Partial Reduction of Acid Derivatives to Aldehydes
When we talk about aldehyde synthesis, we usually think about oxidation of primary alcohols. But that is not always the most convenient method. So in this tutorial I want to talk about the opposite approach to making aldehydes, reducing carboxylic acid derivatives.
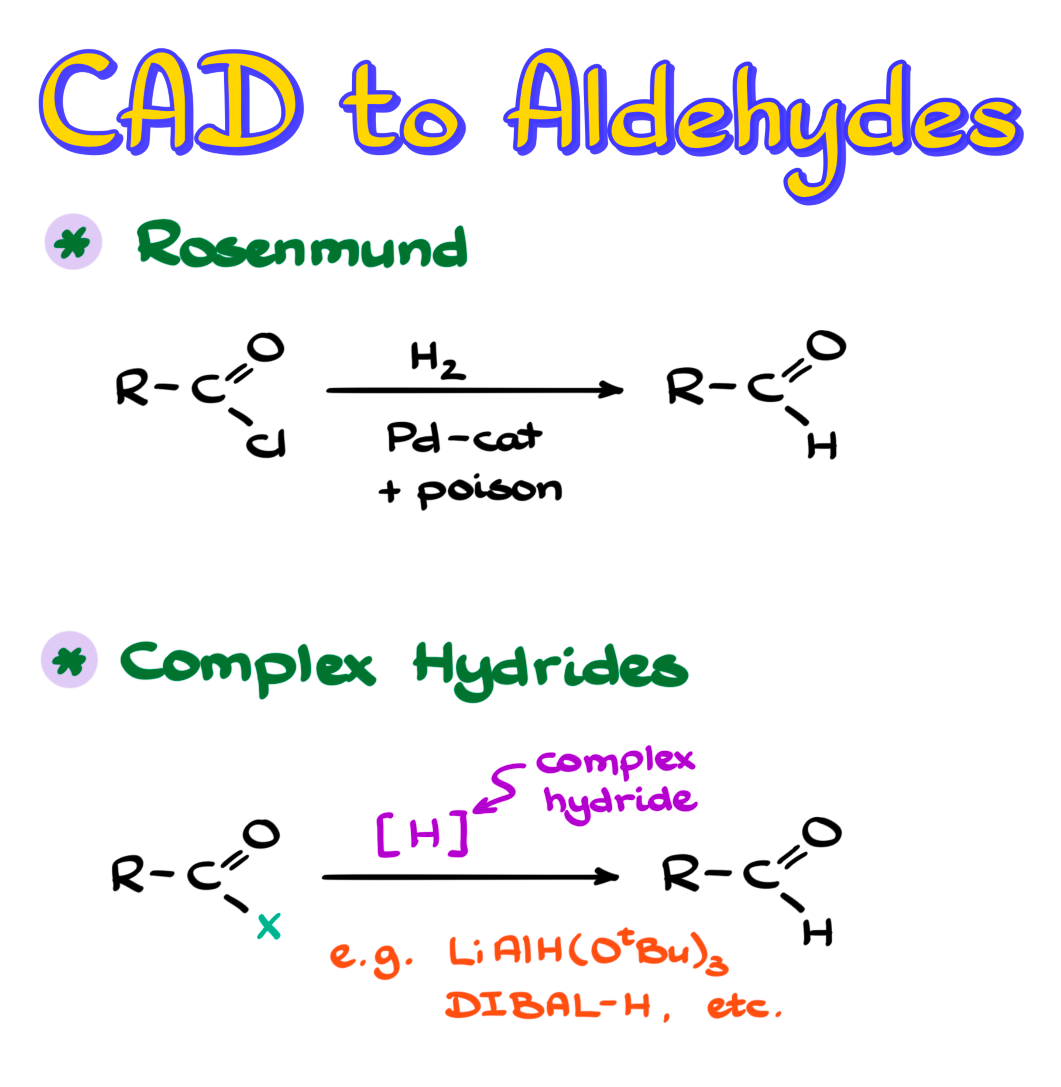
We are going to look at classic methods such as the Rosenmund reduction, and we are also going to talk about reduction of carboxylic acid derivatives with different complex hydrides.
Rosenmund Reduction
Let’s start with the Rosenmund reduction.
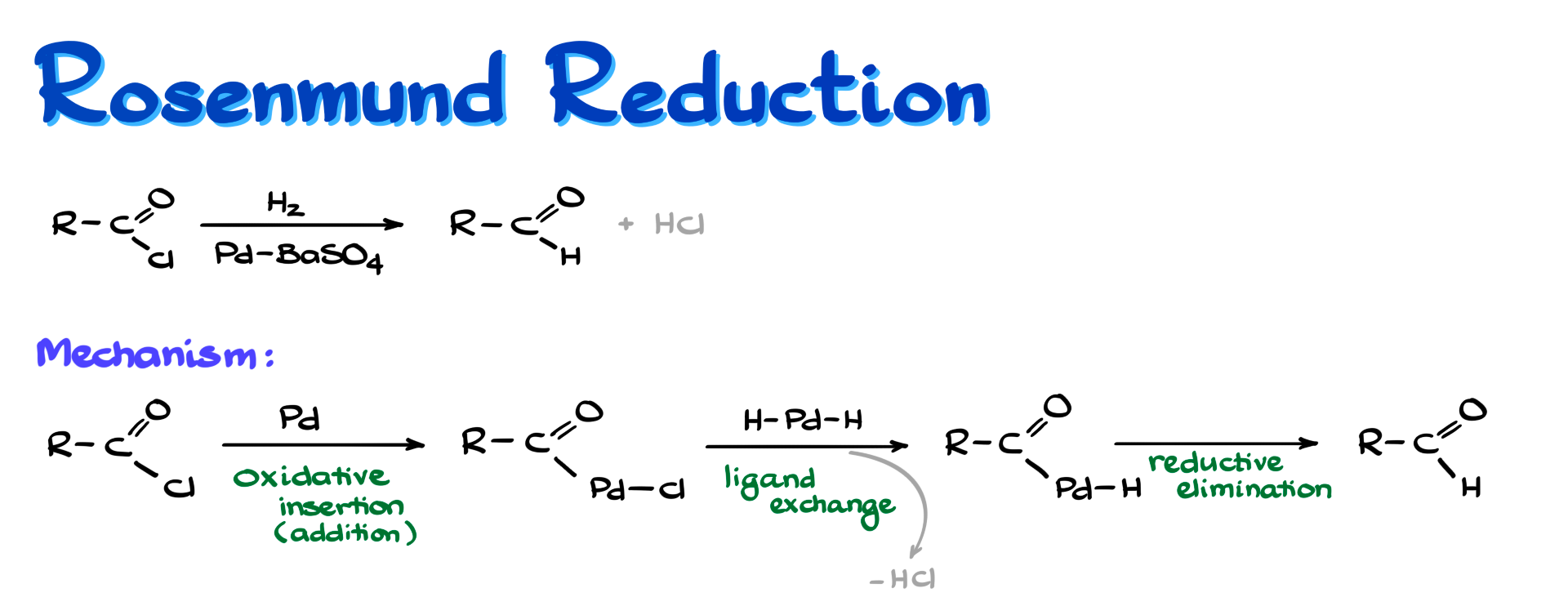
In this reaction we start with an acid chloride and reduce it with H₂ over a poisoned catalyst, typically palladium on barium sulfate, in a system somewhat similar to Lindlar’s catalyst. There are a few variations, so it is not always exactly Lindlar’s catalyst, but the idea is similar. As a result, we end up with our aldehyde, and HCl is formed as a co-product.
From the mechanistic standpoint, this reaction begins with palladium(0) inserting between the carbon and chlorine. As a result of this oxidative insertion, we form a complex in which palladium is attached to carbon, while chlorine is essentially acting as a counterion, if you want to think about it that way.
Next we bring in hydrogen that has been adsorbed on palladium, and we do a ligand exchange where chlorine is replaced by one of those hydrogens. As a result we lose HCl, and now palladium is directly connected to both hydrogen and carbon.
From this point we have a reductive elimination step. Palladium leaves the complex, and we end up with our aldehyde.
While Rosenmund reduction looks good on paper, there are unfortunately some very significant problems with this reaction.
- First of all, it is extremely hard to control and reproduce. The biggest problem is the catalyst itself and the quality of that catalyst. It is very difficult to prepare the exact same catalyst twice, and because of that the results can vary quite a bit.
- Also, since this reaction uses H₂ and a heterogeneous catalyst, the chemoselectivity is often poor. In addition to acid chlorides, we can also reduce double bonds and triple bonds, which can really mess up the molecule. So whenever you use this reaction, you need to make sure that nothing else in the structure can be affected.
Because these limitations are fairly serious, the Rosenmund reduction is used much more often in industry than in the lab. In the lab, we usually prefer other methods.
Reduction with Lithium tri-tert-Butoxyaluminum Hydride
One alternative is to use a complex hydride as the reducing agent to convert an acid chloride into the corresponding aldehyde.
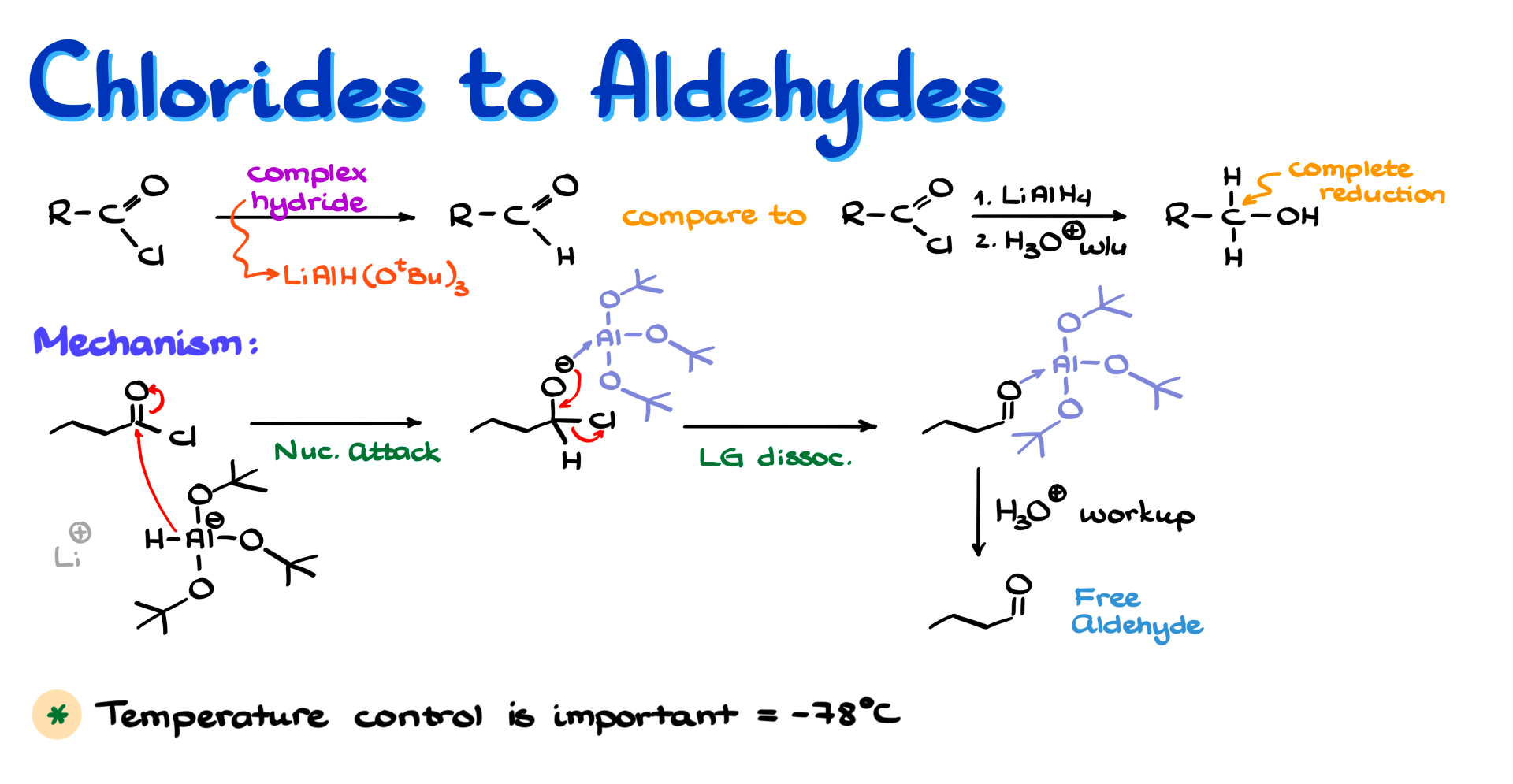
Since both acid chlorides and aldehydes are highly electrophilic, we need a hydride source that is reactive enough to attack the acid chloride, but not reactive enough to continue on to the aldehyde.
Typically the most common reagent here is lithium tri-tert-butoxyaluminum hydride, which is much less reactive than typical aluminum hydrides such as lithium aluminum hydride. If we were to use regular lithium aluminum hydride, the reaction would not stop at the aldehyde. It would keep going until we ended up with the primary alcohol, which is obviously not what we want.
Let’s look at the mechanism of this reaction. For these examples I am going to use butanoyl chloride as my acid chloride.
To show the first step, I will also draw the structure of lithium tri-tert-butoxyaluminum hydride. Naturally we also have a lithium counterion, but we do not care too much about that part.
Like we would normally expect in reactions of complex hydrides with carbonyl compounds, the first step is nucleophilic attack by hydride on the carbonyl carbon. That gives us the corresponding tetrahedral intermediate. One important thing to keep in mind here is that aluminum coordinates to the negatively charged oxygen and pulls some of that negative charge onto itself.
Even so, the chlorine atom is still an excellent leaving group, which means the oxygen can easily kick it out. Importantly, aluminum still remains coordinated to oxygen. As long as we keep the temperature low, and typically we are talking about −78°C, aluminum stays attached to that oxygen and creates a great deal of steric hindrance. That makes the next round of reduction, which would otherwise be possible, much less likely simply because this bulky aluminum species blocks further attack.
From here we do a typical acidic workup to free the aldehyde from aluminum. The aluminum ends up as Al³⁺ salts, we wash those away in the aqueous layer, and we do not think about them again.
As I said, the important feature of this reaction is temperature control. Typically the reaction is run at −78°C, although the literature does report examples at somewhat higher temperatures and even at room temperature. Still, the general rule is that lower temperatures are better, because they help maintain the complex in which aluminum remains attached to oxygen rather than allowing everything to fall apart.
Another obvious problem with this method is that acid chlorides themselves are extremely electrophilic and are not especially tolerant of many other functional groups. That can be an issue if you are working with a larger molecule containing several functional groups.
Reduction with DIBAL-H
So another option is to start with an ester, which is significantly less electrophilic, and reduce it to the corresponding aldehyde.
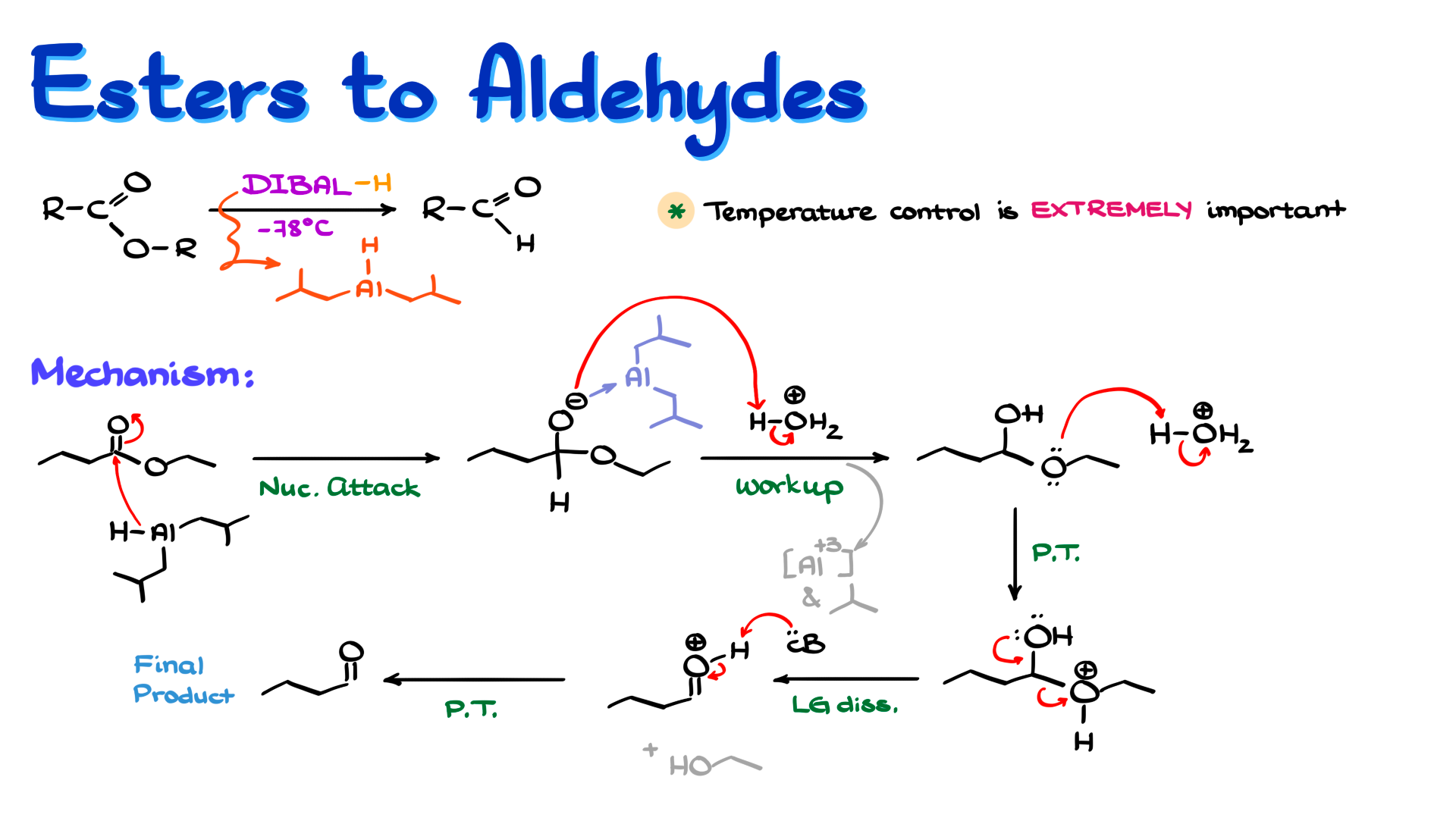
Here we run into a different problem. Esters are less reactive than aldehydes, which means that if we use a hydride reagent reactive enough to attack an ester, it will almost certainly be reactive enough to attack the aldehyde as well.
This is where DIBAL-H, diisobutylaluminum hydride, becomes very useful. It is reactive enough to reduce an ester, but it is so sterically hindered that the reaction can be stopped before overreduction occurs.
Let me show you what I mean through the mechanism. For this example I will use ethyl butanoate as my ester, and I will bring in DIBAL-H as the hydride source.
The first step, just like before, is nucleophilic attack of hydride on the carbonyl carbon. The π electrons go up onto oxygen, and we get the tetrahedral intermediate.
Again, aluminum coordinates to oxygen, just as in the previous example. In this case, though, that bond is even stronger because aluminum does not have extra substituents that can stabilize the negative charge elsewhere, so it really clings to oxygen. Because of this, and because ethoxide is not nearly as good a leaving group as chloride, the intermediate is relatively stable at low temperature, again typically around −78°C.
The next step is the acidic workup. Here the acidic workup does more than just remove aluminum. First, the negatively charged oxygen is protonated, giving an intermediate that is technically a hemiacetal. At the same time, the aluminum species breaks apart into Al³⁺ salts, and the isobutyl fragments are eventually lost as neutral byproducts.
Coming back to the hemiacetal intermediate, this species is not very stable. Since we are working under aqueous acidic conditions, it breaks down readily to the aldehyde.
That part of the mechanism is fairly straightforward. We bring in another equivalent of acid, protonate the alkoxy oxygen to make it into a better leaving group, and form the corresponding protonated intermediate. Then the leaving group departs, giving essentially a protonated aldehyde. Finally, whatever base is present, water, alcohol, or anything else with a lone pair, removes the proton and gives us the aldehyde.
As I mentioned, the most important thing about this reaction is strict temperature control. If you try to do this at a higher temperature, it will behave much more like a regular lithium aluminum hydride reduction, and you will get exhaustive reduction all the way to the primary alcohol instead.
In addition to the importance of temperature, we also need to remember that DIBAL-H is extremely pyrophoric. That means you have to be very careful when handling it, because if you mishandle it, it can easily start a fire. And fire in an organic chemistry lab is never good news.
Still, as long as you are careful, this reaction is well documented, works quite well, and generally gives decent yields. Overall, it is a very good method for making aldehydes from esters.
Weinreb Amide Synthesis
The final method I want to discuss is the partial reduction of Weinreb amides to aldehydes.
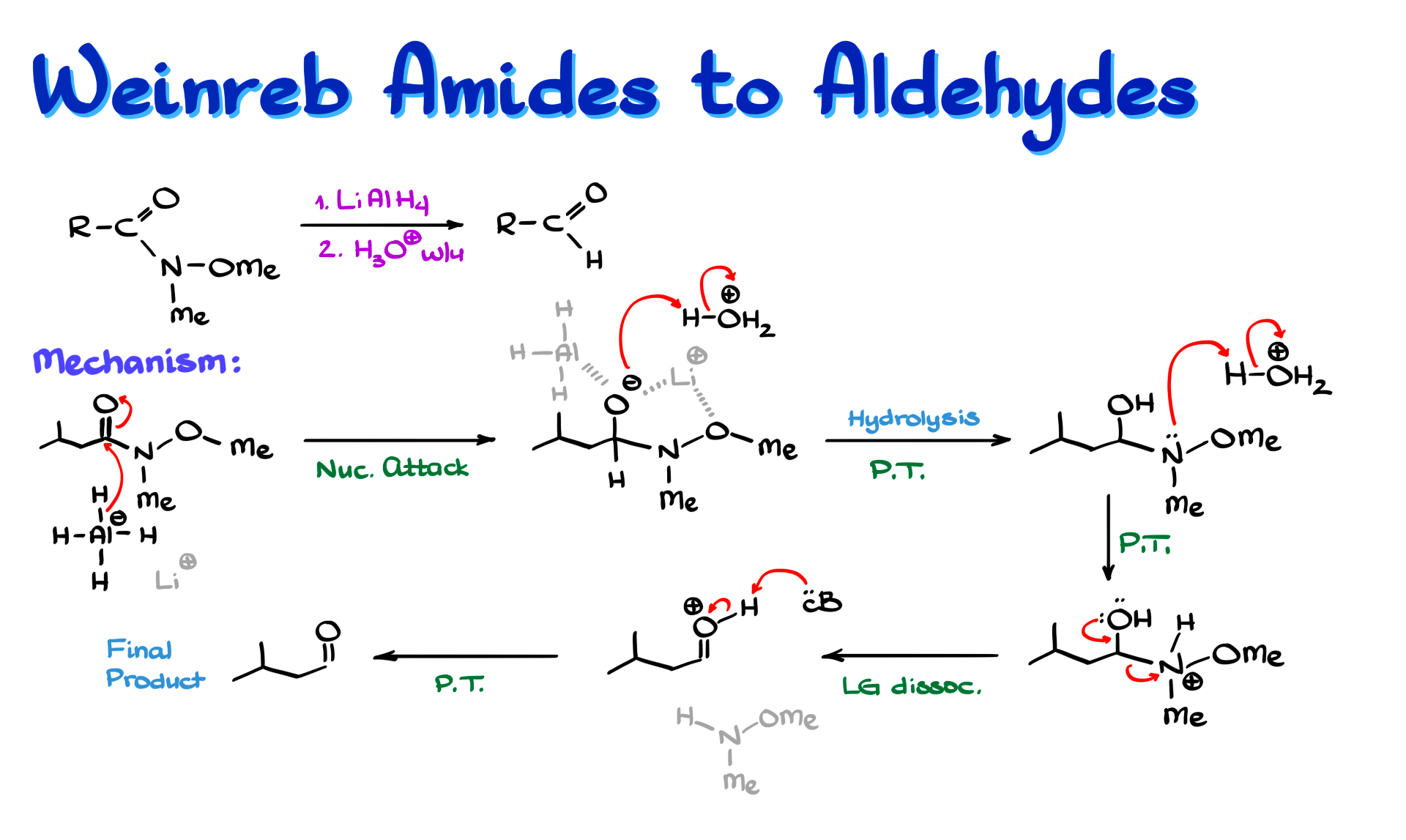
This is probably the best of all the methods I have described so far because Weinreb amides are fairly easy to make. On top of that, they can be reduced with regular lithium aluminum hydride, which is stable, commercially available, and easy to work with. That makes life much easier.
Exactly how we make Weinreb amides, and what else they can do, is a topic for another tutorial. For now, we will simply start with the Weinreb amide directly, and I will just mention that they are commonly prepared from acid chlorides or esters.
So let’s say we have this Weinreb amide. The first step, just like in the previous cases, is a simple hydride attack on the carbonyl. Hydride attacks the carbonyl carbon, and we form the tetrahedral intermediate.
In this case, lithium is actually important. Not only does aluminum coordinate to the oxygen, but lithium also coordinates to both oxygens in the intermediate, stabilizing the whole structure. Because of this double coordination, the Weinreb amide intermediate is very unlikely to collapse or expel the leaving group unless we raise the temperature or perform an acidic workup. These intermediates are fairly stable and can even be isolated in some cases.
If we want the final product, the aldehyde, we then bring in acid. The first step here is a simple proton transfer to give the corresponding protonated intermediate. From there we go through the usual hydrolysis-type steps. Another equivalent of acid protonates the nitrogen, giving a new intermediate in which the nitrogen substituent is now an excellent leaving group.
That leaving group is then expelled, giving us a protonated aldehyde. Finally, whatever base is present, water, amine, or anything else with a lone pair, removes the proton and gives us the aldehyde.
This method is very straightforward and extremely useful.
Recap
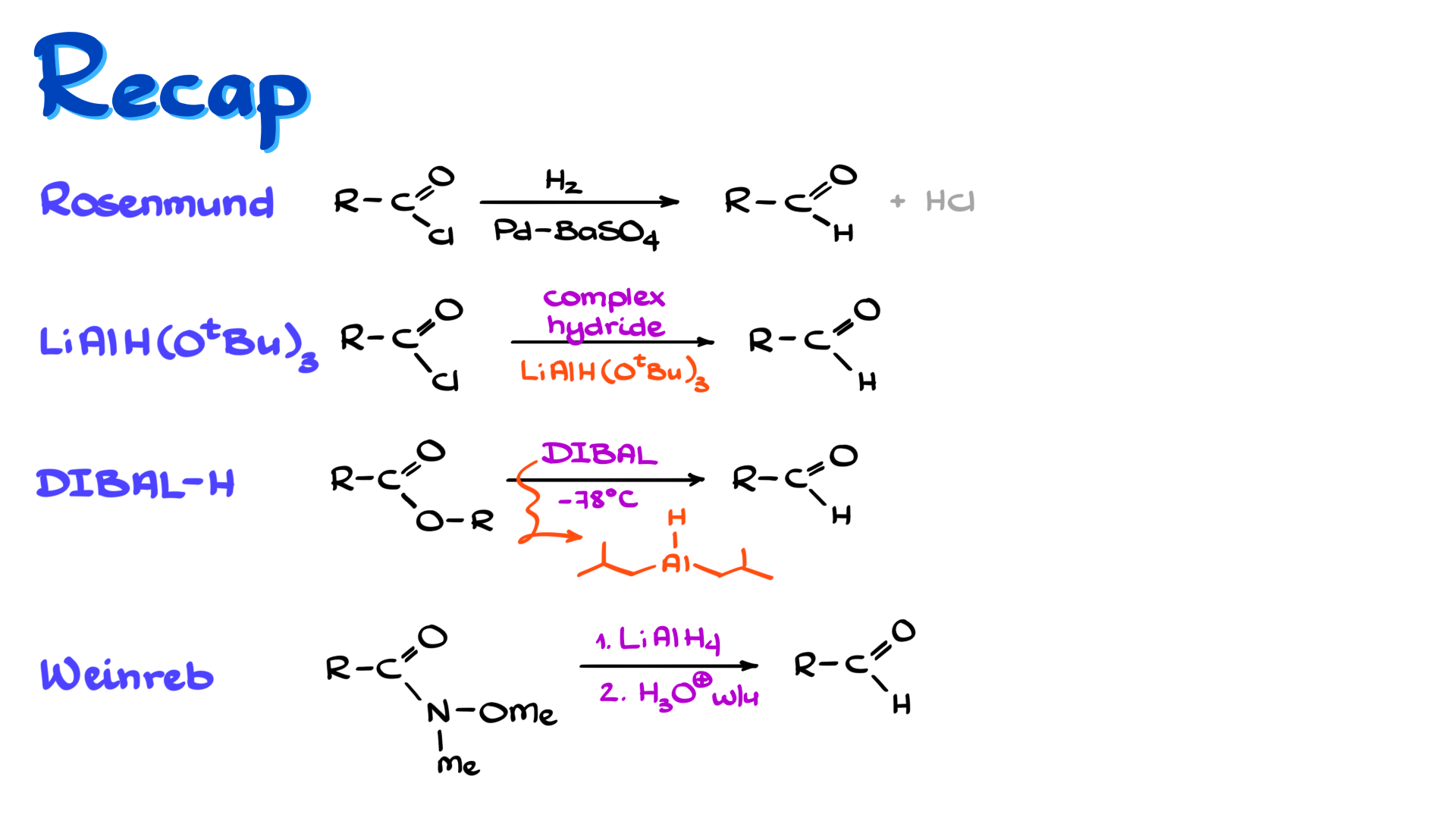
So, as a quick recap, the classic method for reducing carboxylic acid derivatives to aldehydes is the Rosenmund reduction, where we start with an acid chloride and reduce it with H₂ over a poisoned heterogeneous catalyst. As I mentioned, that reaction has a lot of drawbacks, and the biggest one is poor reproducibility, so we usually do not use it much in the lab.
If we want to start with acid chlorides in the lab, we typically use lithium tri-tert-butoxyaluminum hydride. If we want to start with esters, we generally use DIBAL-H. And finally, if we do not want to deal with acid chlorides or do not want to handle pyrophoric DIBAL-H, we can use Weinreb amide reduction with regular lithium aluminum hydride, which also gives us aldehydes.
So now you have yet another tool in your synthetic toolbox. The next time you need to synthesize an aldehyde, you will have more options to choose from.
