SN2 vs E2 Reactions
In this tutorial, I want to walk you through the comparison between SN2 and E2 reactions. This is one of the easier comparisons, but there are still a few important details you need to keep in mind to reliably predict the outcome.
SN2 Reactions
Let’s start with the SN2 reaction. SN2 follows a concerted mechanism, meaning it happens in a single step. The nucleophile attacks the carbon at the same time that the leaving group leaves, so we get the product immediately with no intermediates.
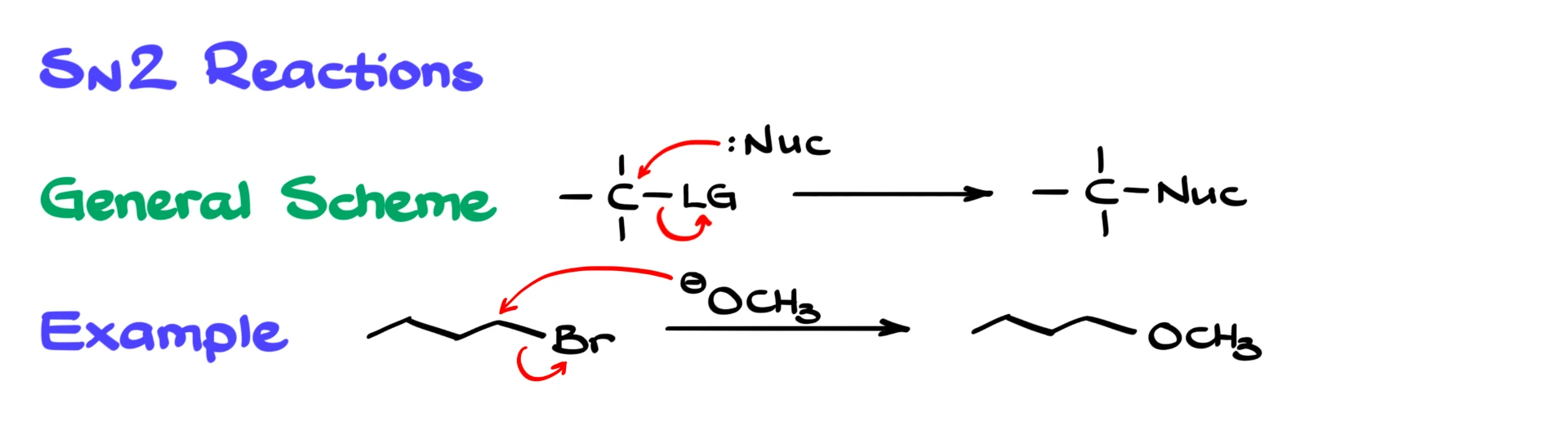
For example, if we take 1-bromobutane and react it with methoxide, the methoxide ion acts as a nucleophile. It attacks the carbon bonded to the bromine, displacing the bromine in one step, and we form the substitution product right away. It is a straightforward backside attack leading directly to the product.
E2 Reactions
Now let’s look at the E2 reaction. E2 is also a concerted process, so again there are no intermediates. However, instead of attacking the carbon, the reagent acts as a base and removes a β-hydrogen. As that proton is removed, the electrons shift to form a double bond while the leaving group departs at the same time.
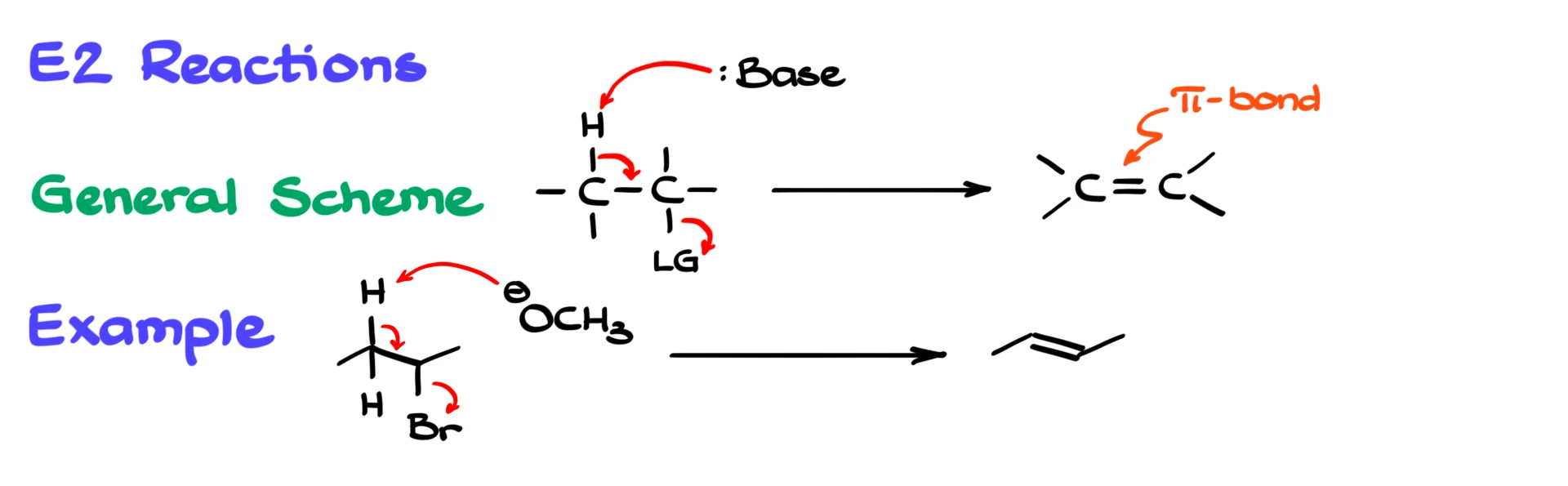
For instance, if we take 2-bromobutane and react it with methoxide, the methoxide removes a β-hydrogen. At the same time, the leaving group is expelled, and we form an alkene, specifically 2-butene.
Comparison of SN2 and E2
When comparing these two reactions, SN2 reactions typically occur with primary or secondary alkyl halides. They require a good nucleophile and are stereospecific, giving inversion of configuration at the reactive center.
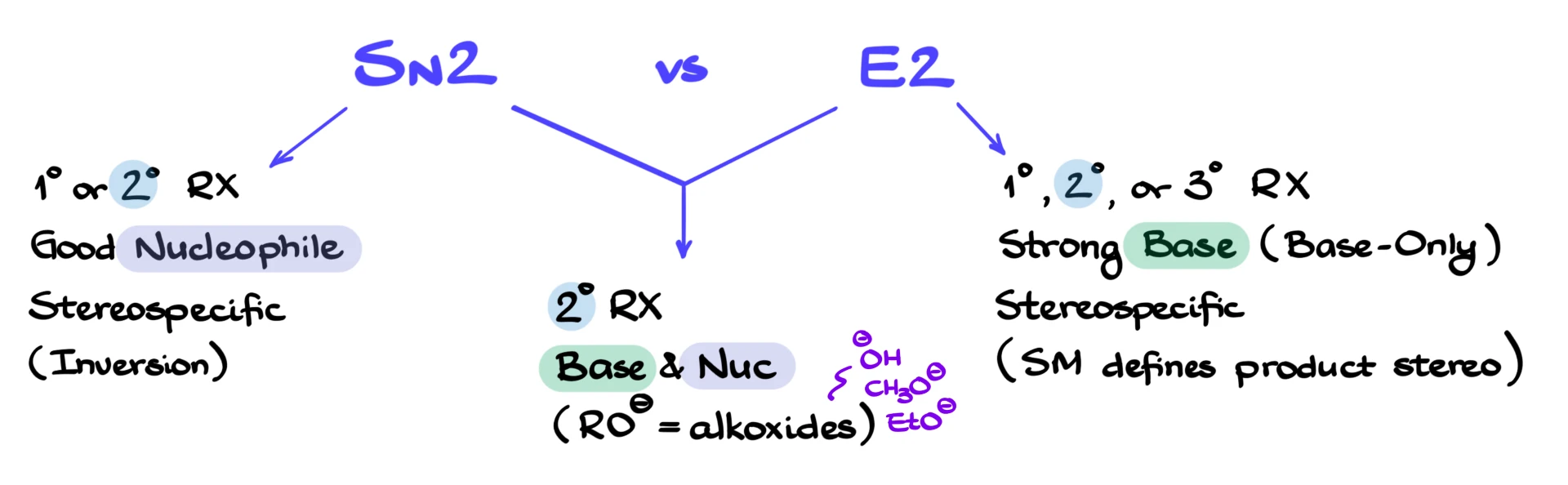
E2 reactions, on the other hand, can occur with primary, secondary, or tertiary substrates. They require a strong base, often described as a base-dominant reagent, and they are also stereospecific because the geometry of the starting material determines the product.
The key point where SN2 and E2 compete is with secondary alkyl halides. This is especially true when the reagent can act as both a nucleophile and a base, which is common for small alkoxides such as hydroxide, methoxide, or ethoxide. These reagents are strong enough to promote elimination but also good enough nucleophiles to give substitution.

A useful rule of thumb helps predict the outcome. With primary alkyl halides, SN2 is usually the major pathway, although a small amount of E2 product may form. With secondary alkyl halides, both SN2 and E2 products are typically formed, but E2 is usually the major pathway. With tertiary alkyl halides, SN2 is essentially blocked due to steric hindrance, so E2 becomes the dominant, and often the only, reaction.
Examples
For example, if you react a primary alkyl halide with sodium ethoxide, you will get both substitution and elimination products, but the SN2 product will be the major one. If you instead use a secondary alkyl halide, you will again get both types of products, but now the elimination products will dominate. Finally, if you start with a tertiary alkyl halide, the reaction will proceed almost exclusively through E2, giving only alkene products.
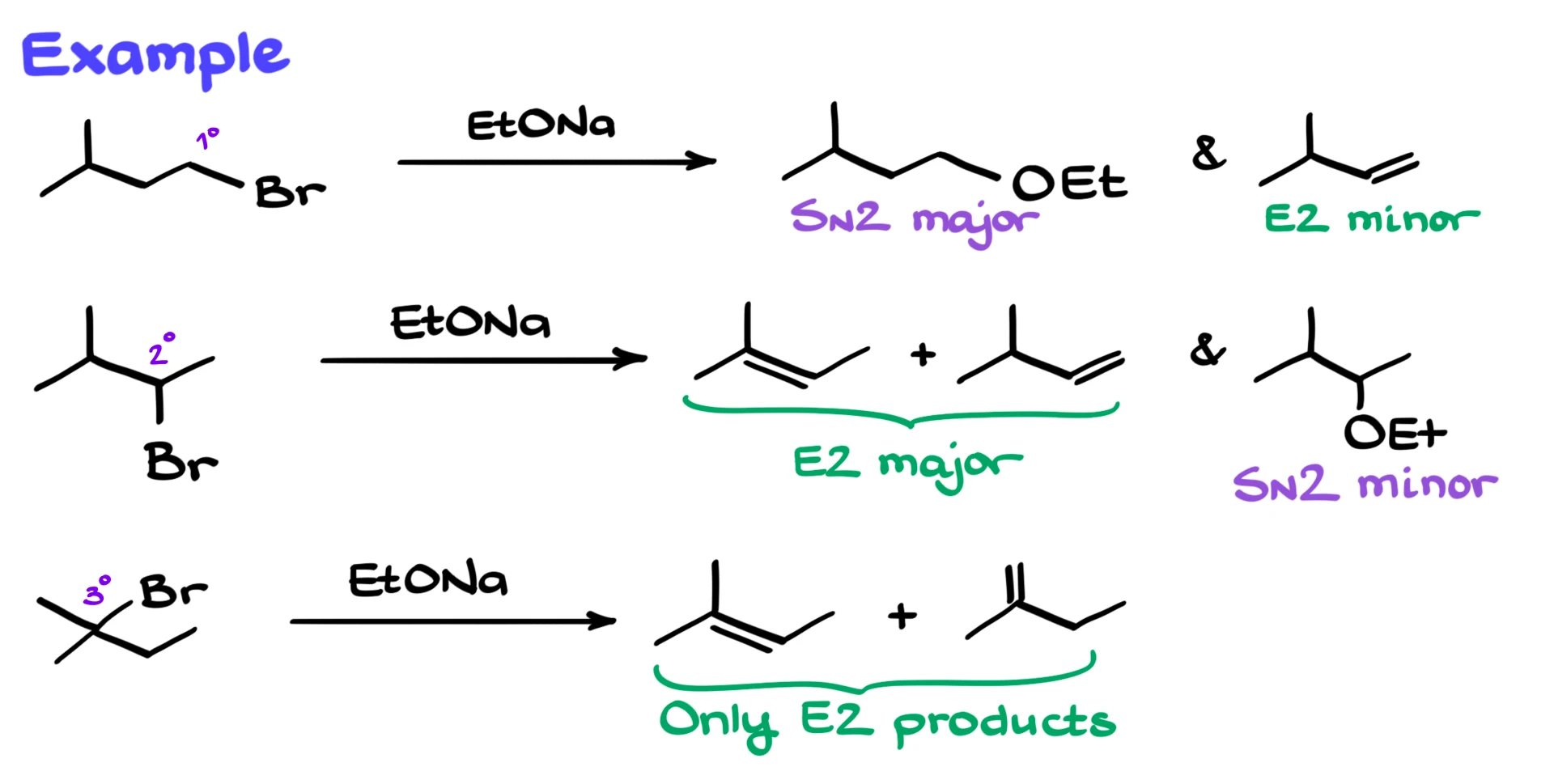
So the main takeaway is that SN2 and E2 compete primarily with secondary substrates and reagents that can act as both bases and nucleophiles. In those cases, elimination usually has the edge.
That is why this comparison is often considered one of the simplest. Once you recognize the substrate and the type of reagent, predicting the outcome becomes much more straightforward.
