SN1 vs E1 Reactions
In this tutorial I want to compare SN1 and E1 reactions and show you how experimental conditions determine which pathway is favored.
Mechanistic Differences
Let’s start with a classic example: tert-butyl bromide reacting with water.
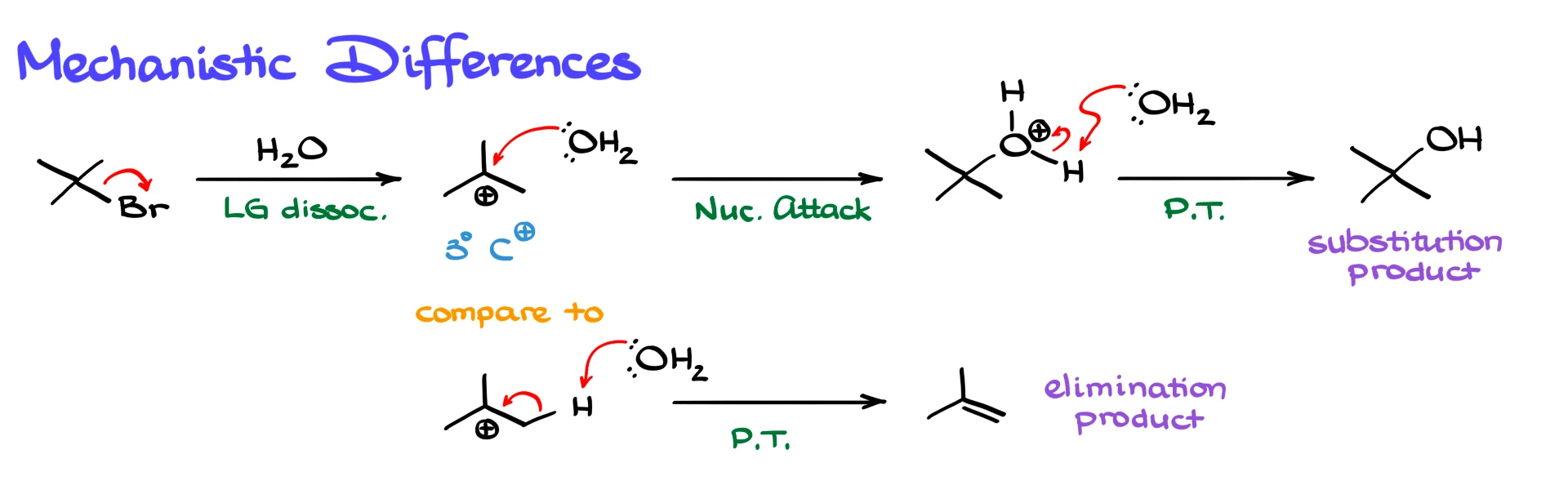
The first step in both SN1 and E1 is the same, leaving group dissociation, which gives a tertiary carbocation. From here, the reaction can go in two different directions.
In the SN1 pathway, water acts as a nucleophile. It attacks the carbocation to form a protonated alcohol, and then another molecule of water removes the proton to give the neutral alcohol product.
In the E1 pathway, water acts as a base instead. It removes a proton from a carbon adjacent to the carbocation, forming a double bond and giving the alkene as the elimination product.
So the key question is, which pathway is preferred?
The answer largely comes down to temperature.
Temperature Effect
To understand why, let’s compare the thermodynamics of the two processes.
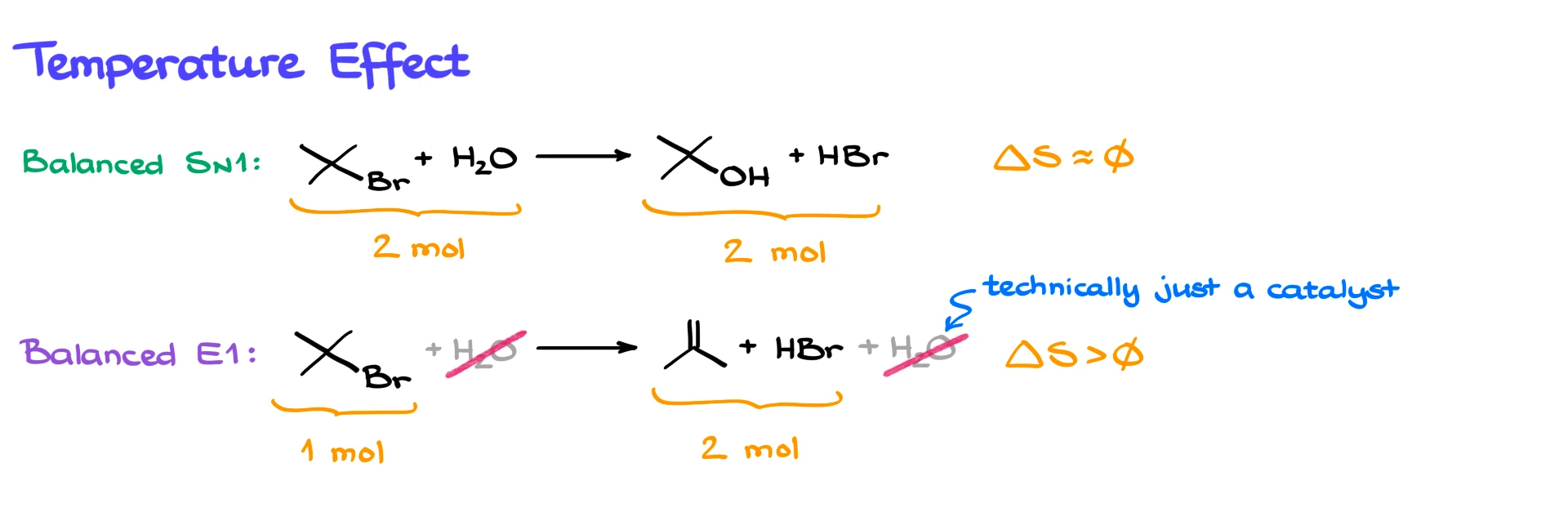
In the SN1 reaction, we start with two molecules, the substrate and water, and we end up with two molecules, the alcohol and the leaving group. That means the change in entropy, ΔS, is roughly zero.
In the E1 reaction, water acts as a catalyst and is not consumed overall. So effectively, we go from one molecule to two molecules. This means ΔS is positive, since we are increasing the number of particles.
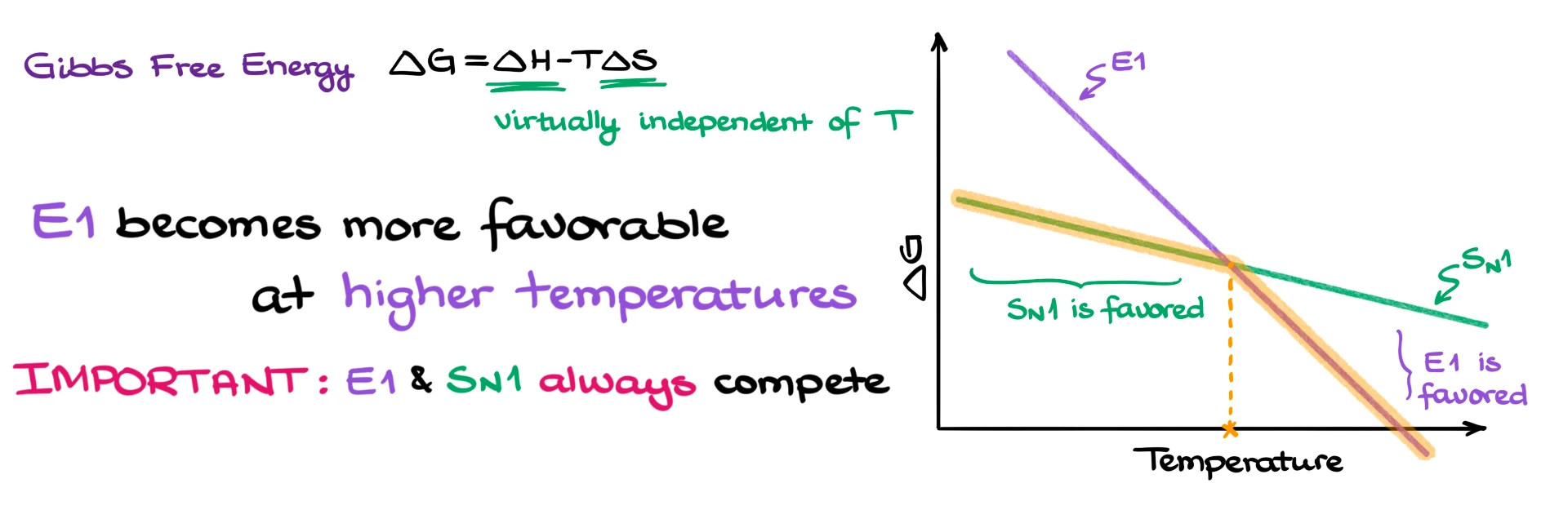
Now, if we think about the Gibbs free energy equation, ΔG = ΔH − TΔS, we can see how temperature plays a role. Since E1 has a larger positive ΔS, the term TΔS becomes more significant as temperature increases, making ΔG more negative.
So at higher temperatures, the E1 pathway becomes more favorable than SN1.
The key takeaway is simple:
E1 is favored at higher temperatures, while SN1 is favored at lower temperatures.
However, there is one very important point. SN1 and E1 are always in competition with each other. It is practically impossible to have only one pathway. If SN1 is happening, E1 is also occurring to some extent, and vice versa. The real question is which pathway gives the major product.
Examples
Let’s go through an example.
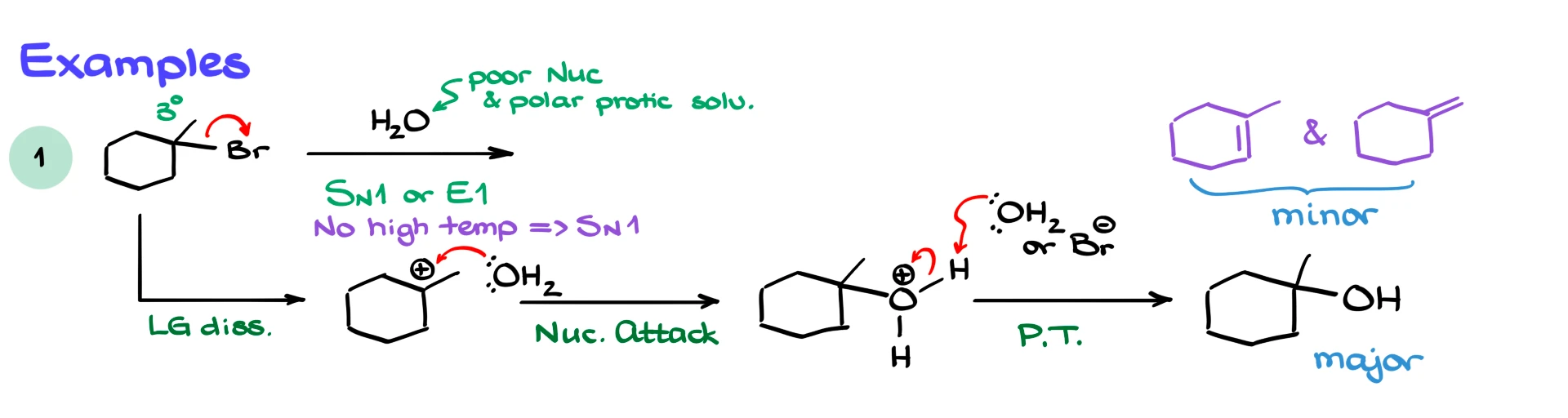
Suppose we react 1-bromo-1-methylcyclohexane with water. Water is a weak nucleophile and a polar protic solvent, and the substrate is tertiary. These conditions clearly favor SN1 and E1 mechanisms.
Since there is no heat indicated, SN1 is favored. The reaction proceeds through carbocation formation, followed by nucleophilic attack by water and deprotonation to give a tertiary alcohol as the major product.
That said, a small amount of elimination product will still form.
Now let’s look at another example.
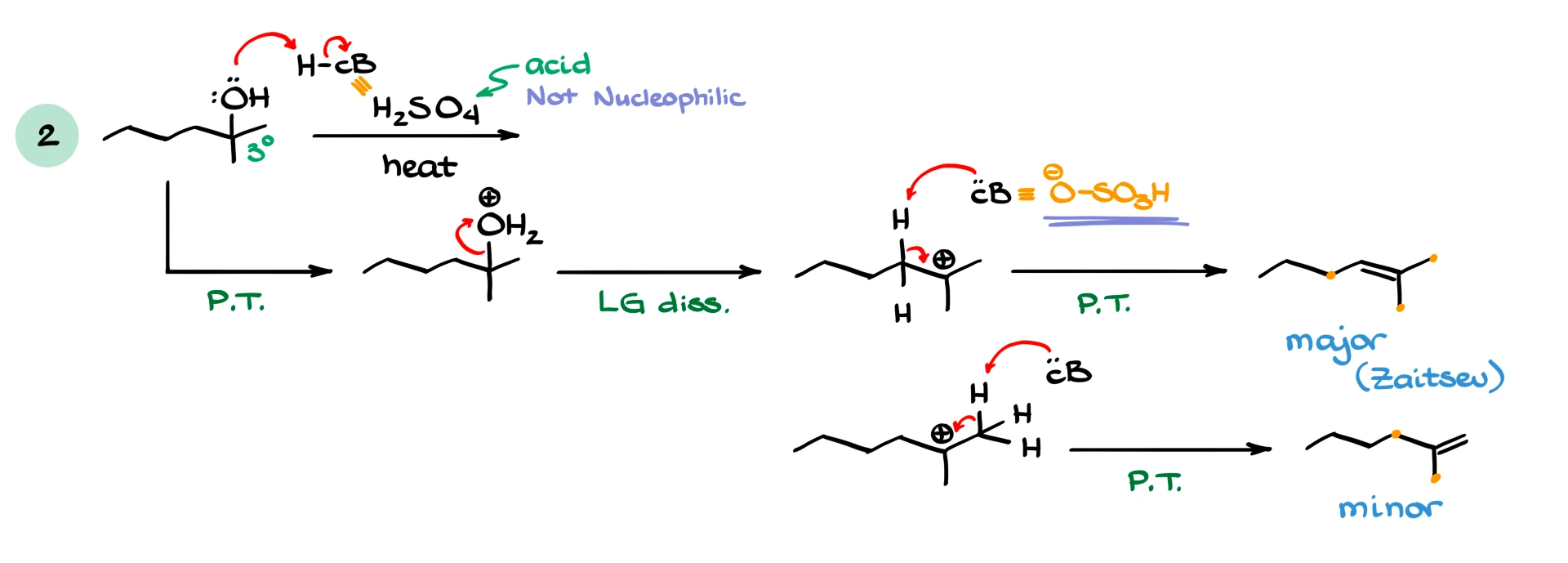
Suppose we have a tertiary alcohol reacting with sulfuric acid under heat. First, the OH group is protonated to turn it into a good leaving group. Then water leaves, forming a tertiary carbocation.
From here, a base removes a proton from a neighboring carbon, forming an alkene. Depending on which proton is removed, we can get different alkene products.
According to Zaitsev’s rule, the more substituted alkene is more stable, so it will be the major product.
Now here is an interesting twist. In this reaction, sulfuric acid and its conjugate base are poor nucleophiles, essentially non-nucleophilic. That means that even though SN1 is theoretically possible, it is very unlikely. So in this case, we observe almost exclusively E1.
If we used a different acid, such as HBr, where the conjugate base is a good nucleophile, then substitution would become a viable pathway again.
So this type of reaction, dehydration of alcohols with strong non-nucleophilic acids, is somewhat of an exception where elimination dominates almost completely.
Concluding Thoughts
To wrap everything up, here are the key ideas.
SN1 and E1 share the same first step, carbocation formation. After that, the reaction either proceeds through nucleophilic attack for substitution or proton loss for elimination.
Temperature plays a major role. Higher temperatures favor elimination because of the positive entropy change.
And finally, these pathways almost always compete, so you are usually asked to identify the major product, not every possible product.
