we take the same tertiary bromide but now use a strong base such as potassium methoxide, the mechanism looks different. The methoxide ion directly abstracts a β-hydrogen, and in that same motion, the leaving group is expelled and the double bond forms instantly. There is no carbocation involved at any point.
Comparison of E1 and E2
When we compare E1 and E2 reactions more broadly, several key differences stand out. E1 reactions typically occur with tertiary substrates because they form stable carbocations. In contrast, E2 reactions can occur with primary, secondary, or tertiary substrates, as long as a strong base is present.

E1 reactions usually take place in protic solvents and do not require a strong base, often relying on the solvent itself. On the other hand, E2 reactions require a strong base and can occur in both protic and aprotic conditions.
Stereochemistry also differs. E1 reactions are not stereospecific and can produce multiple stereoisomers. E2 reactions, however, are stereospecific because the hydrogen and leaving group must be positioned anti-periplanar for the reaction to occur.
Regiochemistry of E1 and E2 Reactions
Now let’s talk about regiochemistry, which is where the double bond forms. In E1 reactions, you often get multiple possible products, but the major product is usually the more substituted alkene, known as the Zaitsev product.
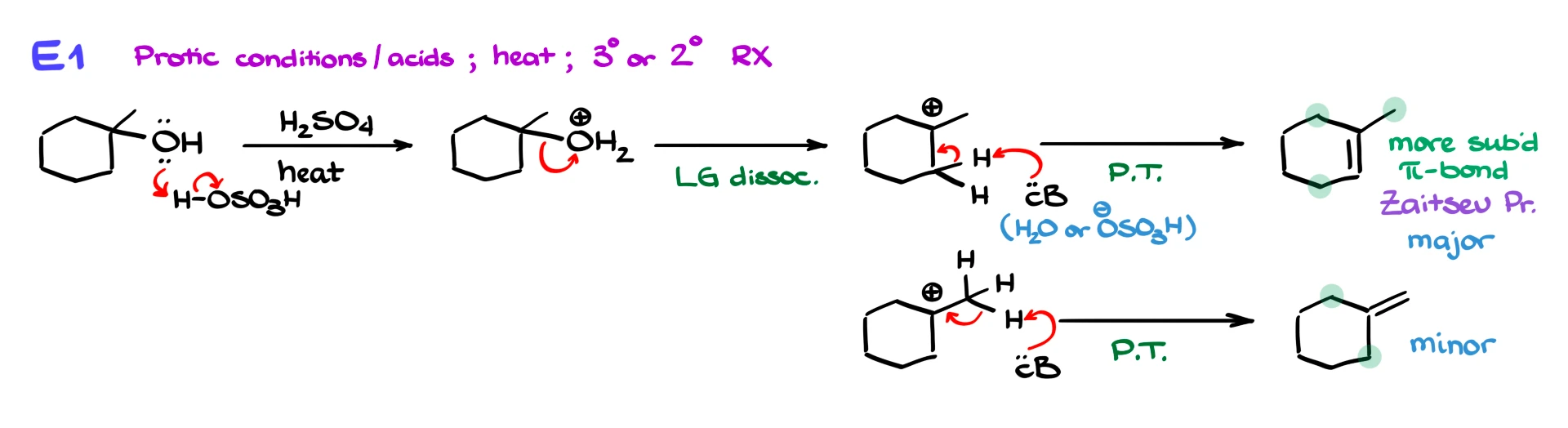
For example, if we take a tertiary alcohol like 1-methyl-1-cyclohexanol and treat it with sulfuric acid, the first step is protonation of the OH group, turning it into a better leaving group. Then water leaves, forming a tertiary carbocation. From there, a base removes a β-hydrogen to form an alkene.
This process can produce more than one alkene. One product has a more substituted double bond, while another has a less substituted one. The more substituted alkene is the major product because it is more stable. The less substituted alkene is the minor product.
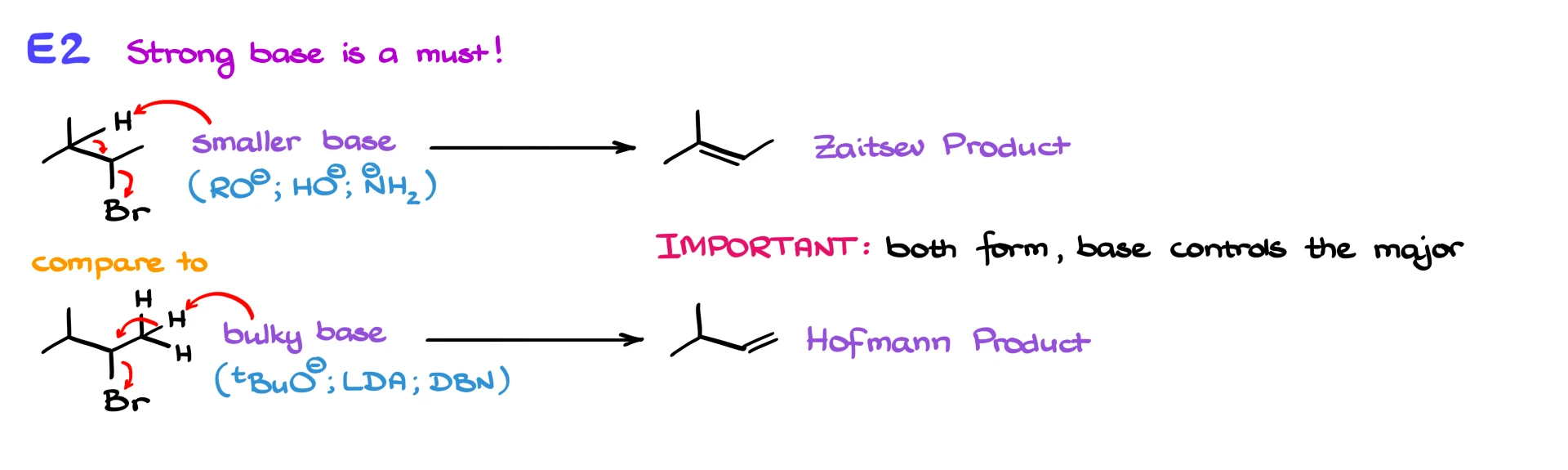
In E2 reactions, the situation depends heavily on the base. If you use a small, strong base like an alkoxide or hydroxide, it can easily access more substituted β-hydrogens, leading to the Zaitsev product. However, if you use a bulky base such as tert-butoxide, LDA, DBN, or DBU, it will preferentially remove a more accessible hydrogen, resulting in the less substituted alkene, known as the Hofmann product.
It is important to note that in both E1 and E2 reactions, multiple products can form unless something prevents one pathway. The key question is which product is major, and in E2 reactions, that is often controlled by the size and strength of the base.
Stereochemistry of E1 and E2 Reactions
Now let’s briefly touch on stereochemistry again. In E1 reactions, you generally get all possible stereoisomers, and the stereochemistry of the starting material does not matter. Even if the starting compound has multiple chiral centers, all stereoisomers can lead to the same set of products.
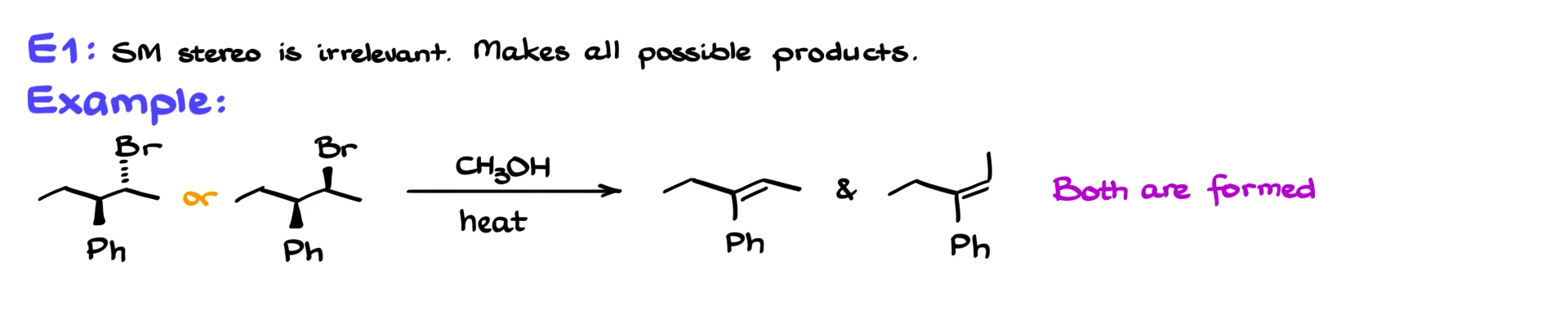
In contrast, E2 reactions are stereospecific. The hydrogen being removed and the leaving group must be anti-periplanar. Because of this requirement, the structure of the starting material directly determines which stereoisomer of the alkene can form. In some cases, only one stereoisomer, either E or Z, is produced.
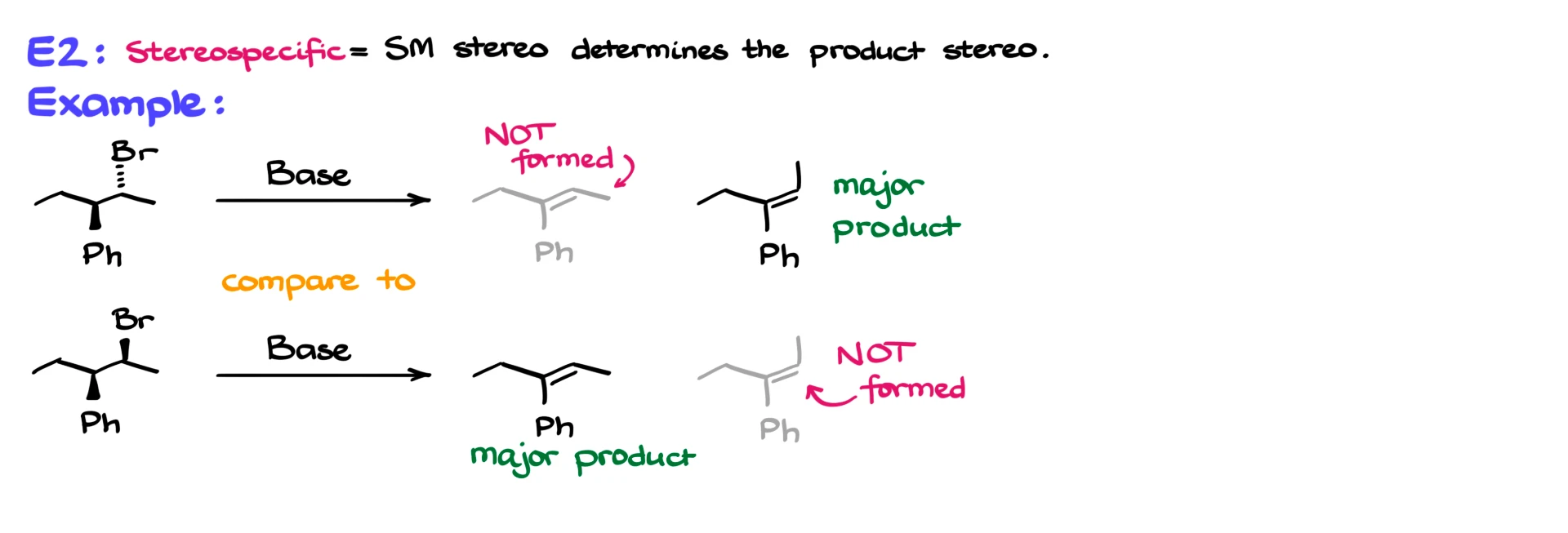
To wrap things up, E1 reactions proceed through a carbocation intermediate, can undergo rearrangements, favor more substituted alkenes, and typically occur in protic conditions. E2 reactions are concerted, require a strong base, have no intermediates, and their regioselectivity is often controlled by the base. They are also stereospecific, meaning the starting structure dictates the final stereochemistry.
Even though both are elimination reactions, their mechanisms and outcomes are quite different, so make sure you pay close attention to these distinctions when solving problems.
