Staudinger Reaction
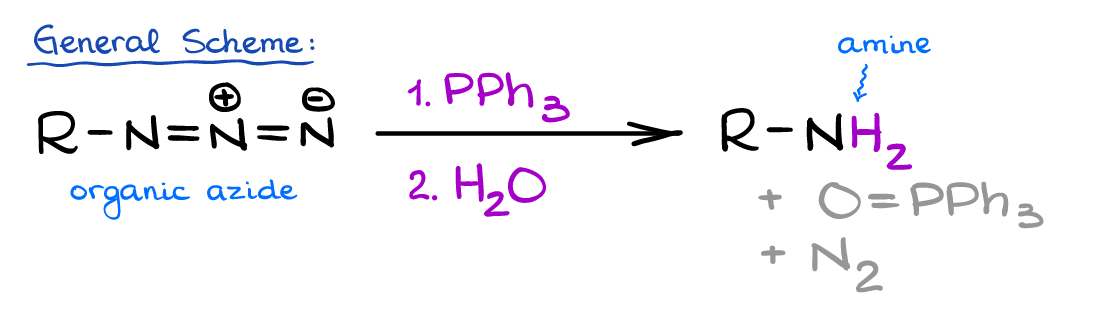
The Staudinger reaction is a mild reduction of organic azides yielding corresponding primary amines. The reaction has been developed by Hermann Staudinger in 1919 when he was a professor at the Swiss Federal Institute of Technology in Zürich.
In the Staudinger reaction the azide reacts with triphenylphosphine yielding the corresponding iminophosphorane intermediate similar to the Wittig ylide. This intermediate then quickly hydrolyzes in aqueous media yielding the final product:
Mechanistically, the first step follows typical for phosphorus nucleophilic attack by P leading to the formation of the 4-membered intermediate, which rapidly decomposes.
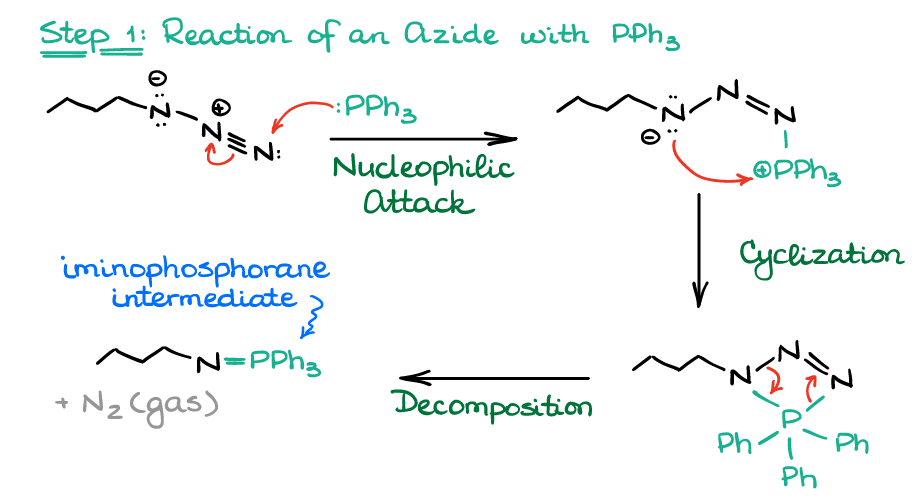
The decomposition of the 4-membered intermediate here is very favorable due to two reasons. First, the 4-membered ring is not very stable due to the angle strain. Second, the side-product of this decomposition is nitrogen gas, which is incredibly thermodynamically stable. Also, the formation of a gas in any reaction means that this reaction will be thermodynamically favorable due to the increase in the reaction entropy.
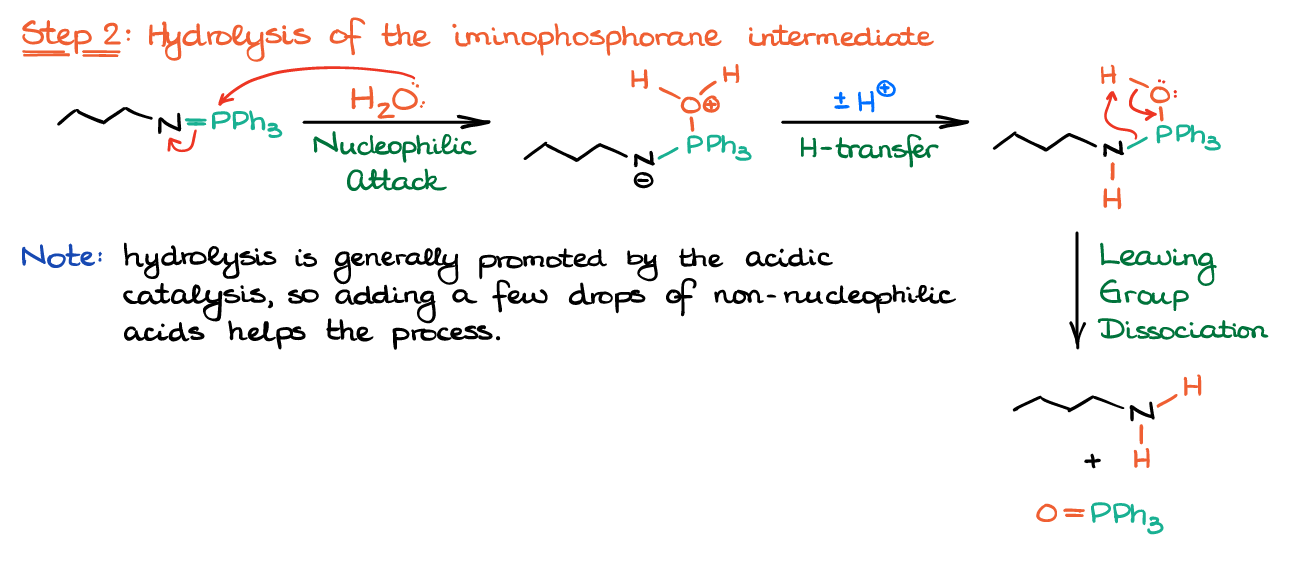
Once you have the iminophosphorane intermediate, you can react it with a number of nucleophiles. The most common ones are water giving the amine, or a carboxylic acid giving an amide (Staudinger-Vilarrasa reaction).
Examples of the Staudinger reaction and its uses
As I’ve mentioned earlier, this is an excellent way to synthesize primary amines:
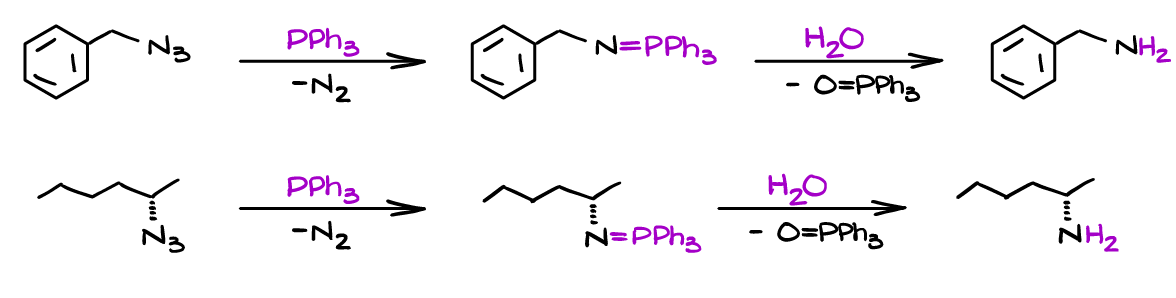
Alternatively, when you use a carboxylic acid, or the acid is a part of the molecule itself you can form corresponding amides or lactams:
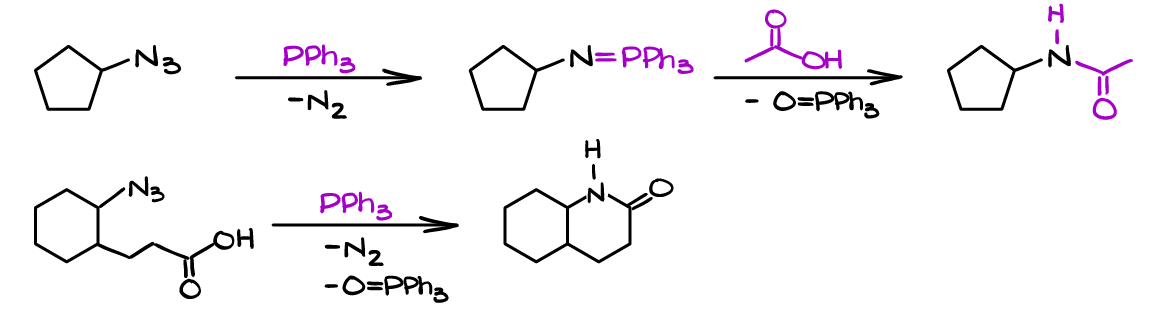
Synthetic applications of the Staudinger Reaction
This reduction method is especially useful in the presence of other functional groups. For instance, look at the following reaction:
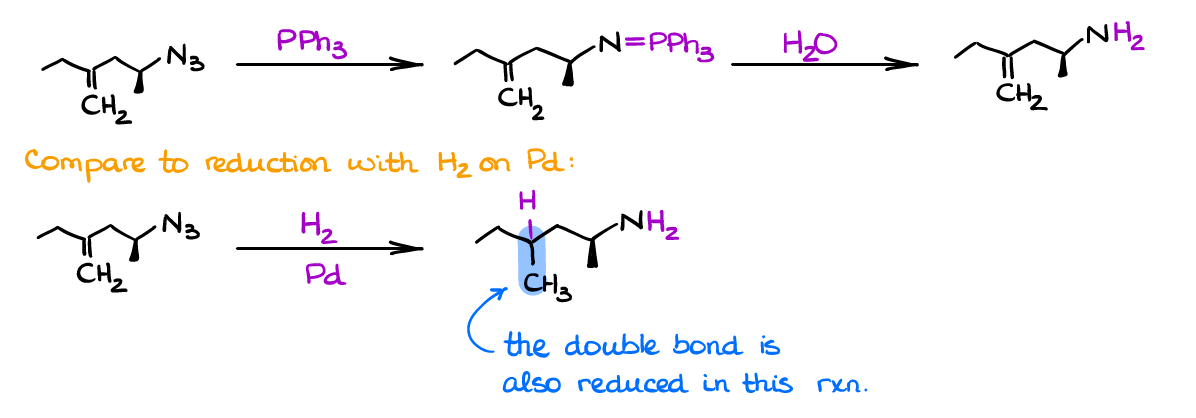
Reduction of azides with hydrogen gas on a heterogeneous catalyst is a common practice. It’s cheap and effective! BUT! If you have double bonds in your compound, then you’re out of luck as you’ll reduce all. Same problem you may see in examples with an aldehyde or a ketone functional groups that can be reduced by hydrogen on palladium catalyst or by complex hydrides that are typically used for the reduction of azides. So, in all those situations when you cannot use your “common” reduction techniques, the Staudinger may be particularly useful.
You can easily introduce an azide functional group into your molecule via a simple SN2 reaction of an alkyl halide with sodium or potassium azide. I would suggest, however, to choose your starting material carefully as the SN2 reaction will cause the inversion of the carbon configuration which may or may not be relevant in your example.
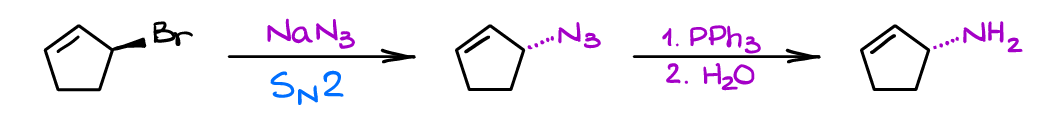
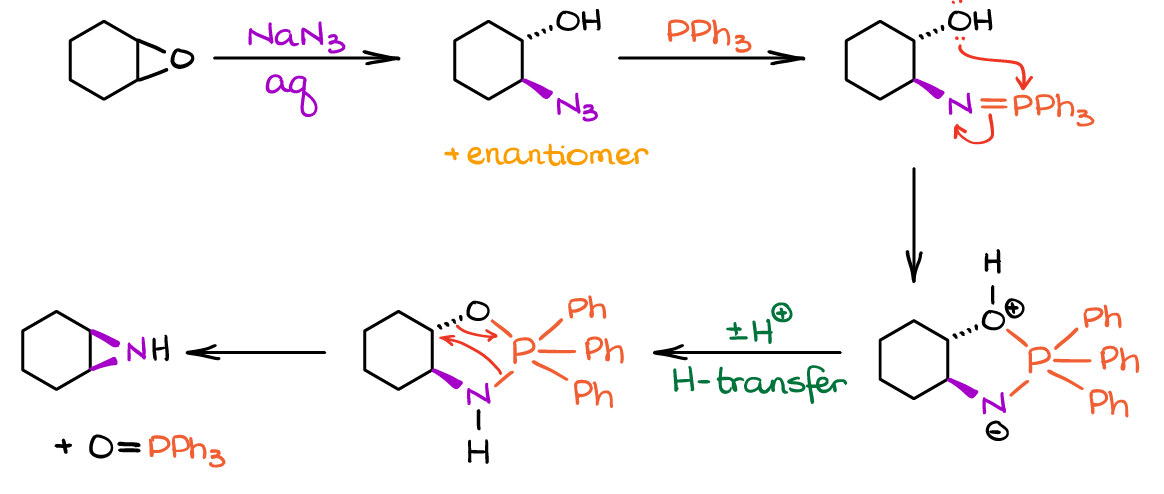
Finally, it’s important to keep in mind the presence of other nucleophilic groups in the molecule. While the Staudinger reactions tolerates many functional groups, it is quite sensitive to oxygen-containing nucleophiles like alcohols or phenols. As phosphorous has high affinity towards oxygen, those -OH nucleophiles may “trap” the intermediate and prevent the second step from happening or divert it to another path. For instance, the adjacent vicinal -OH would give you a 3-membered ring with nitrogen (aziridine) instead of the intended amine: